

Chinese Bulletin of Botany ›› 2019, Vol. 54 ›› Issue (4): 509-514.DOI: 10.11983/CBB19101 cstr: 32102.14.CBB19101
Special Issue: 逆境生物学专辑 (2019年54卷2期)
• TECHNIQUES AND METHODS • Previous Articles Next Articles
Dongfeng Liu,Yongyan Tang,Shengtao Luo,Wei Luo,Zhitao Li,Kang Chong,Yunyuan Xu( )
)
Received:2019-05-29
Accepted:2019-06-11
Online:2019-07-01
Published:2020-01-08
Contact:
Yunyuan Xu
Dongfeng Liu,Yongyan Tang,Shengtao Luo,Wei Luo,Zhitao Li,Kang Chong,Yunyuan Xu. Identification of Chilling Tolerance of Rice Seedlings by Cold Water Bath[J]. Chinese Bulletin of Botany, 2019, 54(4): 509-514.
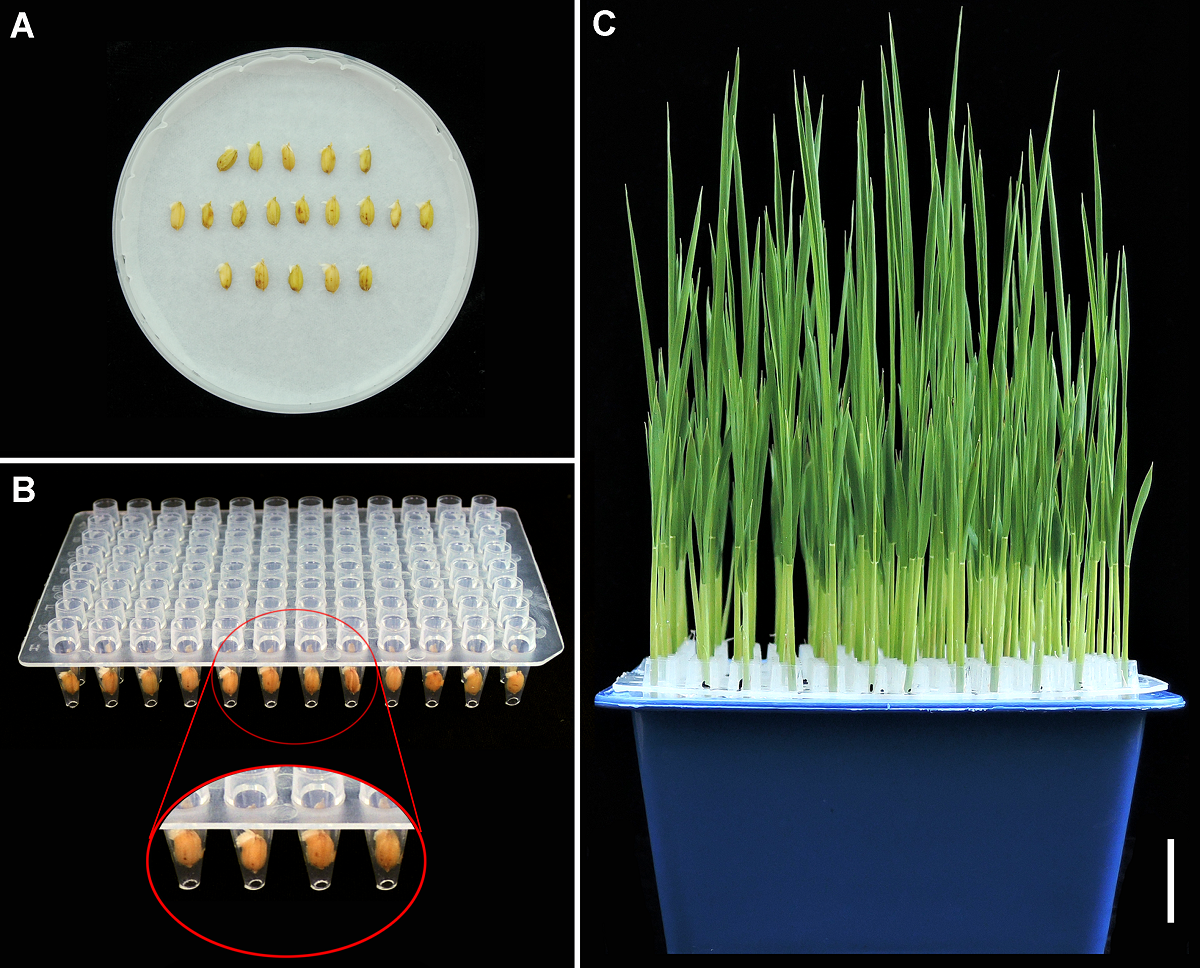
Figure 1 Germination and sowing of rice seeds, and the way of seedling culture (A) Germination of rice seeds; (B) Placement of germination seeds in 96-well plates without bottom; (C) Trefoil stage seedlings (Bar=2 cm)
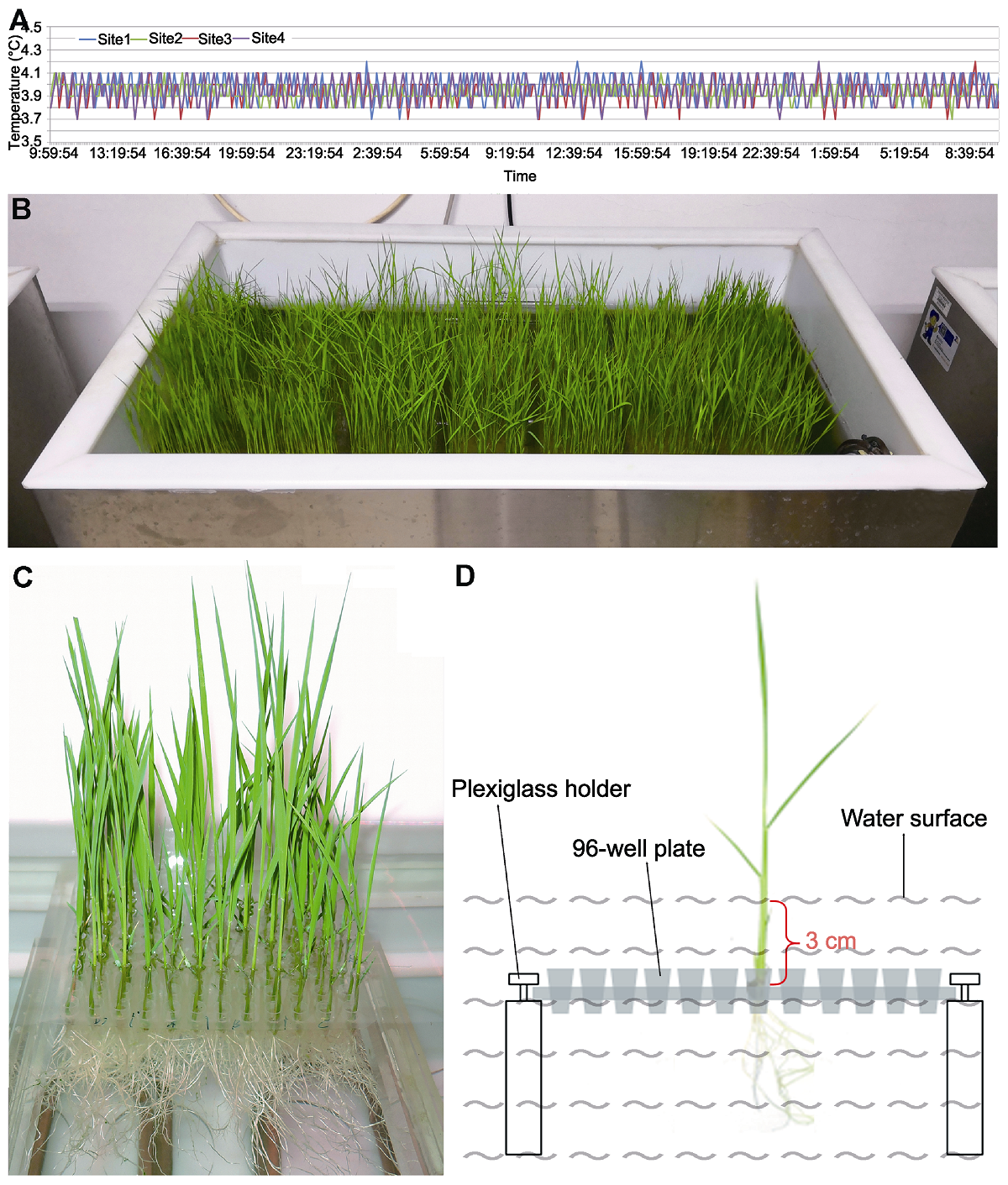
Figure 2 The diagram of low temperature water bath treatment of rice seedlings (A) The temperature of water bath within 24 h; (B) Seedlings under chilling treatment in 4°C water bath; (C) Partially enlarged detail of the seedlings in the cold water bath tank; (D) A diagram of the fixation pattern of seedlings in cold water bath
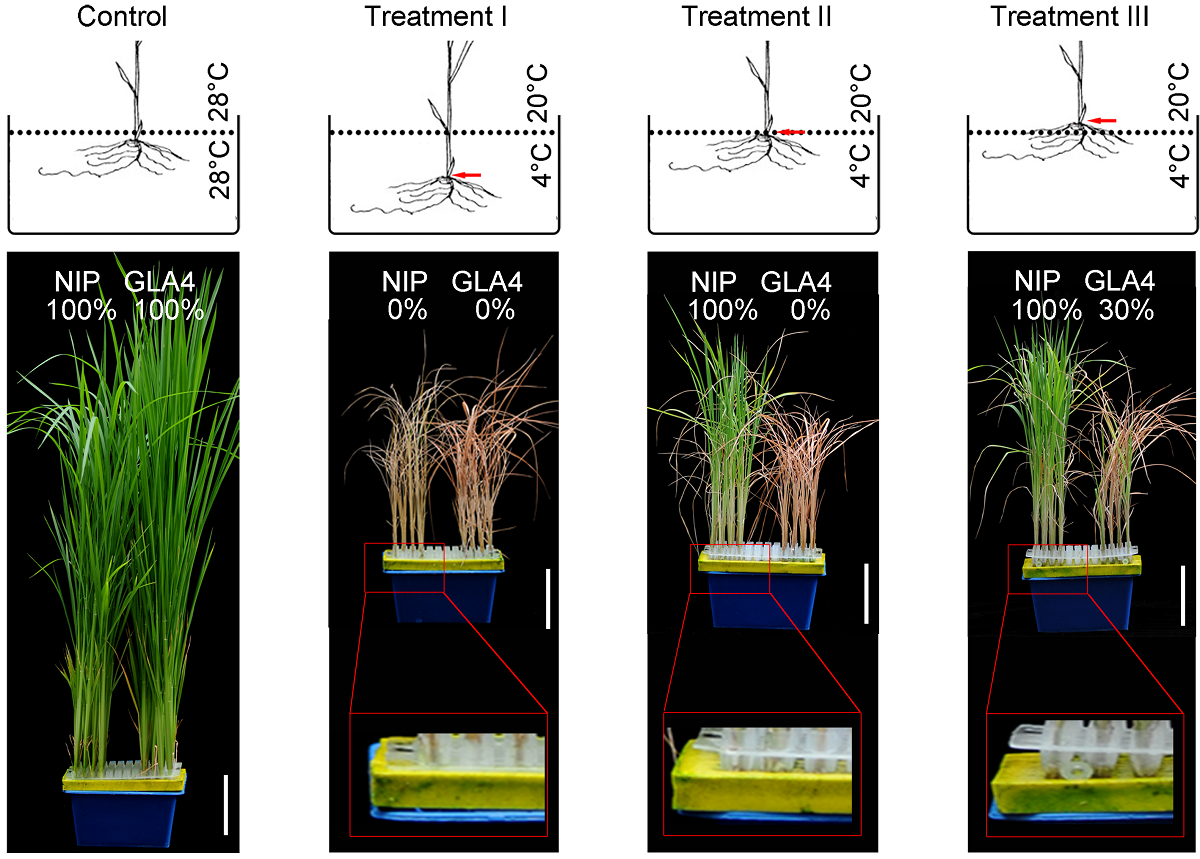
Figure 3 Effect of submerged depth of cold water on low temperature tolerance of rice Red arrow indicated the site of shoot apical meristem under different water depth in chilling treatment I, II and III. Dotted lines represent the water surface. The survival rates were indicated on the top of the seedlings recoveried for 7 days. Treatment with 28oC water was used as control (left). Bars=6 cm
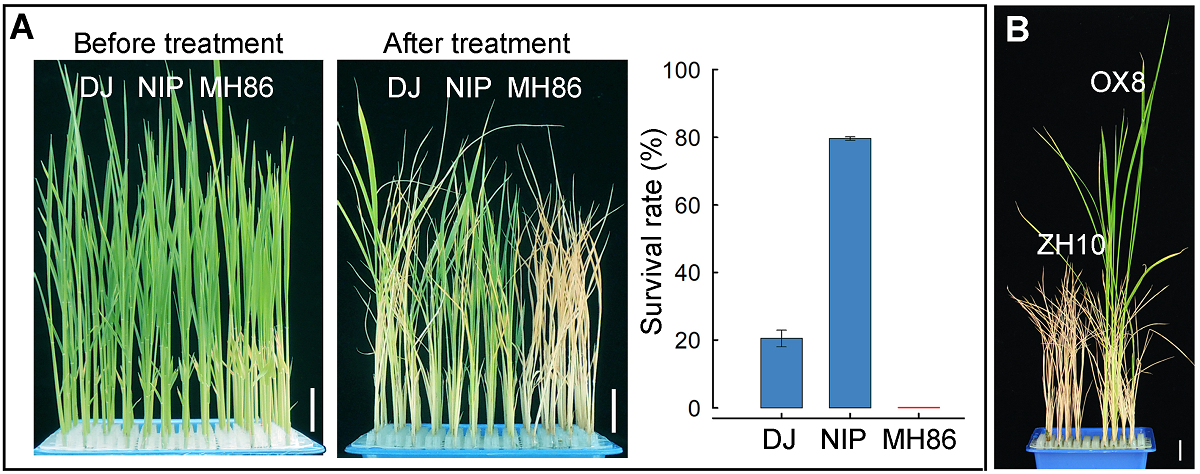
Figure 4 The evaluation of cold tolerance for different rice varieties at seedling stage (A) Phenotypic response to chilling in DJ, NIP and MH86, the survival rate was determined after cold-water treatment at 4°C for 96 h and subsequent recovery at 28°C for 7 days (means±SD, n≥30, three replicates); (B) The phenotype for seedlings of ZH10 and OX8 after the chilling treatment at 4°C for 120 h, subsequently at 28°C for 50 days. Bars=2 cm
| Subspe- cies | Cultivars | Survival rate (>50%) | Survival rate (<10%) |
|---|---|---|---|
| japonica/ geng | KY131, Kitaake | 60-80 h | 96-120 h |
| DJ, HY | 72-84 h | 120-144 h | |
| ZH10, NIP | 72-96 h | 120-144 h | |
| YD | 96-144 h | 168-192 h | |
| indica/ xian | ZF802, GLA4, IR64 | 12-28 h | 40-60 h |
| 93-11, MH86 | 24-40 h | 48-60 h |
Table 2 The recommended treatment time for different survival rates of cultivars of indica/xian and japonica/geng
| Subspe- cies | Cultivars | Survival rate (>50%) | Survival rate (<10%) |
|---|---|---|---|
| japonica/ geng | KY131, Kitaake | 60-80 h | 96-120 h |
| DJ, HY | 72-84 h | 120-144 h | |
| ZH10, NIP | 72-96 h | 120-144 h | |
| YD | 96-144 h | 168-192 h | |
| indica/ xian | ZF802, GLA4, IR64 | 12-28 h | 40-60 h |
| 93-11, MH86 | 24-40 h | 48-60 h |
| [1] | Andaya VC, Tai TH ( 2006). Fine mapping of the qCTS12 locus, a major QTL for seedling cold tolerance in rice. Theor Appl Genet 113, 467-475. |
| [2] | Baba S, Takahashi Y ( 1956). Water and sand culture. In:Togari Y, Matsuo T, Hatamari M, Yamada N, Horada T, Suzuki N, eds. Sakumotsi-Shiken-ho (Laboratory Manual in Crop Science). Kyokai, Tokyo: Nogyo-Gijitsu. pp. 157-185. |
| [3] | Bonnecarrère V, Borsani O, Díaz P, Capdevielle F, Blanco P, Monza J ( 2011). Response to photoxidative stress induced by cold in japonica rice is genotype dependent. Plant Sci 180, 726-732. |
| [4] | Ma Y, Dai XY, Xu YY, Luo W, Zheng XM, Zeng DL, Pan YJ, Lin XL, Liu HH, Zhang DJ, Xiao J, Guo XY, Xu SJ, Niu YD, Jin JB, Zhang H, Xu X, Li LG, Wang W, Qian Q, Ge S, Chong K ( 2015). COLD1 confers chilling tolerance in rice. Cell 160, 1209-1221. |
| [5] | Yoshida S, Forno DA, Cock JH, Gomez KA ( 1976). Laboratory Manual for Physiological Studies of Rice. Las Banos, Laguna: IRRI. pp. 62. |
| [6] | Zhang X, Guo XP, Lei CL, Cheng ZJ, Lin QB, Wang JL, Wu FQ, Wang J, Wan JM ( 2011). Overexpression of SlCZFP1, a novel TFIIIA-type zinc finger protein from tomato, confers enhanced cold tolerance in transgenic Arabidopsis and rice. Plant Mol Biol Rep 29, 185-196. |
| [1] | SHANGGUAN Yao-Yao, SU Shi-Ping, GU Xue-Dan, ZHANG Zheng-Zhong, ZHAO Hu, LI Yi, WEI Xing-Yu. Response of Reaumuria songorica seedlings to photoperiod and light quality ratio [J]. Chin J Plant Ecol, 2025, 49(5): 788-800. |
| [2] | LI Xin-Yi, ZHANG Li-Fang, WU You-Gui, GUO Jing, LAN Rong-Guang, LÜ Hong-Fei, YU Ming-Jian. Growth characteristics of Abies beshanzuensis seedlings at different altitudes and the influencing factors [J]. Chin J Plant Ecol, 2025, 49(4): 610-623. |
| [3] |
Juan Cui, Xiaoyu Yu, Yuejiao Yu, Chengwei Liang, Jian Sun, Wenfu Chen.
Analysis of Texture Factors and Genetic Basis Influencing the Differences in Eating Quality between Northeast China and Japanese Japonica Rice [J]. Chinese Bulletin of Botany, 2025, 60(4): 1-0. |
| [4] | Zhao Ling, Guan Ju, Liang Wenhua, Zhang Yong, Lu Kai, Zhao Chunfang, Li Yusheng, Zhang Yadong. Mapping of QTLs for Heat Tolerance at the Seedling Stage in Rice Based on a High-density Bin Map [J]. Chinese Bulletin of Botany, 2025, 60(3): 342-353. |
| [5] | Xinyu Li, Yue Gu, Feifei Xu, Jinsong Bao. Research Progress on Post-translational Modifications of Starch Biosynthesis-related Proteins in Rice Endosperm [J]. Chinese Bulletin of Botany, 2025, 60(2): 256-270. |
| [6] | Jianguo Li, Yi Zhang, Wenjun Zhang. Iron Plaque Formation and Its Effects on Phosphorus Absorption in Rice Roots [J]. Chinese Bulletin of Botany, 2025, 60(1): 132-143. |
| [7] | Ruifeng Yao, Daoxin Xie. Activation and Termination of Strigolactone Signal Perception in Rice [J]. Chinese Bulletin of Botany, 2024, 59(6): 873-877. |
| [8] | Jinjin Lian, Luyao Tang, Yinuo Zhang, Jiaxing Zheng, Chaoyu Zhu, Yuhan Ye, Yuexing Wang, Wennan Shang, Zhenghao Fu, Xinxuan Xu, Richeng Wu, Mei Lu, Changchun Wang, Yuchun Rao. Genetic Locus Mining and Candidate Gene Analysis of Antioxidant Traits in Rice [J]. Chinese Bulletin of Botany, 2024, 59(5): 738-751. |
| [9] | XU Zi-Yi, JIN Guang-Ze. Variation and trade-offs in fine root functional traits of seedlings of different mycorrhizal types in mixed broadleaf-Korean pine forests [J]. Chin J Plant Ecol, 2024, 48(5): 612-622. |
| [10] | Jiahui Huang, Huimin Yang, Xinyu Chen, Chaoyu Zhu, Yanan Jiang, Chengxiang Hu, Jinjin Lian, Tao Lu, Mei Lu, Weilin Zhang, Yuchun Rao. Response Mechanism of Rice Mutant pe-1 to Low Light Stress [J]. Chinese Bulletin of Botany, 2024, 59(4): 574-584. |
| [11] | Jianmin Zhou. A Combat Vehicle with a Smart Brake [J]. Chinese Bulletin of Botany, 2024, 59(3): 343-346. |
| [12] | GAO Min, GOU Qian-Qian, WANG Guo-Hua, GUO Wen-Ting, ZHANG Yu, ZHANG Yan. Effects of low temperature stress on the physiology and growth of Caragana korshinskii seedlings from different mother tree ages [J]. Chin J Plant Ecol, 2024, 48(2): 201-214. |
| [13] | Chaoyu Zhu, Chengxiang Hu, Zhenan Zhu, Zhining Zhang, Lihai Wang, Jun Chen, Sanfeng Li, Jinjin Lian, Luyao Tang, Qianqian Zhong, Wenjing Yin, Yuexing Wang, Yuchun Rao. Mapping of QTLs Associated with Rice Panicle Traits and Candidate Gene Analysis [J]. Chinese Bulletin of Botany, 2024, 59(2): 217-230. |
| [14] | Zhichao Xu, Meihui Zhu, Zikun Mao, Xugao Wang. Effects of nitrogen addition on seedling dynamics in a broad-leaved Korean pine forest in Northeast China [J]. Biodiv Sci, 2024, 32(12): 24255-. |
| [15] | TAO Qiong, MIAO Ning, YUE Xi-Ming, LUO Jian-Qiong, XUE Pan-Pan, WANG Hui. Influencing factors of biomass accumulation and allocation of Abies fargesii var. faxoniana seedlings in the subalpine region of western Sichuan, China [J]. Chin J Plant Ecol, 2024, 48(11): 1459-1470. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||