

Chinese Bulletin of Botany ›› 2023, Vol. 58 ›› Issue (3): 440-448.DOI: 10.11983/CBB22221 cstr: 32102.14.CBB22221
• TECHNIQUES AND METHODS • Previous Articles Next Articles
Yefei Liu1,2, Haixia Zhao2, Xiping Jiang2, Rui Qiu1,2, Xinyue Zhou1, Yan Zhao1( ), Chunxiang Fu2(
), Chunxiang Fu2( )
)
Received:2022-09-13
Accepted:2022-11-15
Online:2023-05-01
Published:2023-05-17
Contact:
*E-mail: zhaoyannmg@163.com; fucx@qibebt.ac.cn
Yefei Liu, Haixia Zhao, Xiping Jiang, Rui Qiu, Xinyue Zhou, Yan Zhao, Chunxiang Fu. Establishment of Highly Efficient Tissue Culture and Agrobacterium-mediated Callus Infection Systems for Hordeum brevisubulatum[J]. Chinese Bulletin of Botany, 2023, 58(3): 440-448.
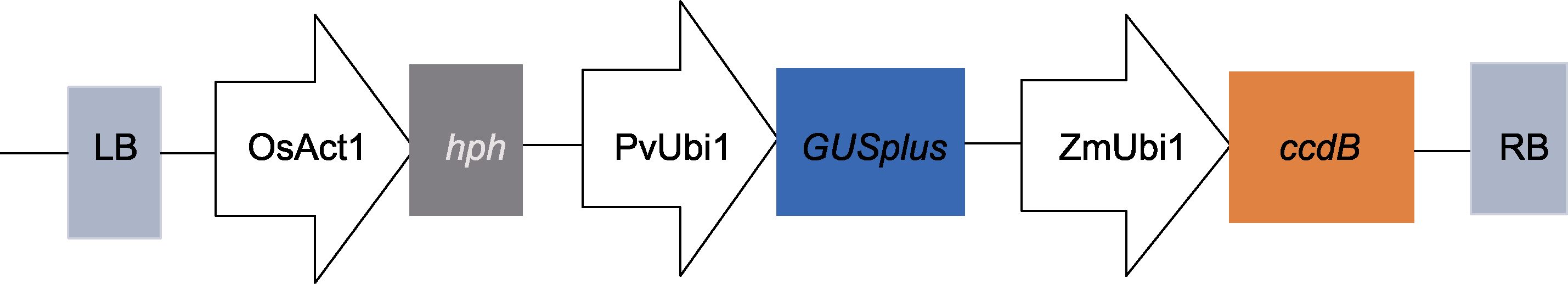
Figure 1 Schematic diagram of PANIC6D(B) carrier LB: Left border of T-DNA; OsAct1: Rice promoter; hph: Screening marker gene for hygromycin resistance; PvUbi1: Switchgrass promoter; GUSplus: Glucosidase reporter gene; ZmUbi1: Maize promoter; ccdB: Lethal gene; RB: Right border of T-DNA
| Disinfection method | Disinfection time | Seed germination induction rate | Pollution rate | |
|---|---|---|---|---|
| A (HgCl2) | 15 min | (61.3±4.0)%ab | (4.0±3.6)%b | |
| B (NaClO) | 15 min | (65.7±3.1)%a | (5.3±1.5)%b | |
| C (H2O2) | C-1 | 4 h | (52.3±4.0)%b | (33.3±1.5)%a |
| C-2 | 8 h | (34.0±2.7)%bc | (4.3±1.5)%b | |
| C-3 | 12 h | (20.1±3.1)%d | (1.3±1.5)%b | |
Table 1 Effect of different sterilization methods on seed induction rate of Hordeum brevisubulatum (means±SE)
| Disinfection method | Disinfection time | Seed germination induction rate | Pollution rate | |
|---|---|---|---|---|
| A (HgCl2) | 15 min | (61.3±4.0)%ab | (4.0±3.6)%b | |
| B (NaClO) | 15 min | (65.7±3.1)%a | (5.3±1.5)%b | |
| C (H2O2) | C-1 | 4 h | (52.3±4.0)%b | (33.3±1.5)%a |
| C-2 | 8 h | (34.0±2.7)%bc | (4.3±1.5)%b | |
| C-3 | 12 h | (20.1±3.1)%d | (1.3±1.5)%b | |
| 2,4-D concen- tration (mg·L-1) | Seed induction rate | Callus status |
|---|---|---|
| 2 | (74.4±5.4)%a | Loose and transparent vitrification callus |
| 5 | (65.0±3.6)%b | Friable embryogenic callus with watery outer layer |
| 7 | (31.1±6.6)%c | Compact and translucent watery callus |
Table 2 Effect of different 2,4-D concentrations on seed callus induction rate of Hordeum brevisubulatum (means±SE)
| 2,4-D concen- tration (mg·L-1) | Seed induction rate | Callus status |
|---|---|---|
| 2 | (74.4±5.4)%a | Loose and transparent vitrification callus |
| 5 | (65.0±3.6)%b | Friable embryogenic callus with watery outer layer |
| 7 | (31.1±6.6)%c | Compact and translucent watery callus |

Figure 2 Seed callus status of Hordeum brevisubulatum induced by different concentrations of 2,4-D (2 mg·L-1 (A), 5 mg·L-1 (B) and 7 mg·L-1 (C)) (Bars=5 mm)

Figure 4 Differentiation and rooting of embryogenic callus of Hordeum brevisubulatum (A) 15 days’ culture in light; (B) 25 days’ culture in light; (C) 40 days’ culture in light; (D) Rooting; (E) Transplanting
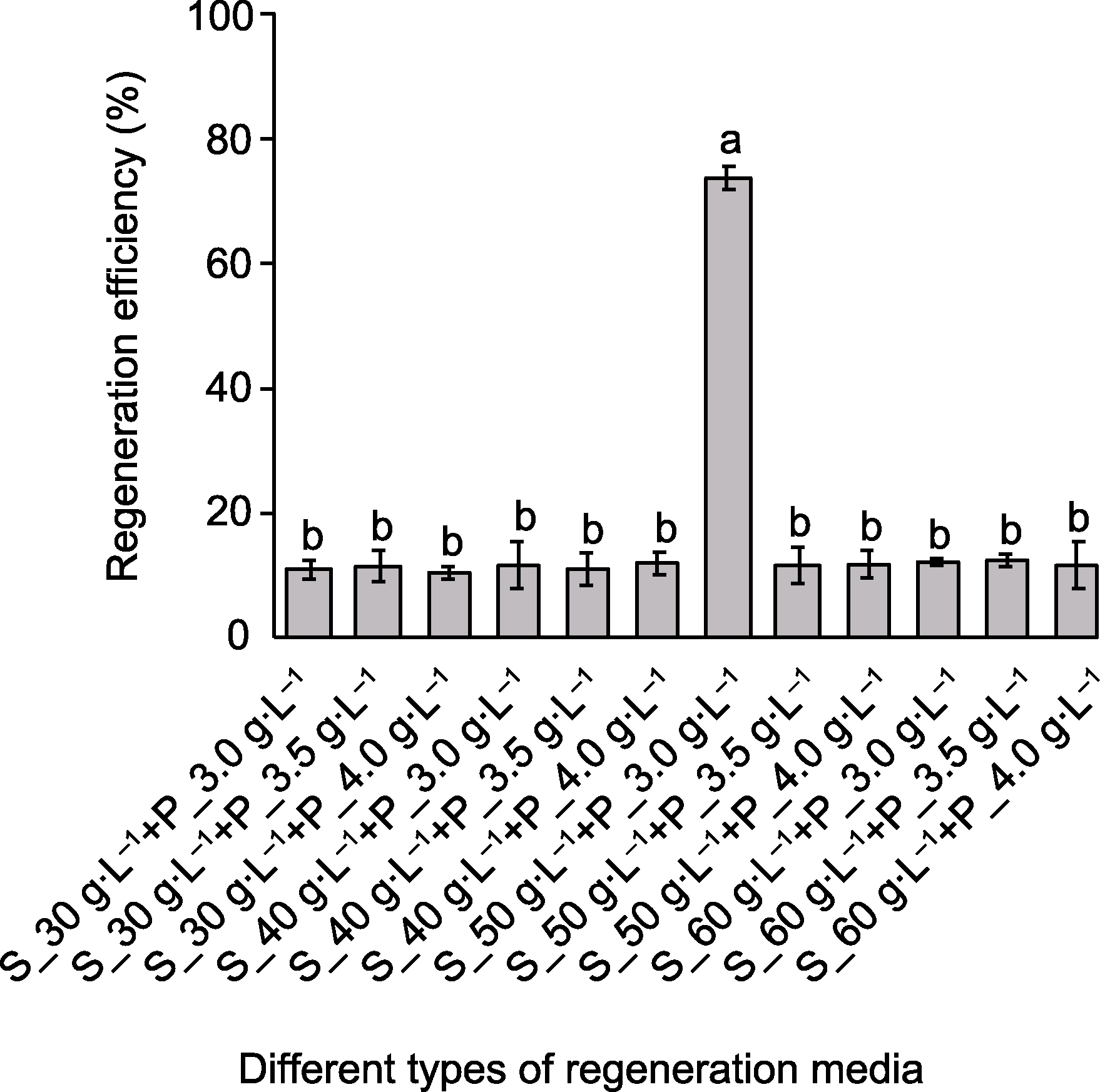
Figure 5 Screening of regeneration medium for Hordeum brevisubulatum The horizontal axis represents different types of regeneration medium. The medium composition includes MS+2 mg·L-1 2,4-D+0.05 mg·L-1 KT, and supplemented with different levels of sucrose (30, 40, 50 and 60 g·L-1) and phytagel (3.0, 3.5 and 4.0 g·L-1). S: Sucrose; P: Phytagel. Different lowercase letters indicate significant differences among different treatments (One-way ANOVA, Duncan’s test, P<0.05).
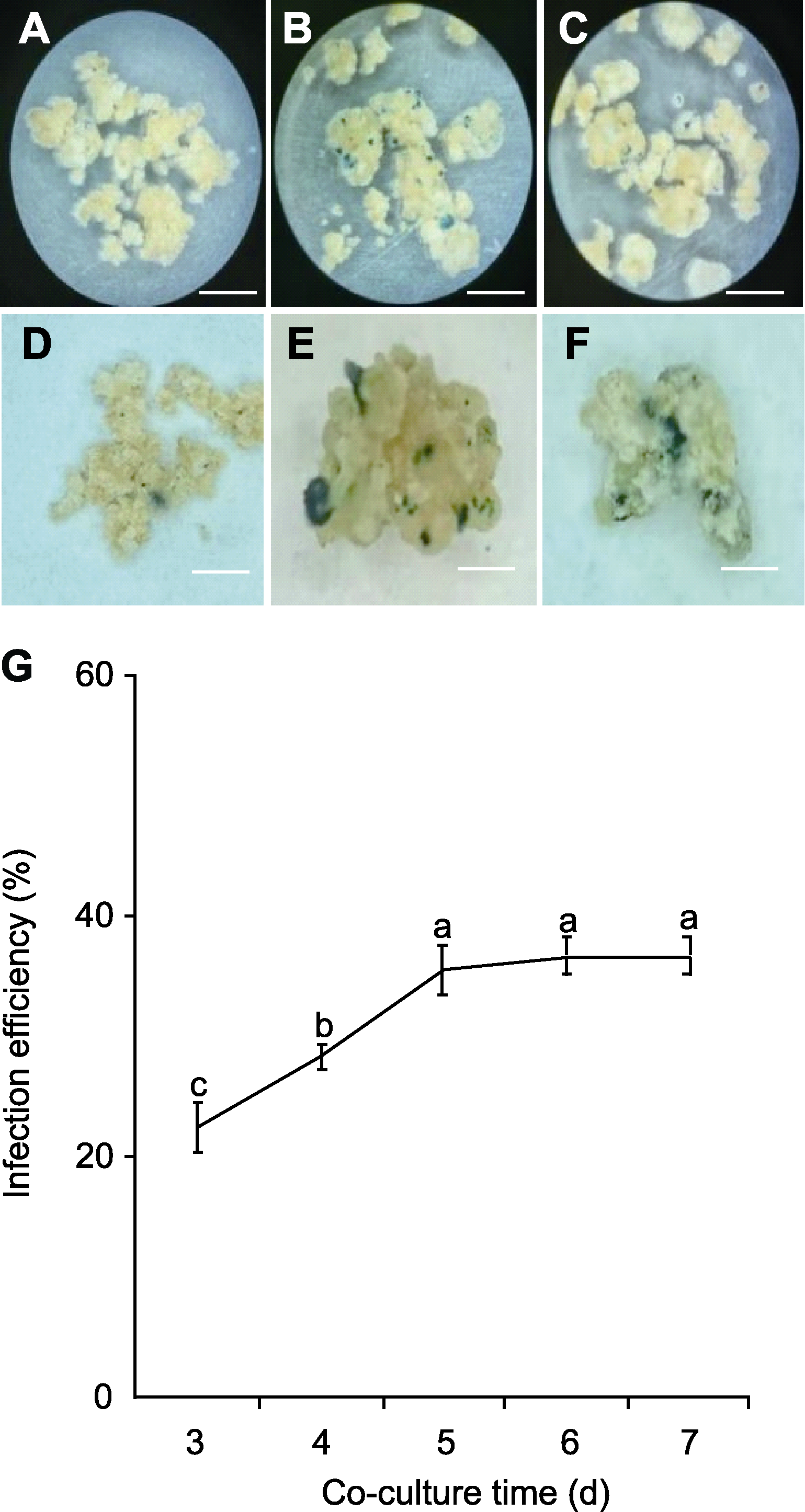
Figure 6 Effect of co-culture time of Agrobacterium tumefaciens on infection efficiency (A)-(C) GUS staining of co-culture for 3, 4 and 5 days; (D)- (F) Enlarged view of GUS staining positive calli in (A), (B) and (C); (G) The relationship between infection efficiency and co-culture time. Different lowercase letters indicate significant differences among different treatments (One-way ANOVA, Duncan’s test, P<0.05). Bars=100 μm
| [1] | 蔡联炳, 郭本兆 (1988). 中国大麦属的演化与地理分布的探讨. 西北植物学报 (02), 73-84. |
| [2] | 程肖蕊, 张亚兰, 杨松涛, 李彦舫 (1997). 野大麦幼根的愈伤组织诱导及植株再生. 吉林农业科学 (02), 94-96. |
| [3] | 丁雪梅, 何汉琼, 张英, 李玉梅, 赵云, 成军, 饶家辉, 王宏娟, 沈景林 (2010). 野大麦种子萌发条件的研究. 东北农业大学学报 41, 11-16. |
| [4] | 杜雪玲 (2004). 多年生黑麦草和结缕草再生体系及以农杆菌为介导的抗草甘瞵遗传转化体系建立的研究. 硕士论文. 兰州: 甘肃农业大学. pp. 11-17. |
| [5] | 付凤玲, 李晚忱, 刘玉贞 (1999). 玉米幼穗培养及植株再生. 四川农业大学学报 (3), 278-281. |
| [6] | 李红, 杨允菲, 张成武 (2000). 松嫩平原碱化草甸野大麦无性系构建的定量分析. 草业学报 (4), 13-19. |
| [7] | 李庆华 (2021). 裸燕麦转基因体系的建立与优化. 硕士论文. 呼和浩特: 内蒙古农业大学. pp. 4-7. |
| [8] |
梁流芳, 佘建明, 吴瑛瑛 (2008). 海雀稗幼穗离体培养植株再生. 草地学报 (6), 590-593.
DOI |
| [9] | 罗雪梅, 金晓玲, 王征, 刘雪梅 (2012). 组培微环境对再生苗生长的影响. 经济林研究 30, 141-144. |
| [10] | 权军利, 刘正全, 陈耀锋, 韩德俊, 李春莲, 任慧莉 (1999). 蔗糖与激素对小麦幼穗体细胞无性系形成及生长特性的影响研究. 西北植物学报 19(6), 87-91. |
| [11] | 石德成, 奚惕 (1988). 野大麦幼叶组织培养及植株再生. 四川草原 (3), 13-15. |
| [12] | 唐静仪 (2010). 蔗糖和ABA对铁皮石斛体细胞胚胎发生的影响. 硕士论文. 成都: 西南交通大学. pp. 3-5. |
| [13] | 王春梅, 张茜, 张怀山, 汪晓斌, 朱新强, 夏曾润, 王晓力 (2013). 野大麦耐盐性研究进展. 中国草食动物科学 33, 48-52. |
| [14] | 王伟 (2020). 玉米高效遗传转化受体材料的筛选. 硕士论文. 泰安: 山东农业大学. pp. 8-20. |
| [15] | 杨海营 (2016). 玉米芽再生能力调控基因的全基因组关联分析. 硕士论文. 泰安: 山东农业大学. pp. 7-10. |
| [16] | 云玲格, 李造哲, 马青枝, 谢菲, 李月强 (2016). 披碱草×野大麦杂种F1幼穗培养再生体系的建立. 中国草地学报 38, 14-18. |
| [17] | 张立营, 李喜文, 许红力, 庞晓斌, 李彦舫 (2004). 野大麦组织培养及植株再生的研究. 内蒙古民族大学学报(自然科学版) (3), 290-292. |
| [18] | 张亚兰, 程肖蕊, 李彦舫 (1997). 野大麦幼叶组织培养. 植物生理学通讯 (5), 359. |
| [19] | 赵智燕, 潘俊松, 何亚丽, 王琛, 闫军辉 (2009). 两个高羊茅无性系的营养器官组织培养及再生体系的建立. 草业学报 18, 168-175. |
| [20] | 朱志国, 黄承钧, 陶陶, 郭丽 (2006). 红叶石楠组培增殖技术研究. 安徽农业科学 (15), 3668, 3691. |
| [21] |
Feng ZY, Zhang BT, Ding WN, Liu XD, Yang DL, Wei PL, Cao FQ, Zhu SH, Zhang F, Mao YF, Zhu JK (2013). Ef-ficient genome editing in plants using a CRISPR/Cas system. Cell Res 23, 1229-1232.
DOI |
| [22] |
Mann DG, Lafayette PR, Abercrombie LL, King ZR, Maza-rei M, Halter MC, Poovaiah CR, Baxter H, Shen H, Dixon RA, Parrott WA, Neal Stewart C Jr (2012). Gate-way-compatible vectors for high-throughput gene functio-nal analysis in switchgrass (Panicum virgatum L.) and other monocot species. Plant Biotechnol J 10, 226-236.
DOI URL |
| [23] |
Mao YF, Zhang H, Xu NF, Zhang BT, Gao F, Zhu JK (2013). Application of the CRISPR-Cas system for efficient genome engineering in plants. Mol Plant 6, 2008-2011.
DOI PMID |
| [24] |
Vasil IK (1994). Molecular improvement of cereals. Plant Mol Biol 25, 925-937.
DOI PMID |
| [1] |
Tong Li, Churan Li, Zhiyu Zhang, Xiaoman Fu, Yun Liu, Yingjun Zhang, Liying Yang, Ping Zhao.
A Preliminary Study on Tissue Culture and Rapid Propagation Technology of Phyllanthus acidus [J]. Chinese Bulletin of Botany, 2025, 60(4): 1-0. |
| [2] | Wen Feng, Yuguo Wang. Establishment of an In Vitro Regeneration System for Stem Segments of Cultivated Dioscorea polystachya [J]. Chinese Bulletin of Botany, 2024, 59(5): 792-799. |
| [3] | Yuze Liu, Yifei Wang, Weizhen Ren, Hao Li, Bin Lu, Bingshe Lu, Xiaoyue Yu. Establishment of Immature Embryo Rescue and Regeneration System for Pyrus calleryana cv. ‘Cleveland’ [J]. Chinese Bulletin of Botany, 2024, 59(5): 800-809. |
| [4] | Hao Zeng, Peifang Li, Zhihui Guo, Chunlin Liu, Ying Ruan. Establishment of a Regeneration System for Lunaria annua [J]. Chinese Bulletin of Botany, 2024, 59(3): 433-440. |
| [5] | Shangwen Zhang, Shiyu Huang, Tianwei Yang, Ting Li, Xiangjun Zhang, Manrong Gao. Establishment of a Tissue Culture and Rapid Propagation System for Erythropalum scandens Based on Orthogonal Test [J]. Chinese Bulletin of Botany, 2024, 59(1): 99-109. |
| [6] | Chungang Xie, Zhe Liu, Shusheng Zhang, Haitao Hu. Establishment of In Vitro Regeneration System of Citrus australasica [J]. Chinese Bulletin of Botany, 2023, 58(6): 926-934. |
| [7] | Jinchun Lu, Lina Cao, Guanjie Tong, Xinying Wang, Liying Zhang, Xin Yu, Huifang Li, Yanhui Li. Establishment of Callus Induction and Regeneration System of Anemone silvestris [J]. Chinese Bulletin of Botany, 2022, 57(2): 217-226. |
| [8] | Mengyue Li, Liu Liu, Yan Liu, Xiaoman Zhang. Establishment of Tissue Culture System for Axillary Bud Regeneration of Primula × pubescens [J]. Chinese Bulletin of Botany, 2021, 56(6): 732-739. |
| [9] | Qian Luo, Yansha Zhang, Jing Ou. Callus Induction and Plant Regeneration of Cerasus serrulata var. lannesiana cv. ‘Grandiflora’ [J]. Chinese Bulletin of Botany, 2021, 56(4): 451-461. |
| [10] | Pengfei Du, Yu Wang, Yingping Cao, Song Yang, Zhichao Sun, Decai Mao, Jiajun Yan, Daxu Li, Meizhen Sun, Chunxiang Fu, Shiqie Bai. Establishment of Biolistic Mediated Transformation System for Elymus sibiricus [J]. Chinese Bulletin of Botany, 2021, 56(1): 62-70. |
| [11] | Sha Deng, Yanni Wu, Kunlin Wu, Lin Fang, Lin Li, Songjun Zeng. Breeding characteristics and artificial propagation of 14 species of Wild Plant with Extremely Small Populations (WPESP) in China [J]. Biodiv Sci, 2020, 28(3): 385-400. |
| [12] | Hong Luo, Xiaohui Wen, Yuanyuan Zhou, Silan Dai. Establishment of In Vitro Regeneration System of Helenium aromaticum [J]. Chinese Bulletin of Botany, 2020, 55(3): 318-328. |
| [13] | Yan Xiao,Zhenxing Wang,Dongming Li,Yanhua Qi, Enhebayaer. Optimization of Tissue Culture and Plant Regeneration System of Mature Embryo of Leymus chinensis [J]. Chinese Bulletin of Botany, 2020, 55(2): 192-198. |
| [14] | Wenting Zhang,Yanhong He,Ning Shu,Jingjing Xing,Baojun Liu,Manzhu Bao,Guofeng Liu. Plant Regeneration and Rapid Propagation System of Lilium bakerianum var. aureum [J]. Chinese Bulletin of Botany, 2019, 54(6): 773-778. |
| [15] | Fengluan Tang,Jian Zhao,Zhiguo Zhao,Ke Xia,Shuo Qiu. Tissue Culture and Rapid Propagation of Ardisia gigantifolia [J]. Chinese Bulletin of Botany, 2019, 54(3): 378-384. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||