

Chinese Bulletin of Botany ›› 2019, Vol. 54 ›› Issue (4): 486-496.DOI: 10.11983/CBB18139 cstr: 32102.14.CBB18139
• EXPERIMENTAL COMMUNICATIONS • Previous Articles Next Articles
Yegeng Fan1,Lihang Qiu1,Xing Huang1,Huiwen Zhou1,Chongkun Gan2,Yangrui Li1,Rongzhong Yang1,Jianming Wu1,*( ),Rongfa Chen1,*(
),Rongfa Chen1,*( )
)
Received:2018-06-19
Accepted:2018-12-10
Online:2019-07-01
Published:2020-01-08
Contact:
Jianming Wu,Rongfa Chen
Yegeng Fan,Lihang Qiu,Xing Huang,Huiwen Zhou,Chongkun Gan,Yangrui Li,Rongzhong Yang,Jianming Wu,Rongfa Chen. Expression Analysis of Key Genes in Gibberellin Biosynthesis and Related Phytohormonal Dynamics During Sugarcane Internode Elongation[J]. Chinese Bulletin of Botany, 2019, 54(4): 486-496.
| Genes | Sequences of primers (5'-3') | Tm (°C) | Product length (bp) |
|---|---|---|---|
| 25S rRNA | F: CGAATACGAACCGTGAAAGC | 58.4 | 164 |
| R: ACAATGATAGGAAGAGCCGACA | 59.0 | ||
| GA20-Oxidase1 | F: CCGTCGTGGACTACTTCG | 54.0 | 205 |
| R: CACGGCGGGTAGTAGTTGAG | 58.3 | ||
| GID1 | F: GGTCATCCTCTTCTTCCACGG | 60.8 | 119 |
| R: AGTTGACGGACACCACGACG | 61.3 | ||
| GAI | F: GCGACGACAAGGATGAGGA | 58.8 | 186 |
| R: TGAAGCGGTCAAGGAATGTG | 59.2 |
Table 1 Primers for real-time quantitative PCR
| Genes | Sequences of primers (5'-3') | Tm (°C) | Product length (bp) |
|---|---|---|---|
| 25S rRNA | F: CGAATACGAACCGTGAAAGC | 58.4 | 164 |
| R: ACAATGATAGGAAGAGCCGACA | 59.0 | ||
| GA20-Oxidase1 | F: CCGTCGTGGACTACTTCG | 54.0 | 205 |
| R: CACGGCGGGTAGTAGTTGAG | 58.3 | ||
| GID1 | F: GGTCATCCTCTTCTTCCACGG | 60.8 | 119 |
| R: AGTTGACGGACACCACGACG | 61.3 | ||
| GAI | F: GCGACGACAAGGATGAGGA | 58.8 | 186 |
| R: TGAAGCGGTCAAGGAATGTG | 59.2 |
| Period | Plant height (cm) | Elongation rate (cm·d-1) | Internode number | Average internode length (cm) |
|---|---|---|---|---|
| Ls1 | 39.38±1.10 | 0.44±0.01 | 4±1 | 6.09±0.28 |
| Ls2 | 68.37±1.25** | 0.97±0.07** | 7±1** | 8.05±0.18** |
| Ls3 | 147.13±0.85** | 2.63±0.06** | 12±1** | 11.43±0.07** |
Table 2 The elongation of sugarcane at different stages (means±SD)
| Period | Plant height (cm) | Elongation rate (cm·d-1) | Internode number | Average internode length (cm) |
|---|---|---|---|---|
| Ls1 | 39.38±1.10 | 0.44±0.01 | 4±1 | 6.09±0.28 |
| Ls2 | 68.37±1.25** | 0.97±0.07** | 7±1** | 8.05±0.18** |
| Ls3 | 147.13±0.85** | 2.63±0.06** | 12±1** | 11.43±0.07** |

Figure 1 Changes of sugarcane elongation at different stages (A) The sugarcane plant height of non-elongation stage (Ls1); (B) The sugarcane plant height of early elongation stage (Ls2); (C) The sugarcane plant height of elongation stage (Ls3). Bars=10 cm
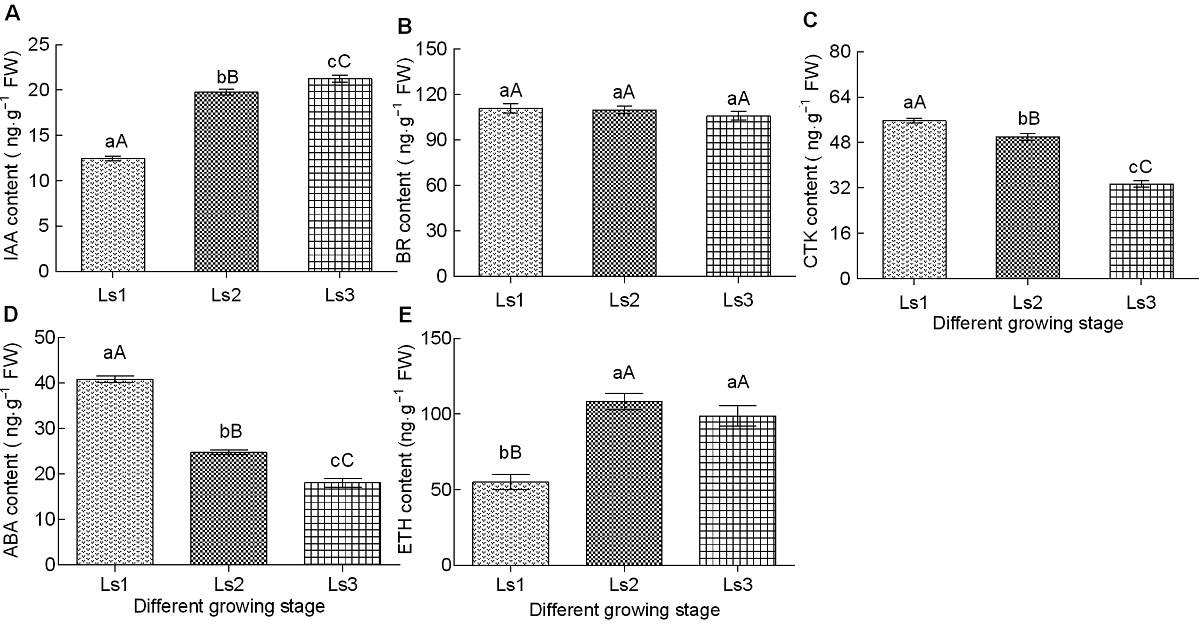
Figure 2 Changes of endogenous hormone contents in sugarcane internode during different elongation stages (A) Indole acetic acid (IAA); (B) Brassinosteroids (BR); (C) Cytokinin (CTK); (D) Abscisic acid (ABA); (E) Ethylene (ETH). The different lowercase letters indicate significant differences (P<0.05), and the different capital letters indicate extremely significant differences (P<0.01). Ls1-Ls3 see Table 2.
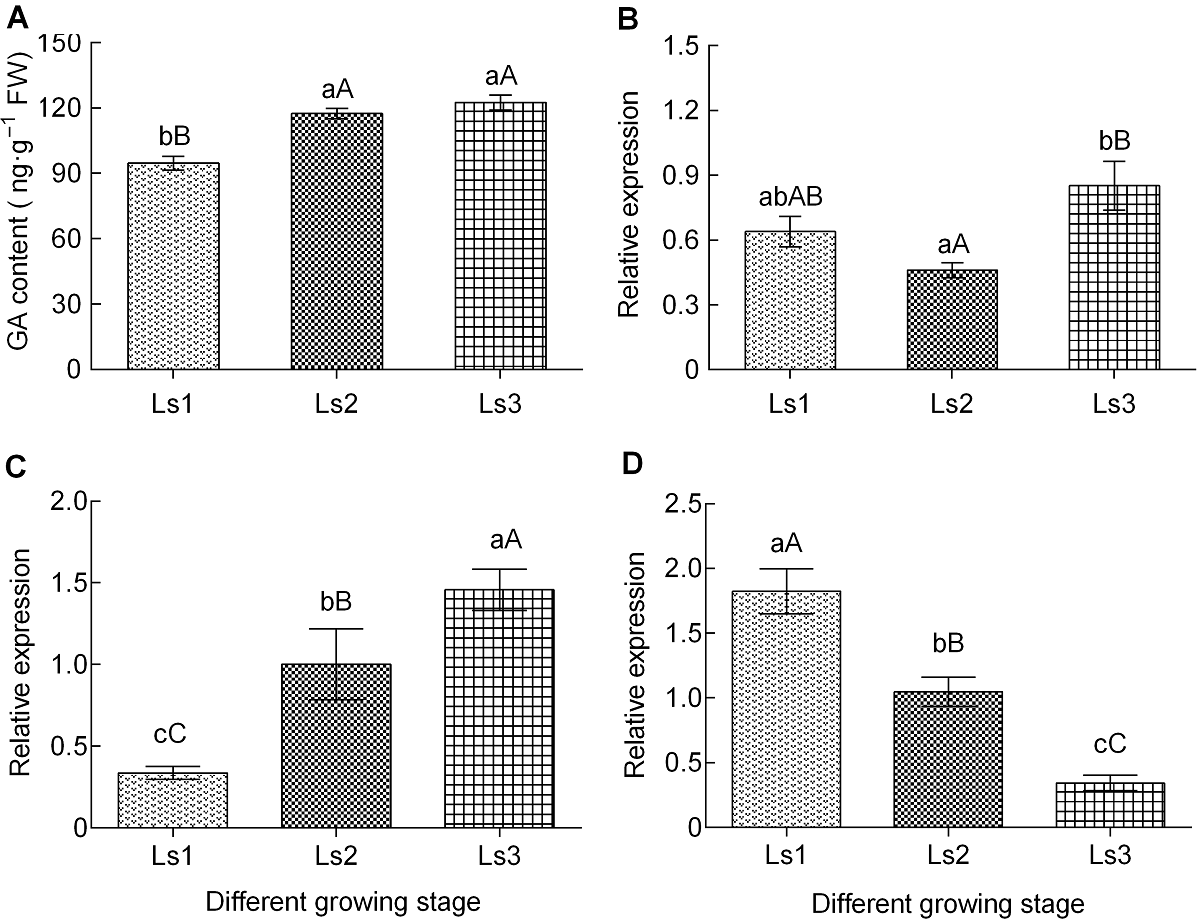
Figure 3 The changes of gibberellin content and genes differential expression of synthetic pathways during sugarcane internode elongation (A) The content of gibberellic acid (GA); (B) The expression level of GA20-Oxidase1; (C) The expression level of GID1; (D) The expression level of GAI. The different lowercase letters indicate significant differences (P<0.05), and the different capital letters indicate extremely significant differences (P<0.01). Ls1-Ls3 see Table 2.
| [1] | 陈登举, 高培军, 吴兴波, 高岩, 温国胜, 王玉魁, 高荣孚, 张汝民 ( 2013). 毛竹茎秆叶绿体超微结构及其发射荧光光谱特征. 植物学报 48, 635-642. |
| [2] | 陈明辉, 黄杏, 谢晓娜, 杨丽涛, 李杨瑞, 陈保善 ( 2013). 宿根矮化病菌对甘蔗生长和内源激素的影响. 华中农业学报 32(3), 57-61. |
| [3] | 陈宇杰, 刘飞, 梁菲菲, 石君, 赵焕梦 ( 2017). 蓖麻DELLA蛋白家族GAI基因克隆、表达及生物信息学分析. 内蒙古民族大学学报(自然科学版) 32, 320-327. |
| [4] | 刁家连, 何钟佩 ( 1998). 冬小麦茎秆生长过程中内源激素动态的系统研究. 中国农业大学学报 3(增刊), 16-20. |
| [5] | 丁兴萃 ( 1997). 毛竹笋体生长发育过程中内源激素的动态分析. 竹子研究汇刊 16(2), 53-62. |
| [6] | 董静, 尹梦回, 杨帆, 赵娟, 覃珊, 侯磊, 罗明, 裴炎, 肖月华 ( 2009). 棉花赤霉素不敏感矮化GID1同源基因的克隆和表达分析. 作物学报 35, 1822-1830. |
| [7] | 杜维, 丁健, 阮成江 ( 2018). 沙棘果实发育过程中内源激素水平的动态变化. 植物学报 53, 219-226. |
| [8] | 段娜, 贾玉奎, 徐军, 陈海玲, 孙鹏 ( 2015). 植物内源激素研究进展. 中国农学通报 31(2), 159-165. |
| [9] | 方楷, 杨光耀, 杨清培, 黄俊宝, 施建敏, 于芬 ( 2011). 毛竹成竹过程中内源激素动态变化. 江西农业大学学报 33, 1107-1111. |
| [10] | 冯丹, 陈贵林 ( 2011). 独脚金内酯调控侧枝发育的研究进展. 生态学杂志 30, 349-356. |
| [11] | 冯孟杰, 徐恒, 张华, 朱英 ( 2015). 茉莉素调控植物生长发育的研究进展. 植物生理学报 51, 407-412. |
| [12] | 甘小洪, 丁雨龙 ( 2004). 毛竹茎秆纤维发育过程的超微结构观察. 植物学通报 21, 180-188. |
| [13] | 甘小洪, 唐翠彬, 温中斌, 丁雨龙 ( 2013). 寿竹笋的营养成分研究. 天然产物研究与开发 25, 494-499. |
| [14] | 郭光生, 曾炳山, 范春节, 裘珍飞 ( 2018). 外施GA3对巨桉木质部发育的影响. 分子植物育种 16, 602-606. |
| [15] | 季兰, 杨仁崔 ( 2002). 水稻茎伸长生长与植物激素. 植物学通报 19, 109-115. |
| [16] | 江雪, 楼崇, 袁娜, 岳晋军, 顾小平 ( 2016). 外源GA3对毛竹实生苗新分蘖竹株秆形与竹材纤维质量的影响. 南京林业大学学报(自然科学版) 40(2), 121-126. |
| [17] | 姜勇, 胡尚连, 曹颖, 卢学琴, 黄艳, 徐刚 ( 2017). 外源GA3对毛竹实生苗茎秆生长及CesA基因表达的影响. 植物研究 37, 744-750. |
| [18] | 李欢庆, 李桂玲, 崔香环, 安国勇, 宋纯鹏 ( 2008). 茉莉酸甲酯抑制拟南芥根伸长生长电生理学机制. 广西植物 28, 414-419. |
| [19] | 李惠民, 李宏亮, 高吉仁 ( 2010). 水杨酸对黄瓜种子萌发和胚根伸长的影响及其与乙烯的关系. 西安工程大学学报 24, 773-775. |
| [20] | 黎舒佳, 高谨, 李家洋, 王永红 ( 2015). 独脚金内酯调控水稻分蘖的研究进展. 植物学报 50, 539-548. |
| [21] | 李霞, 郭振华 ( 2014). 麻竹节间伸长过程的初步研究. 植物分类与资源学报 36, 22-28. |
| [22] | 李兴亮, 郭献平, 沈元月, 曹庆芹, 冯永庆, 秦岭 ( 2011). 板栗赤霉素缺陷型短雄花序芽变的初步鉴定及CmGID1基因的表达分析. 园艺学报 38, 1251-1258. |
| [23] | 刘芳, 袁华招, 沈欣杰, 廖雄, 李天红 ( 2013). 外源GA3和PP333对甜樱桃新梢生长及赤霉素代谢关键基因表达的影响. 核农学报 27, 272-278. |
| [24] | 阙友雄, 许莉萍, 徐景升, 张积森, 张木清, 陈如凯 ( 2009). 甘蔗基因表达定量PCR分析中内参基因的选择. 热带作物学报 30, 274-278. |
| [25] | 王利军, 战吉成, 黄卫东 ( 2002). 水杨酸与植物抗逆性. 植物生理学通讯 38, 619-624. |
| [26] | 王伦旺, 廖江雄, 谭芳, 唐仕云, 黄家雍, 李翔, 杨荣仲, 李杨瑞, 黄海荣, 经艳, 邓宇驰 ( 2015). 高产高糖抗倒伏甘蔗新品种桂糖42号的选育及高产栽培技术. 南方农业学报 46, 1361-1366. |
| [27] | 魏强, 丁雨龙 ( 2017). 矢竹地下茎转录组测序及节间生长相关基因表达分析. 南京林业大学学报(自然科学版) 41(5), 42-48. |
| [28] | 卫晓轶, 张明才, 张燕, 李召虎, 段留生 ( 2011). 乙烯利对不同基因型玉米节间伸长和内源激素的影响. 农药学学报 13, 475-479. |
| [29] | 吴建明, 李杨瑞, 王爱勤, 杨柳, 杨丽涛 ( 2010 a). 赤霉素处理对甘蔗节间伸长及产质量的影响. 中国糖料 ( 4), 24-26. |
| [30] | 吴建明, 李杨瑞, 王爱勤, 杨柳, 杨丽涛 ( 2010 b). 赤霉素诱导甘蔗节间伸长的效应与相关酶活性的关系. 西北植物学报 30, 962-967. |
| [31] | 吴建明, 李杨瑞, 杨柳, 王爱勤, 杨丽涛 ( 2009). 赤霉素诱导甘蔗节间伸长与内源激素变化的关系. 热带作物学报 30, 1452-1457. |
| [32] | 吴良如, 萧江华 ( 1998). 大小年毛竹林中内源激素节律变化特征的研究. 竹子研究汇刊 17, 24-30. |
| [33] | 肖辉海 ( 2008). 长穗颈温敏核不育水稻穗颈节间伸长与内源激素的关系. 植物生理学通讯 44, 656-660. |
| [34] | 肖辉海, 郝小花, 王文龙 ( 2011). 温度对温敏核不育水稻eui突变体最上节间伸长的影响. 植物学报 46, 138-146. |
| [35] | 许婷婷, 杨光耀, 杨清培, 于芬 ( 2016). 厚壁毛竹快速高生长期竹秆ATP酶超微细胞化学定位. 西北植物学报 36, 1566-1574. |
| [36] | 徐有明, 郝培应, 费本华 ( 2008). 竹笋形态发育构建过程中解剖结构及化学成分的动态变化. 东北林业大学学报 36(4), 8-11, 16. |
| [37] | 杨晓婉, 郑国琦, 杨涓, 许兴, 卢迪, 杨乐 ( 2014). 宁夏枸杞果实内源激素的变化及其与细胞壁成分和相关酶的关系. 植物学报 49, 30-40. |
| [38] | 杨奕, 董文渊, 邱月群, 李雯, 杨佳俊, 韩妍 ( 2015). 筇竹笋生长过程中营养成分的变化. 东北林业大学学报 43, 80-82, 87. |
| [39] | 张春玲 ( 2014). 毛竹笋-竹生长发育过程系统分析与生长素相关基因研究. 博士论文. 北京: 中国林业科学研究院. pp. 42-68. |
| [40] | 张珂 ( 2014). 玉米节间伸长过程中相关基因甲基化及表达分析. 硕士论文. 武汉: 华中农业大学. pp. 37-38. |
| [41] | 张同祯, 李永生, 李玥, 姚海梅, 赵娟, 王婵, 赵阳, 王汉宁, 方永丰, 胡晋 ( 2016). 多胺氧化酶(PAO)调控光诱导玉米中胚轴伸长的生理机制. 作物学报 42, 734-742. |
| [42] | 张莹, 田埂, 路慧萍, 冉洪, 胡陶, 郭起荣 ( 2015). 厚壁毛竹六个节气笋芽发育的转录组分析. 江西农业大学学报 37, 466-474. |
| [43] | Abeles FB, Morgan PW, Saltveit ME ( 1992). Ethylene in Plant Biology. San Diego: Academic Press. pp. 123-135. |
| [44] | Ayano M, Kani T, Kojima M, Sakakibara H, Kitaoka T, Kuroha T, Angeles-Shim RB, Kitano H, Nagai K, Ashikari M ( 2014). Gibberellin biosynthesis and signal transduction is essential for internode elongation in deepwater rice. Plant Cell Environ 37, 2313-2324. |
| [45] | Choi YH, Yoshizawa K, Kobayashi M, Sakurai A ( 1995). Distribution of endogenous gibberellins in vegetative shoots of rice. Plant Cell Physiol 36, 997-1001. |
| [46] | Coles JP, Phillips AL, Croker SJ, García-Lepe R, Lewis MJ, Hedden P ( 1999). Modification of gibberellin production and plant development in Arabidopsis by sense and antisense expression of gibberellin 20-oxidase genes. Plant J 17, 547-556. |
| [47] | Cui K, He CY, Zhang JG, Duan AG, Zeng YF ( 2012). Temporal and spatial profiling of internode elongation-associated protein expression in rapidly growing culms of bamboo. J Proteome Res 11, 2492-2507. |
| [48] | Cutler AJ, Krochko JE ( 1999). Formation and breakdown of ABA. Trends Plant Sci 4, 472-478. |
| [49] | De Lucas M, Davière JM, Rodríguez-Falcón M, Pontin M, Iglesias-Pedraz JM, Lorrain S, Fankhauser C, Blázquez MA, Titarenko E, Prat S ( 2008). A molecular framework for light and gibberellin control of cell elongation. Nature 451, 480-484. |
| [50] | Eriksson ME, Hoffman D, Kaduk M, Mauriat M, Moritz T ( 2015). Transgenic hybrid aspen trees with increased gibberellin (GA) concentrations suggest that GA acts in parallel with FLOWERING LOCUS T2 to control shoot elongation. New Phytol 205, 1288-1295. |
| [51] | Feng SH, Martinez C, Gusmaroli G, Wang Y, Zhou JL, Wang F, Chen LY, Yu L, Iglesias-Pedraz JM, Kircher S, Schäfer E, Fu XD, Fan LM, Deng XW ( 2008). Coordinated regulation of Arabidopsis thaliana development by light and gibberellins. Nature 451, 475-479. |
| [52] | Gao J, Zhang Y, Zhang CL, Qi FY, Li XP, Mu SH, Peng ZH ( 2014). Characterization of the floral transcriptome of Moso bamboo (Phyllostachys edulis) at different flowering developmental stages by transcriptome sequencing and RNA-seq analysis. PLoS One 9, e98910. |
| [53] | Gao XH, Huang XZ, Xiao SL, Fu XD ( 2008). Evolutionarily conserved DELLA-mediated gibberellin signaling in plants. J Integr Plant Biol 50, 825-834. |
| [54] | Hattori Y, Nagai K, Furukawa S, Song XJ, Kawano R, Sakakibara H, Wu JZ, Matsumoto T, Yoshimura A, Kitano H, Matsuoka M, Mori H, Ashikari M ( 2009). The ethylene response factors SNORKEL1 and SNORKEL2 allow rice to adapt to deep water. Nature 460, 1026-1030. |
| [55] | He CY, Cui K, Zhang JG, Duan AG, Zeng YF ( 2013). Next-generation sequencing-based mRNA and microRNA expression profiling analysis revealed pathways involved in the rapid growth of developing culms in Moso bamboo. BMC Plant Biol 13, 119. |
| [56] | Hedden P, Phillips AL ( 2000). Gibberellin metabolism: new insights revealed by the genes. Trends Plant Sci 5, 523-530. |
| [57] | Kende H, van der Knaap E, Cho HT ( 1998). Deepwater rice: a model plant to study stem elongation. Plant Physiol 118, 1105-1110. |
| [58] | Lawit SJ, Wych HM, Xu DP, Kundu S, Tomes DT ( 2010). Maize DELLA proteins dwarf plant8 and dwarf plant9 as modulators of plant development. Plant Cell Physiol 51, 1854-1868. |
| [59] | Lee S, Cheng H, King KE, Wang W, He YW, Hussain A, Lo J, Harberd NP, Peng JR ( 2002). Gibberellin regulates Arabidopsis seed germination via RGL2, a GAI/RGA-like gene whose expression is up-regulated following imbibition. Genes Dev 16, 646-658. |
| [60] | Li LF, Zhang WB, Zhang LL, Li N, Peng JZ, Wang YQ, Zhong CM, Yang YP, Sun SL, Liang S, Wang XJ ( 2015). Transcriptomic insights into antagonistic effects of gibberellin and abscisic acid on petal growth in Gerbera hybrida. Front Plant Sci 6, 168. |
| [61] | Magome H, Nomura T, Hanada A, Takeda-Kamiya N, Ohnishi T, Shinma Y, Katsumata T, Kawaide H, Kamiya Y, Yamaguchi S ( 2013). CYP714B1 and CYP 714B2 encode gibberellin 13-oxidases that reduce gibberellin activity in rice. Proc Natl Acad Sci USA 110, 1947-1952. |
| [62] | Mauriat M, Moritz T ( 2009). Analyses of GA20ox- and GID1-over-expressing aspen suggest that gibberellins play two distinct roles in wood formation. Plant J 58, 989-1003. |
| [63] | Peng JR, Carol PO, Richards DE, King KE, Cowling RJ, Murphy GP, Harberd NP ( 1997). The Arabidopsis GAI gene defines a signaling pathway that negatively regulates gibberellin responses. Genes Dev 11, 3194-3205. |
| [64] | Peng ZH, Zhang CL, Zhang Y, Hu T, Mu SH, Li XP, Gao J ( 2013). Transcriptome sequencing and analysis of the fast growing shoots of Moso bamboo (Phyllostachys edulis). PLoS One 8, e78944. |
| [65] | Pierik R, Cuppens MLC, Voesenek LACJ, Visser EJW ( 2004). Interactions between ethylene and gibberellins in phytochrome-mediated shade avoidance responses in tobacco. Plant Physiol 136, 2928-2936. |
| [66] | Pierik R, Visser EJW, De Kroon H, Voesenek LACJ ( 2003). Ethylene is required in tobacco to successfully compete with proximate neighbours. Plant Cell Environ 26, 1229-1234. |
| [67] | Qin XQ, Zeevaart JAD ( 2002). Overexpression of a 9-cis-epoxycarotenoid dioxygenase gene in Nicotiana plumbaginifolia increases abscisic acid and phaseic acid levels and enhances drought tolerance. Plant Physiol 128, 544-551. |
| [68] | Rieu I, Ruiz-Rivero O, Fernandez-Garcia N, Griffiths J, Powers SJ, Gong F, Linhartova T, Eriksson S, Nilsson O, Thomas SG, Phillips AL, Hedden P ( 2008). The gibberellin biosynthetic genes AtGA20ox1 and AtGA20- ox2 act, partially redundantly, to promote growth and development throughout the Arabidopsis life cycle. Plant J 53, 488-504. |
| [69] | Ross JJ, O’Neill DP, Smith JJ, Kerckhoffs LHJ, Elliot RC ( 2000). Evidence that auxin promotes gibberellin A1 biosynthesis in pea. Plant J 21, 547-552. |
| [70] | Takatsuka H, Umeda M ( 2014). Hormonal control of cell division and elongation along differentiation trajectories in roots. J Exp Bot 65, 2633-2643. |
| [71] | Thomas GS, Hedden P ( 2018). Gibberellin metabolism and signal transduction. In: Hedden P, Thomas SG, eds. Annual Plant Reviews. Washington: John Wiley & Sons. pp. 147-184. |
| [72] | Ueguchi-Tanaka M, Ashikari M, Nakajima M, Itoh H, Katoh E, Kobayashi M, Chow TY, Hsing YI, Kitano H, Yamaguchi I, Matsuoka M ( 2005). Gibberellin insensitive Dwarf 1 encodes a soluble receptor for gibberellin. Nature 437, 693-698. |
| [73] | Voorend W, Nelissen H, Vanholme R, De Vliegher A, Van Breusegem F, Boerjan W, Roldán-Ruiz I, Muylle H, Inzé D ( 2016). Overexpression of GA20-OXIDASE1 impacts plant height, biomass allocation and saccharification efficiency in maize. Plant Biotechnol J 14, 997-1007. |
| [74] | Wang HY, Cui K, He CY, Zeng YF, Liao SX, Zhang JG ( 2015). Endogenous hormonal equilibrium linked to bamboo culm development. Genet Mol Res 14, 11312-11323. |
| [75] | Wang YJ, Deng DX ( 2014). Molecular basis and evolutionary pattern of GA-GID1-DELLA regulatory module. Mol Genet Genomics 289, 1-9. |
| [76] | Xiao YH, Ye YF, Feng Y, Li XB, Luo M, Hou L, Luo XY, Li DM, Pei Y ( 2006). Functional expression of the cotton gibberellic acid oxidase homologous gene GhGA20ox1 in tobacco. J Plant Physiol Mol Biol 32, 563-569. |
| [1] | Liu Xupeng, Wang Min, Han Shou'an, Zhu Xuehui, Wang Yanmeng, Pan Mingqi, Zhang Wen. Research Progress on Factors and Molecular Mechanisms Regulating Plant Organ Abscission [J]. Chinese Bulletin of Botany, 2025, 60(3): 472-482. |
| [2] | Tingxin Chen, Min Fu, Na Li, Leilei Yang, Lingfei Li, Chunmei Zhong. Identification and Expression Analysis of DNA Methyltransferase in Begonia masoniana [J]. Chinese Bulletin of Botany, 2024, 59(5): 726-737. |
| [3] | Ming-Yi Bai, Jinrong Peng, Xiangdong Fu. Coordinated Regulation of Gibberellin and Brassinosteroid Signalings Drives Toward a Sustainable “Green Revolution” by Breeding the New Generation of High-yield Wheat [J]. Chinese Bulletin of Botany, 2023, 58(2): 194-198. |
| [4] | Li Yue, Hu Desheng, Tan Jinfang, Mei Hao, Wang Yi, Li Hui, Li Fang, Han Yanlai. Chaetomium uniseriatum Promotes Maize Growth by Accelerating Straw Degradation and Regulating the Expression of Hormone Responsive Genes [J]. Chinese Bulletin of Botany, 2022, 57(4): 422-433. |
| [5] | Yanyan Meng, Nan Zhang, Yan Xiong. Novel Links in the Plant Target of Rapamycin Signaling Networks [J]. Chinese Bulletin of Botany, 2022, 57(1): 1-11. |
| [6] | Xiaoting Zhao, Kaitao Mao, Jiahui Xu, Chuan Zheng, Xiaofeng Luo, Kai Shu. Protein Phosphorylation and Its Regulatory Roles in Seed Dormancy and Germination [J]. Chinese Bulletin of Botany, 2021, 56(4): 488-499. |
| [7] | Qilu Yu, Jiangzhe Zhao, Xiaoxian Zhu, Kewei Zhang. Regulation of Rice Growth by Root-secreted Phytohormones [J]. Chinese Bulletin of Botany, 2021, 56(2): 175-182. |
| [8] | Ruifeng Yao,Daoxin Xie. New Insight into Strigolactone Signaling [J]. Chinese Bulletin of Botany, 2020, 55(4): 397-402. |
| [9] | Jiahui Xu, Yujia Dai, Xiaofeng Luo, Kai Shu, Weiming Tan. Thoughts and Applications of Chemical Biology in Phytohormonal Research [J]. Chinese Bulletin of Botany, 2020, 55(3): 369-381. |
| [10] | Jia Guo,Yansu Li,Chaoxing He,Yan Yan,Xianchang Yu. Establishing a High-efficiency Regeneration System in Pumpkin (Cucurbita moschata) [J]. Chinese Bulletin of Botany, 2019, 54(4): 539-546. |
| [11] | Liu Ming, Liu Xia, Sun Ran, Li Yuling, Du Kejiu. Polychlorinated Biphenyls Promotes Differentiation on Adventitious Roots of Populous tomentosa [J]. Chinese Bulletin of Botany, 2018, 53(6): 764-772. |
| [12] | Shi Ce, Luo Pan, Zou Jie, Sun Mengxiang. The Role of DELLA Proteins in Sexual Reproduction of Angiosperms [J]. Chinese Bulletin of Botany, 2018, 53(6): 745-755. |
| [13] | Haiwei Shuai, Yongjie Meng, Feng Chen, Wenguan Zhou, Xiaofeng Luo, Wenyu Yang, Kai Shu. Phytohormone-mediated Plant Shade Responses [J]. Chinese Bulletin of Botany, 2018, 53(1): 139-148. |
| [14] | Zheng Jun, Qiao Ling, Zhao Jiajia, Qiao Linyi, Zhang Shichang, Chang Jianzhong, Tang Caiguo, Yang Sanwei. Whole-genome Analysis of CCT Gene Family and Their Responses to Phytohormones in Aegilops tauschii [J]. Chinese Bulletin of Botany, 2017, 52(2): 188-201. |
| [15] | Chunmei Zhong, Xiaojing Wang. Progress in Cysteine-rich Gibberellic Acid-stimulated Arabidopsis Protein [J]. Chinese Bulletin of Botany, 2016, 51(1): 1-8. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||