

植物学报 ›› 2023, Vol. 58 ›› Issue (6): 956-965.DOI: 10.11983/CBB22252 cstr: 32102.14.CBB22252
张御格1,2, 袁笑妍1,2, 张贵芳1,2, 李雨健1,2, 殷金环1,2, 林金星1,2, 李晓娟1,2,*( )
)
收稿日期:2022-10-25
接受日期:2023-02-28
出版日期:2023-11-01
发布日期:2023-11-27
通讯作者:
* E-mail: lixj@bjfu.edu.cn
基金资助:
Yuge Zhang1,2, Xiaoyan Yuan1,2, Guifang Zhang1,2, Yujian Li1,2, Jinhuan Yin1,2, Jinxing Lin1,2, Xiaojuan Li1,2,*( )
)
Received:2022-10-25
Accepted:2023-02-28
Online:2023-11-01
Published:2023-11-27
Contact:
* E-mail: lixj@bjfu.edu.cn
摘要: 点击化学又称“链接化学”或“速配结合式组合化学”。其可通过碳-杂原子键(C-X-C)连接产生出诸多功能强大、高度可靠且具较强特异性的反应, 是一种快速合成大量化合物的新方法。近几年, 点击化学在药物开发、新材料合成、材料表面功能化修饰和生物大分子标记等方面取得了较大进展。2022年, 点击化学的开拓者获得了诺贝尔化学奖。该文简要介绍点击化学的原理和反应类型, 重点总结其在标记生物大分子上的研究进展, 特别是在植物细胞壁聚糖标记方面的应用, 以期为解析植物细胞壁结构、合成和动态转运机制提供新思路。
张御格, 袁笑妍, 张贵芳, 李雨健, 殷金环, 林金星, 李晓娟. 点击化学反应在植物细胞标记中的应用. 植物学报, 2023, 58(6): 956-965.
Yuge Zhang, Xiaoyan Yuan, Guifang Zhang, Yujian Li, Jinhuan Yin, Jinxing Lin, Xiaojuan Li. The Application of Click Chemistry Reactions in Plant Cell Labeling. Chinese Bulletin of Botany, 2023, 58(6): 956-965.
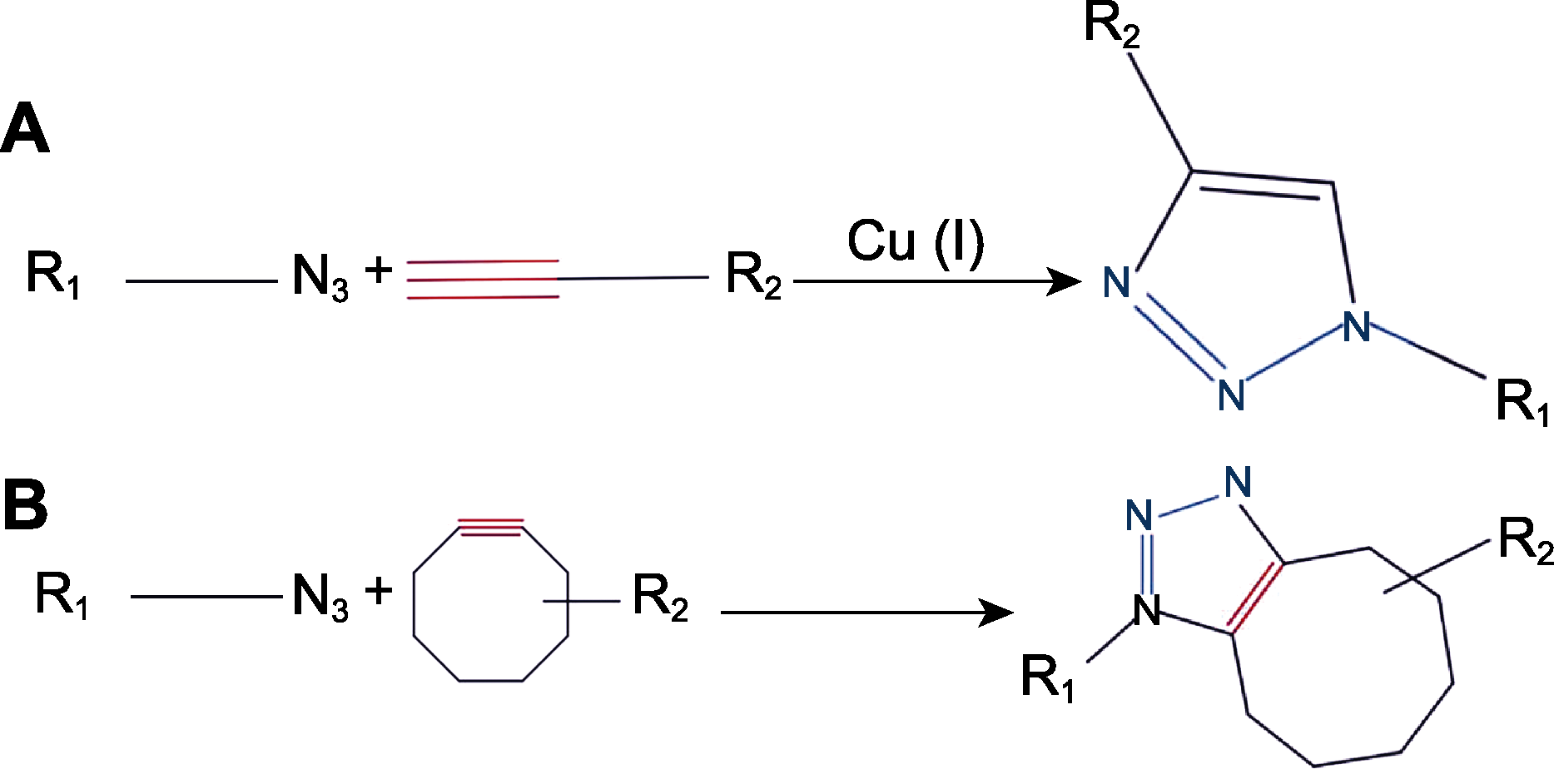
图1 环加成反应的2种反应类型CuAAC (A)和SPAAC (B) CuAAC采用端基炔烃和叠氮化物在铜离子催化下形成1,4-二取代-1,2,3-三唑; SPAAC利用环辛炔和叠氮化物反应形成稳定的三唑。CuAAC: 铜(I)催化的叠氮-炔烃环化加成反应; SPAAC: 张力促进的叠氮-炔烃环化加成反应
Figure 1 Two types of cycloadditions reactions CuAAC (A) and SPAAC (B) CuAAC uses end-group alkynes and azides to form 1,4-disubstituted-1,2,3-triazole catalyzed by copper ions; SPAAC uses cyclooctyne and azide reactions to form stable triazoles. CuAAC: Copper (I) catalysed azide-alkyne cycloaddition; SPAAC: Strain-promoted azide-alkyne cycloaddition
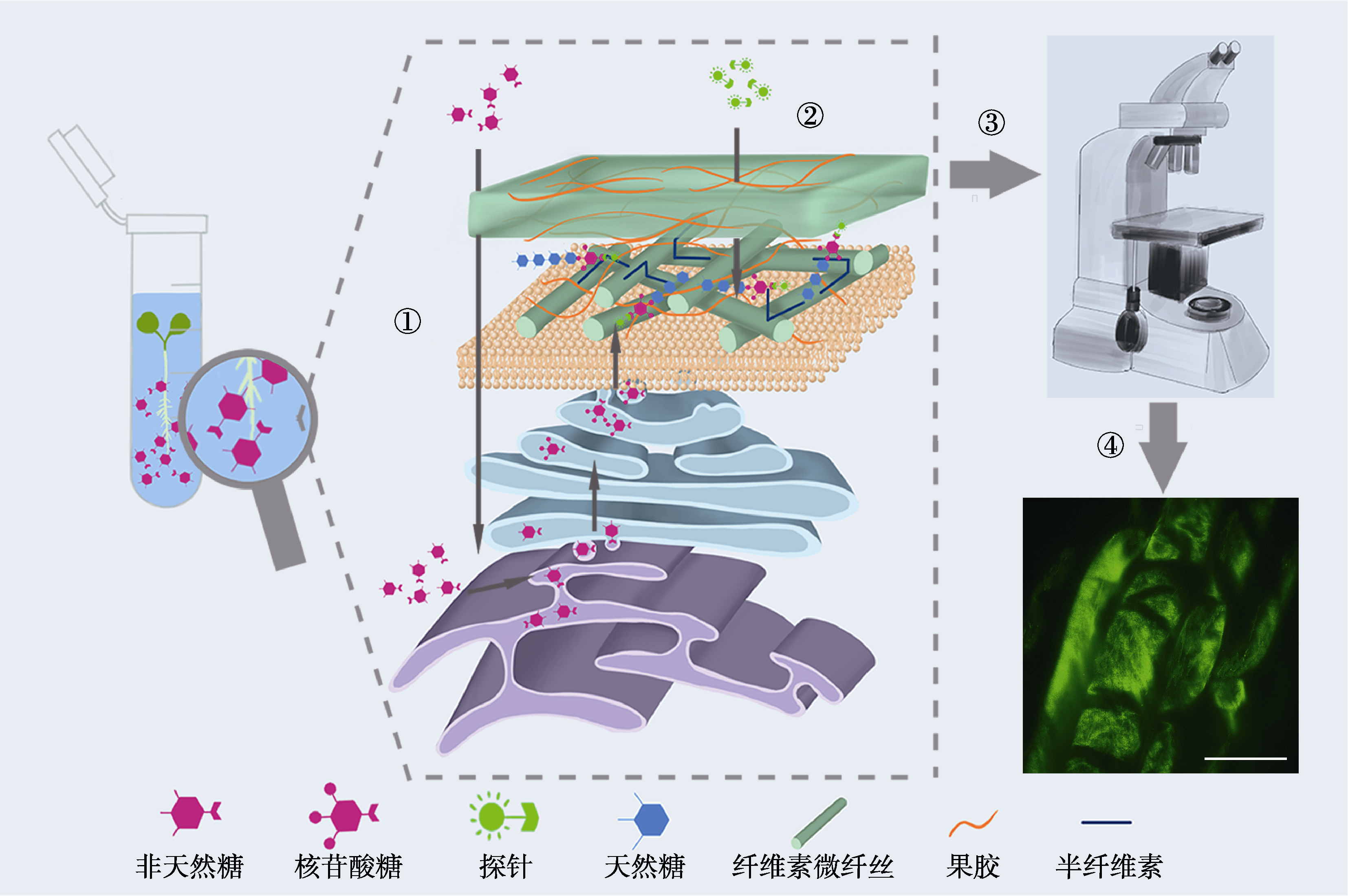
图2 点击化学标记在细胞壁聚糖中应用流程图 图中非天然糖指细胞壁中的单糖组分, 不特指具体糖类。① 外源单糖类似物通过聚糖合成的补救途径代谢进入细胞; ② 含荧光团的探针与沉积在细胞壁上并含有单糖类似物的聚糖发生点击反应; ③ 荧光成像检测单糖类似物在植物体内的排布, 如下方荧光成像图所示植物细胞壁中的果胶RG-II; ④ 荧光显微镜成像点击化学标记的果胶在植物根中的排布(bar=50 μm)。
Figure 2 The flow diagram of applying click chemistry to labeling cell wall glycan The non-natural sugars in the figure refer to the monosaccharide components in the cell wall, and do not refer to specific sugar molecules. ① Exogenous monosaccharide analogues are metabolized into cells through the remedial glycan synthesis pathway; ② Probes containing fluorophores click with glycans that contain monosaccharide analogues and are deposited on the cell wall; ③ Fluorescence is detected to investigate the arrangement of monosaccharide analogues in plants, as shown in the fluorescent image of pectin RG-II in plant cell wall; ④ Fluorescence microscopy imaging of pectin distribution in plant roots labelled by click chemistry (bar=50 μm).
| 外源糖 | 毒害作用 | 组织定位 |
|---|---|---|
| 炔基化岩藻糖 | 无毒性 | 分布于根表皮的细胞壁中 |
| 8-叠氮-8-脱氧-3-脱氧- D-甘露-2-辛酮糖酸 | 无毒性 | 分布于整个初生壁中, 但在质膜附近标记更强 |
| 叠氮基N-乙酰氨基葡萄糖 | 无毒性 | 根中均有分布, 分生区分布较多 |
| 叠氮乙酰基阿拉伯糖 | 无毒性 | 根中均有分布 |
| 叠氮乙酰基岩藻糖 | 无毒性 | 根中均有分布 |
| 6-脱氧-炔基葡萄糖 | 抑制根生长 | 定位于根毛凸起和突出的根毛尖端 |
表1 拟南芥中含有炔基或叠氮官能团的糖类似物
Table 1 Sugar analogues containing alkyne or azide functional groups in Arabidopsis thaliana
| 外源糖 | 毒害作用 | 组织定位 |
|---|---|---|
| 炔基化岩藻糖 | 无毒性 | 分布于根表皮的细胞壁中 |
| 8-叠氮-8-脱氧-3-脱氧- D-甘露-2-辛酮糖酸 | 无毒性 | 分布于整个初生壁中, 但在质膜附近标记更强 |
| 叠氮基N-乙酰氨基葡萄糖 | 无毒性 | 根中均有分布, 分生区分布较多 |
| 叠氮乙酰基阿拉伯糖 | 无毒性 | 根中均有分布 |
| 叠氮乙酰基岩藻糖 | 无毒性 | 根中均有分布 |
| 6-脱氧-炔基葡萄糖 | 抑制根生长 | 定位于根毛凸起和突出的根毛尖端 |
| [1] | 成波, 陈兴 (2020). 唾液酸化聚糖的化学标记和解析. 科学通报 65, 2984-2997. |
| [2] | 刘佩佩, 张耿, 李晓娟 (2021). 植物果胶的生物合成与功能. 植物学报 56, 191-200. |
| [3] |
刘玥, 尹悦佳, 梁重阳, 黄殿帅, 王阳, 刘艳芝, 窦瑶, 冯树丹, 郝东云 (2015). 3D-SIM结构照明超分辨率显微镜实现蛋白质在植物亚细胞器内的定位. 植物学报 50, 495-503.
DOI |
| [4] | 肖银燕, 袁伟娜, 刘静, 孟建, 盛奇明, 谭烨欢, 徐春香 (2020). 木葡聚糖及其在植物抗逆过程中的功能研究进展. 植物学报 55, 777-787. |
| [5] |
杨麦云, 陈鹏 (2015). 生物正交标记反应研究进展. 化学学报 73, 783-792.
DOI |
| [6] | 占方玲, 高思宇, 谢元栋, 张金铭, 李毅, 刘宁 (2020). 点击化学反应在蛋白质组学分析中的研究进展. 分析化学 48, 431-438. |
| [7] | Finn MG, Kolb HC, Fokin VV, Sharpless KB (张欣豪, 吴云东译)(2008). 点击化学——释义与目标. 化学进展 20, 1-4. |
| [8] |
Agard NJ, Prescher JA, Bertozzi CR (2004). A strain-promoted [3+2] azide-alkyne cycloaddition for covalent modification of biomolecules in living systems. J Am Chem Soc 126, 15046-15047.
DOI URL |
| [9] |
Ancajas CF, Ricks TJ, Best MD (2020). Metabolic labeling of glycerophospholipids via clickable analogs derivatized at the lipid headgroup. Chem Phys Lipids 232, 104971.
DOI URL |
| [10] |
Anderson CT, Wallace IS, Somerville CR (2012). metabolic click-labeling with a fucose analog reveals pectin delivery, architecture, and dynamics in Arabidopsis cell walls. Proc Natl Acad Sci USA 109, 1329-1334.
DOI PMID |
| [11] |
Beatty KE, Xie F, Wang Q, Tirrell DA (2005). Selective dye-labeling of newly synthesized proteins in bacterial cells. J Am Chem Soc 127, 14150-14151.
PMID |
| [12] |
Beller NC, Hummon AB (2022). Advances in stable isotope labeling: dynamic labeling for spatial and temporal proteo-mic analysis. Mol Omics 18, 579-590.
DOI URL |
| [13] |
Bidhendi AJ, Chebli Y, Geitmann A (2020). Fluorescence visualization of cellulose and pectin in the primary plant cell wall. J Microsc 278, 164-181.
DOI URL |
| [14] |
Bird RE, Lemmel SA, Yu X, Zhou QA (2021). Bioorthogonal chemistry and its applications. Bioconjug Chem 32, 2457-2479.
DOI URL |
| [15] |
Breugst M, Reissig HU (2020). The Huisgen reaction: milestones of the 1,3-dipolar cycloaddition. Angew Chem Int Ed Engl 59, 12293-12307.
DOI URL |
| [16] |
Chang XL, Chen LY, Liu BN, Yang ST, Wang HF, Cao AN, Chen CY (2022). Stable isotope labeling of nanomaterials for biosafety evaluation and drug development. Chin Chem Lett 33, 3303-3314.
DOI URL |
| [17] |
Cheng B, Tang Q, Zhang C, Chen X (2021). Glycan labeling and analysis in cells and in vivo. Annu Rev Anal Chem (Palo Alto Calif) 14, 363-387.
DOI URL |
| [18] |
Depmeier H, Hoffmann E, Bornewasser L, Kath-Schorr S (2021). Strategies for covalent labeling of long RNAs. Chembiochem 22, 2826-2847.
DOI PMID |
| [19] |
Devaraj NK, Finn MG (2021). Introduction: click chemistry. Chem Rev 121, 6697-6698.
DOI PMID |
| [20] |
Devree BT, Steiner LM, Głazowska S, Ruhnow F, Her-burger K, Persson S, Mravec J (2021). Current and future advances in fluorescence-based visualization of plant cell wall components and cell wall biosynthetic machineries. Biotechnol Biofuels 14, 78.
DOI |
| [21] |
Dumont M, Lehner A, Vauzeilles B, Malassis J, Marchant A, Smyth K, Linclau B, Baron A, Mas Pons J, Ander-son CT, Schapman D, Galas L, Mollet JC, Lerouge P (2016). Plant cell wall imaging by metabolic click-mediated labeling of rhamnogalacturonan II using azido 3-deoxy-D-manno-oct-2-ulosonic acid. Plant J 85, 437-447.
DOI URL |
| [22] |
Fantoni NZ, El-Sagheer AH, Brown T (2021). A hitchhiker’s guide to click-chemistry with nucleic acids. Chem Rev 121, 7122-7154.
DOI PMID |
| [23] |
Gothelf KV, Jørgensen KA (1998). Asymmetric 1,3-dipolar cycloaddition reactions. Chem Rev 98, 863-910.
PMID |
| [24] |
Hoogenboom J, Berghuis N, Cramer D, Geurts R, Zuilhof H, Wennekes T (2016). Direct imaging of glycans in Arabidopsis roots via click labeling of metabolically incorporated azido-monosaccharides. BMC Plant Biol 16, 220.
PMID |
| [25] |
Huisgen R (1963). 1,3-dipolar cycloadditions. Past and future. Angew Chem Int Ed Engl 2, 565-598.
DOI URL |
| [26] |
Jao CY, Roth M, Welti R, Salic A (2009). Metabolic labeling and direct imaging of choline phospholipids in vivo. Proc Natl Acad Sci USA 106, 15332-15337.
DOI URL |
| [27] |
Jao CY, Salic A (2008). Exploring RNA transcription and turnover in vivo by using click chemistry. Proc Natl Acad Sci USA 105, 15779-15784.
DOI URL |
| [28] |
John CJ, Carolyn RB (2010). Cu-free click cycloaddition reactions in chemical biology. Chem Soc Rev 39, 1272-1279.
PMID |
| [29] |
Kim E, Koo H (2019). Biomedical applications of copper- free click chemistry: in vitro, in vivo, and ex vivo. Chem Sci, 10, 7835-7851.
DOI URL |
| [30] |
Kolb HC, Finn MG, Sharpless KB (2001). Click chemistry: diverse chemical function from a few good reactions. Angew Chem Int Ed Engl 40, 2004-2021.
DOI URL |
| [31] |
Kuerschner L, Thiele C (2022). Tracing lipid metabolism by alkyne lipids and mass spectrometry: the state of the art. Front Mol Biosci 9, 880559.
DOI URL |
| [32] |
Li L, Zhang ZY (2016). Development and applications of the copper-catalyzed azide-alkyne cycloaddition (CuAAC) as a bioorthogonal reaction. Molecules 21, 1393.
DOI URL |
| [33] |
Lion C, Simon C, Huss B, Blervacq AS, Tirot L, Toybou D, Spriet C, Slomianny C, Guerardel Y, Hawkins S, Biot C (2017). BLISS: a bioorthogonal dual-labeling strategy to unravel lignification dynamics in plants. Cell Chem Biol 24, 326-338.
DOI PMID |
| [34] |
Ming X, Leonard P, Heindl D, Seela F (2008). Azide-alkyne "click" reaction performed on oligonucleotides with the universal nucleoside 7-octadiynyl-7-deaza-2'-deoxyinosine. Nucleic Acids Symp Ser 52, 471-472.
DOI URL |
| [35] |
Neef AB, Luedtke NW (2014). An azide-modified nucleoside for metabolic labeling of DNA. Chembiochem 15, 789-793.
DOI URL |
| [36] |
Neef AB, Pernot L, Schreier VN, Scapozza L, Luedtke NW (2015). A bioorthogonal chemical reporter of viral infection. Angew Chem Int Ed Engl 54, 7911-7914.
DOI URL |
| [37] |
Paper JM, Mukherjee T, Schrick K (2018). Bioorthogonal click chemistry for fluorescence imaging of choline phospholipids in plants. Plant Methods 14, 31.
DOI |
| [38] |
Parker CG, Pratt MR (2020). Click chemistry in proteomic investigations. Cell 180, 605-632.
DOI PMID |
| [39] |
Prescher JA, Bertozzi CR (2005). Chemistry in living sys-tems. Nat Chem Biol 1, 13-21.
DOI |
| [40] |
Rodríguez DF, Moglie Y, Ramírez-Sarmiento CA, Singh SK, Dua K, Zacconi FC (2022). Bio-click chemistry: a bridge between biocatalysis and click chemistry. RSC Adv 12, 1932-1949.
DOI PMID |
| [41] |
Ropitaux M, Hays Q, Baron A, Fourmois L, Boulogne I, Vauzeilles B, Lerouge P, Mollet JC, Lehner A (2022). Dynamic imaging of cell wall polysaccharides by metabolic click-mediated labeling of pectins in living elongating cells. Plant J 110, 916-924.
DOI URL |
| [42] |
Rostovtsev VV, Green LG, Fokin VV, Sharpless KB (2002). A stepwise Huisgen cycloaddition process: copper (I)-catalyzed regioselective "ligation" of azides and terminal alkynes. Angew Chem Int Ed Engl 41, 2596-2599.
DOI URL |
| [43] |
Takayama Y, Kusamori K, Nishikawa M (2019). Click chemistry as a tool for cell engineering and drug delivery. Molecules 24, 172.
DOI URL |
| [44] |
Teramoto H, Kojima K (2015). Incorporation of methionine analogues into Bombyx mori silk fibroin for click modifica-tions. Macromol Biosci 15, 719-727.
DOI URL |
| [45] |
Tobimatsu Y (2017). A “double click” for illuminating plant cell walls. Cell Chem Biol 24, 246-247.
DOI PMID |
| [46] |
Truong L, Ferré-D'Amaré AR (2019). From fluorescent proteins to fluorogenic RNAs: tools for imaging cellular macromolecules. Protein Sci 28, 1374-1386.
DOI PMID |
| [47] |
Turner MA, Lwin TM, Amirfakhri S, Nishino H, Hoffman RM, Yazaki PJ, Bouvet M (2021). The use of fluorescent anti-CEA antibodies to label, resect and treat cancers: a review. Biomolecules 11, 1819.
DOI URL |
| [48] |
Wang JG, Zhang JB, Lee YM, Ng S, Shi Y, Hua ZC, Lin QS, Shen HM (2017). Nonradioactive quantification of autophagic protein degradation with L-azidohomoalanine labeling. Nat Protoc 12, 279-288.
DOI PMID |
| [49] |
Wittig G, Krebs A (1961). Zur existenz niedergliedriger cycloalkine, 1. Chem Ber 94, 3260-3275.
DOI URL |
| [50] | Wu J, Yu YH (2015). Recent progress on application of “click” chemistry in labeling of biomolecules. J Jianghan Univ (Nat Sci Ed) 43, 138-145. |
| [51] |
Xiong HT, Zou HY, Liu H, Wang M, Duan LL (2021). Sur-face functionalization of a γ-graphyne-like carbon material via click chemistry. Chem Asian J 16, 922-925.
DOI URL |
| [52] |
Yao TT, Xu XW, Huang R (2021). Recent advances about the applications of click reaction in chemical proteomics. Molecules 26, 5368.
DOI URL |
| [53] |
Yoon HY, Lee D, Lim DK, Koo H, Kim K (2022). Cop-per-free click chemistry: applications in drug delivery, cell tracking, and tissue engineering. Adv Mater 34, 2107192.
DOI URL |
| [54] |
Zhu YT, Chen X (2017). Expanding the scope of metabolic glycan labeling in Arabidopsis thaliana. Chembiochem 18, 1286-1296.
DOI URL |
| [55] |
Zhu YT, Wu J, Chen X (2016). Metabolic labeling and imaging of N-linked glycans in Arabidopsis thaliana. Angew Chem Int Ed Engl 55, 9301-9305.
DOI URL |
| [56] |
Zou J, Dong XY, Li YL, Tong SQ, Wang JW, Liao MX, Huang GF (2019). Deep sequencing identification of differentially expressed miRNAs in the spinal cord of resinifera-toxin-treated rats in response to electroacupuncture. Neurotox Res 36, 387-395.
DOI |
| [1] | 覃思颖, 罗燕, 张禾, 胡君, 廖菊够. 花粉管细胞壁原子力显微镜观测制样方法优化[J]. 植物学报, 2024, 59(5): 783-791. |
| [2] | 赵晗茜, 宋佳怡, 杨洁, 赵永晶, 夏文念, 顾伟卓, 汪仲毅, 杨楠, 胡慧贞. 金鱼草XTH家族基因鉴定及抗核盘菌和雄蕊瓣化相关基因筛选[J]. 植物学报, 2024, 59(2): 188-203. |
| [3] | 郭彦君, 陈枫, 罗敬文, 曾为, 许文亮. 植物细胞壁木聚糖的生物合成及其应用[J]. 植物学报, 2023, 58(2): 316-334. |
| [4] | 冯旭飞, 雷长英, 张玉洁, 向导, 杨明凤, 张旺锋, 张亚黎. 棉花花铃期叶片氮分配对光合氮利用效率的影响[J]. 植物生态学报, 2023, 47(11): 1600-1610. |
| [5] | 王琦, 许艳丽, 闫鹏, 董好胜, 张薇, 卢霖, 董志强. PAC对谷子花后土壤氮素供应和叶片抗氧化特性的影响[J]. 植物学报, 2023, 58(1): 90-107. |
| [6] | 熊映杰, 于果, 魏凯璐, 彭娟, 耿鸿儒, 杨冬梅, 彭国全. 天童山阔叶木本植物叶片大小与叶脉密度及单位叶脉长度细胞壁干质量的关系[J]. 植物生态学报, 2022, 46(2): 136-147. |
| [7] | 肖银燕, 袁伟娜, 刘静, 孟建, 盛奇明, 谭烨欢, 徐春香. 木葡聚糖及其在植物抗逆过程中的功能研究进展[J]. 植物学报, 2020, 55(6): 777-787. |
| [8] | 张雨, 赵明洁, 张蔚. 植物次生细胞壁生物合成的转录调控网络[J]. 植物学报, 2020, 55(3): 351-368. |
| [9] | 马骊, 孙万仓, 袁金海, 刘自刚, 武军艳, 方彦, 许耀照, 蒲媛媛, 白静, 董小云, 何辉立. 白菜型冬油菜β-1,3-葡聚糖酶基因在低温胁迫下的表达[J]. 植物学报, 2017, 52(5): 568-578. |
| [10] | 靳振明, 平宝哲, 沈浩珺, 杜淮清, 李瑞乾, 朱璐, 张大兵, 袁政. 水稻脆秆突变体bc-s1的表型分析和基因定位[J]. 植物学报, 2016, 51(2): 167-174. |
| [11] | 解敏敏, 晁江涛, 孔英珍. 参与木葡聚糖合成的糖基转移酶基因研究进展[J]. 植物学报, 2015, 50(5): 644-. |
| [12] | 刘冉, 王振宇, 李婷婷, 王芳, 安静. 壳聚糖对红松幼苗多酚积累和抗氧化防御酶的诱导作用[J]. 植物生态学报, 2014, 38(7): 749-756. |
| [13] | 韩笑, 郭凯, 李新新, 刘绪, 王炳锐, 夏涛, 彭良才, 丰胜求. 拟南芥纤维素合酶基因时空表达模式与功能预测[J]. 植物学报, 2014, 49(5): 539-547. |
| [14] | 许欢欢, 康健, 梁明祥. 植物果聚糖的代谢途径及其在植物抗逆中的功能研究进展[J]. 植物学报, 2014, 49(2): 209-220. |
| [15] | 杨晓婉, 郑国琦, 杨涓, 许兴, 卢迪, 杨乐. 宁夏枸杞果实内源激素的变化及其与细胞壁成分和相关酶的关系[J]. 植物学报, 2014, 49(1): 30-40. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||