

Chinese Bulletin of Botany ›› 2020, Vol. 55 ›› Issue (5): 605-612.DOI: 10.11983/CBB20030 cstr: 32102.14.CBB20030
• TECHNIQUES AND METHODS • Previous Articles Next Articles
Jianfei Liu1, Yan Liu1, Kejian Liu2, Yang Chi3, Zhifa Huo3, Yonghong Huo3, Xiangling You1,*( )
)
Received:2020-02-26
Accepted:2020-06-05
Online:2020-09-01
Published:2020-09-03
Contact:
Xiangling You
Jianfei Liu, Yan Liu, Kejian Liu, Yang Chi, Zhifa Huo, Yonghong Huo, Xiangling You. Optimization of the Regeneration System from Somatic Embryogenesis in Larix olgensis[J]. Chinese Bulletin of Botany, 2020, 55(5): 605-612.
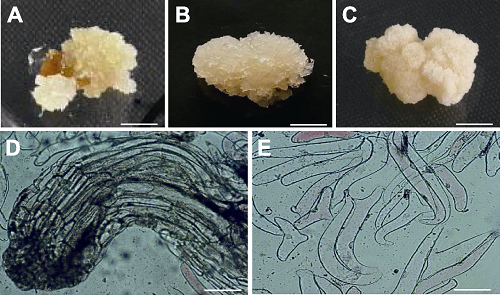
Figure 1 Callus and its microscopic images of Larix olgensis (A) Callus; (B) Embryogenic callus; (C) Non-embryogenic callus; (D) The image of embryogenic callus; (E) The image of non-embryogenic callus. (A)-(C) Bars=3 mm; (D) Bar= 200 μm; (E) Bar=500 μm
| Excellent trees | Callus induction rate (%) | Embryogenic callus formation rate (%) |
|---|---|---|
| 061-1009 | 89.43±48.59 a | 0 |
| 091-1008 | 81.56±47.91 c | 0 |
| 058-842 | 91.25±53.17 a | 4.53±2.73 a |
| 024-922 | 85.91±37.29 b | 0 |
Table 1 Callus and embryogenic callus induction in different Larix olgensis lines (means±SD)
| Excellent trees | Callus induction rate (%) | Embryogenic callus formation rate (%) |
|---|---|---|
| 061-1009 | 89.43±48.59 a | 0 |
| 091-1008 | 81.56±47.91 c | 0 |
| 058-842 | 91.25±53.17 a | 4.53±2.73 a |
| 024-922 | 85.91±37.29 b | 0 |
| The combination of plant growth regulators in the medium | Somatic embryogenesis (%) | Somatic embryo amount (numbers·g-1) |
|---|---|---|
| 0.1 mg?L-1 2,4-D+0.04 mg·L-1 BA+0.02 mg·L-1 KT | 45.27±23.31 | 16.47±1.67 |
| 0.5 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 52.65±31.83 b | 25.31±9.85 c |
| 0.4 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 57.38±15.41 ab | 31.53±5.37 b |
| 0.3 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 64.41±11.79 a | 39.74±4.29 ab |
| 0.2 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 72.36±10.57 a | 48.56±3.77 a |
| 0.1 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 63.70±43.86 a | 41.92±7.22 a |
Table 2 Effects of plant growth regulators at different concentration ratios on somatic embryogenesis and somatic embryo amount in Larix olgensis (means±SD)
| The combination of plant growth regulators in the medium | Somatic embryogenesis (%) | Somatic embryo amount (numbers·g-1) |
|---|---|---|
| 0.1 mg?L-1 2,4-D+0.04 mg·L-1 BA+0.02 mg·L-1 KT | 45.27±23.31 | 16.47±1.67 |
| 0.5 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 52.65±31.83 b | 25.31±9.85 c |
| 0.4 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 57.38±15.41 ab | 31.53±5.37 b |
| 0.3 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 64.41±11.79 a | 39.74±4.29 ab |
| 0.2 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 72.36±10.57 a | 48.56±3.77 a |
| 0.1 mg?L-1 NAA+0.5 mg·L-1 BA+0.5 mg·L-1 KT | 63.70±43.86 a | 41.92±7.22 a |
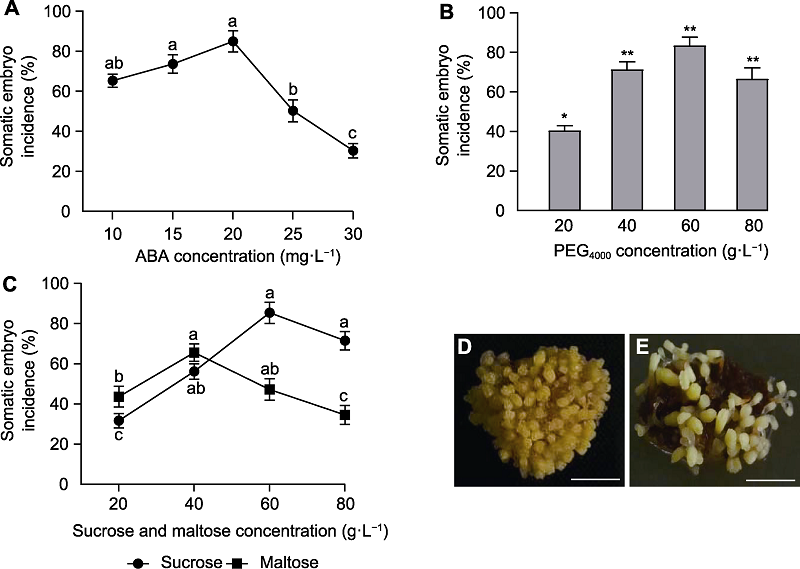
Figure 3 Effects of different factors on the rate of somatic embryogenesis in Larix olgensis (means±SE) (A) ABA; (B) PEG4000; (C) Sucrose and maltose; (D) Cultured with sucrose for 6 weeks; (E) Cultured with maltose for 6 weeks. (D), (E) Bars=2.5 mm. Different lowercase letters indicate significant differences (P<0.05). * P<0.05; ** P<0.01

Figure 4 Somatic embryo seedlings at different developmental states of Larix olgensis and their transplanting (A) Cotyledonary embryo; (B) Cotyledons germinate; (C) Control of somatic embryo seedling; (D) Optimization somatic embryo seedling; (E) Rooting somatic embryo seedlings; (F) Transplanting somatic embryo seedling. (A) Bar=1.5 mm; (B) Bar=6.5 mm; (C), (D) Bars=1.0 cm; (E), (F) Bars=1.5 cm
| Growth regulator combination | Somatic embryo seedlings | |||
|---|---|---|---|---|
| NAA (mg·L-1) | IBA (mg·L-1) | Seedling rate (%) | Stem length (mm) | Main root length (mm) |
| 0.4 | 0.5 | 38.83±29.38 ab | 1.59±1.44 c | 16.38±12.74 b |
| 0.4 | 1.0 | 46.21±41.29 a | 1.75±0.77 ab | 19.17±14.52 a |
| 0.4 | 1.5 | 35.37±33.34 b | 1.67±1.75 b | 12.60±10.34 c |
| 0.8 | 0.5 | 22.57±25.43 c | 1.76±0.91 ab | 12.31±9.54 c |
| 0.8 | 1.0 | 22.65±15.83 c | 1.81±0.88 a | 15.18±13.37 bc |
| 0.8 | 1.5 | 21.43±19.68 c | 1.78±0.67 ab | 11.40±8.53 c |
Table 3 Effects of growth regulator combination on the growth of somatic embryo seedling in Larix olgensis (means±SD)
| Growth regulator combination | Somatic embryo seedlings | |||
|---|---|---|---|---|
| NAA (mg·L-1) | IBA (mg·L-1) | Seedling rate (%) | Stem length (mm) | Main root length (mm) |
| 0.4 | 0.5 | 38.83±29.38 ab | 1.59±1.44 c | 16.38±12.74 b |
| 0.4 | 1.0 | 46.21±41.29 a | 1.75±0.77 ab | 19.17±14.52 a |
| 0.4 | 1.5 | 35.37±33.34 b | 1.67±1.75 b | 12.60±10.34 c |
| 0.8 | 0.5 | 22.57±25.43 c | 1.76±0.91 ab | 12.31±9.54 c |
| 0.8 | 1.0 | 22.65±15.83 c | 1.81±0.88 a | 15.18±13.37 bc |
| 0.8 | 1.5 | 21.43±19.68 c | 1.78±0.67 ab | 11.40±8.53 c |
| Plant regeneration | GA3 concentration (mg·L-1) | ||
|---|---|---|---|
| 0 | 0.5 | 1.0 | |
| Seedling rate (%) | 45.61±52.27 b | 55.44±37.91 a | 45.91±35.81 b |
| Stem length (mm) | 1.63±1.33 b | 2.65±2.61 a | 0.91±1.11 c |
| Main root length (mm) | 17.98±14.22 a | 12.41±16.55 b | 8.65±5.49 c |
Table 4 Effects of different concentration of GA3 on the growth of somatic embryo seedling in Larix olgensis (means± SD)
| Plant regeneration | GA3 concentration (mg·L-1) | ||
|---|---|---|---|
| 0 | 0.5 | 1.0 | |
| Seedling rate (%) | 45.61±52.27 b | 55.44±37.91 a | 45.91±35.81 b |
| Stem length (mm) | 1.63±1.33 b | 2.65±2.61 a | 0.91±1.11 c |
| Main root length (mm) | 17.98±14.22 a | 12.41±16.55 b | 8.65±5.49 c |
| [1] | 黄健秋, 卫志明, 许智宏 ( 1995). 马尾松成熟合子胚的体细胞胚胎发生和植株再生. 科学通报 40, 72-75. |
| [2] | 齐力旺 ( 2000). 华北落叶松体细胞胚胎发生与遗传转化系统建立的研究. 博士论文. 北京: 中国林业科学研究院. pp. 44-74. |
| [3] | 宋跃, 甄成, 张含国, 李淑娟 ( 2016). 长白落叶松胚性愈伤组织诱导及体细胞胚胎发生. 林业科学 52(10), 45-54. |
| [4] | 王伟达, 李成浩, 杨静莉, 张含国, 张淑玲 ( 2009). 杂种落叶松未成熟胚的体细胞胚发生和植株再生. 林业科学 12(8), 34-38. |
| [5] |
Attree SM, Pomeroy MK, Fowke LC ( 1992). Manipulation of conditions for the culture of somatic embryos of white spruce for improved triacylglycerol biosynthesis and desiccation tolerance. Planta 187, 395-404.
DOI URL PMID |
| [6] |
Chalupa V, Durzan DJ, Vithayasai C ( 1976). Growth and metabolism of cells and tissue of Jack pine ( Pinus banksiana). 2. The quantitative analysis of the growth of callus from hypocotyls and radicles. Can J Bot 54, 446-455.
DOI URL |
| [7] |
Durzan DJ, Gupta PK ( 1987). Somatic embryogenesis and polyembryogenesis in Douglas-fir cell suspension cultures. Plant Sci 52, 229-235.
DOI URL |
| [8] |
Hakman I, Fowke LC, Von Arnold S, Eriksson T ( 1985). The development of somatic embryos in tissue cultures initiated from immature embryos of Picea abies (Norway Spruce). Plant Sci 38, 53-59.
DOI URL |
| [9] |
Kim YW, Youn Y, Noh ER, Kim JC ( 1998). Somatic embryogenesis and plant regeneration from immature zygotic embryos of Japanese larch (Larix leptolepis). Plant Cell Tissue Organ Cult 55, 95-101.
DOI URL |
| [10] |
Klimaszewska K ( 1989). Plantlet development from immature zygotic embryos of hybrid larch through somatic embryogenesis. Plant Sci 63, 95-103.
DOI URL |
| [11] | Klimaszewska K, Devantier Y, Lachance D, Lelu MA, Charest PJ ( 1997). Larix laricinar (tamarack): somatic embryogenesis and genetic transformation. Can J For Res 27, 538-550. |
| [12] | Klimaszewska K, Hargreaves C, Lelu-Walter MA, Trontin JF (2016). Advances in conifer somatic embryogenesis since year 2000. In: Germana MA, Lambardi M, eds. In Vitro Embryogenesis in Higher Plants. New York: Humana Press. pp. 131-162. |
| [13] |
Klimaszewska K, Smith DR ( 1997). Maturation of somatic embryos of Pinus strobus is promoted by a high concentration of gellan gum. Physiol Plant 100, 949-957.
DOI URL |
| [14] |
Kvaalen H, von Arnold S ( 1991). Effects of various partial pressures of oxygen and carbon dioxide on different stages of somatic embryogenesis in Picea abies. Plant Cell Tissue Organ Cult 27, 49-57.
DOI URL |
| [15] |
Lelu MA, Klimaszewska K, Charest PJ ( 1994). Somatic embryogenesis from immature and mature zygotic embryos and from cotyledons and needles of somatic plantlets of Larix. Can J For Res 24, 100-106.
DOI URL |
| [16] |
Li QF, Deng C, Zhu TQ, Li JJ, Zhang HG, Kong LS, Zhang SG, Wang JH, Chen XY ( 2019). Dynamics of physiological and miRNA changes after long-term proliferation in somatic embryogenesis of Picea balfouriana. Trees 33, 469-480.
DOI URL |
| [17] |
Lulsdorf MM, Tautorus TE, Kikcio SI, Bethune TD, Dunstan DI ( 1993). Germination of encapsulated embryos of interior spruce (Picea glauca engelmannii complex) and black spruce (Picea mariana Mill.). Plant Cell Rep 12, 385-389.
DOI URL PMID |
| [18] |
Montalbán IA, De Diego N, Moncaleán P ( 2010). Bottlenecks in Pinus radiata somatic embryogenesis: improving maturation and germination. Trees 24, 1061-1071.
DOI URL |
| [19] |
Nunes S, Marum L, Farinha N, Pereira VT, Almeida T, Sousa D, Mano N, Figueiredo J, Dias MC, Santos C ( 2018). Somatic embryogenesis of hybrid Pinus elliottii var. elliottii × P. caribaea var. hondurensis and ploidy assessment of somatic plants. Plant Cell Tissue Organ Cult 132, 71-84.
DOI URL |
| [20] |
Plačková L, Hrdlička J, Smýkalová I, Cvečková M, Novák O, Griga M, Doležal K ( 2015). Cytokinin profiling of long-term in vitro pea (Pisum sativum L.) shoot cultures. Plant Growth Regul 77, 125-132.
DOI URL |
| [21] |
Roberts DR, Sutton BCS, Flinn BS ( 1990). Erratum: synchronous and high frequency germination of interior spruce somatic embryos following partial drying at high relative humidity. Can J Bot 68, 1832.
DOI URL |
| [22] |
Stasolla C, Yeung EC ( 2003). Recent advances in conifer somatic embryogenesis: improving somatic embryo quality. Plant Cell Tissue Organ Cult 74, 15-35.
DOI URL |
| [23] |
Tautorus TE, Fowke LC, Dunstan DI ( 1991). Somatic embryogenesis in conifers. Can J Bot 69, 1873-1899.
DOI URL |
| [24] |
Thompson RG, von Aderkas P ( 1992). Somatic embryogenesis and plant regeneration from immature embryos of western larch. Plant Cell Rep 11, 379-385.
DOI URL PMID |
| [25] | Tret’yakova IN, Barsukova AV ( 2012). Somatic embryogenesis in in vitro culture of three larch species. Russ J Dev Biol 43, 353-361. |
| [26] | von Aderkas P, Bonga JM, Nagmani R ( 1987). Promotion of embryogenesis in cultured megagametophytes of Larix decidua. Can J For Res 17, 1293-1296. |
| [27] | Yang XY, Zhang XL ( 2010). Regulation of somatic embryogenesis in higher plants. Crit Rev Plant Sci 29, 36-57. |
| [1] | Yuchen Li, Haixia Zhao, Xiping Jiang, Xintian Huang, Yaling Liu, Zhenying Wu, Yan Zhao, Chunxiang Fu. Establishment of Agrobacterium-mediated Transformation System for Agropyron mongolicum [J]. Chinese Bulletin of Botany, 2024, 59(4): 600-612. |
| [2] | WEN Xiao-Shi, CHEN Bin-Hang, ZHANG Shu-Bin, XU Kai, YE Xin-Yu, NI Wei-Jie, WANG Xiang-Ping. Relationships of radial growth with climate change in larch plantations of different stand ages and species [J]. Chin J Plant Ecol, 2019, 43(1): 27-36. |
| [3] | Ya-Lin XIE, Hai-Yan WANG, Xiang-Dong LEI. Effects of climate change on net primary productivity in Larix olgensis plantations based on process modeling [J]. Chin J Plan Ecolo, 2017, 41(8): 826-839. |
| [4] | YU Jian,XU Qian-Qian,LIU Wen-Hui,LUO Chun-Wang,YANG Jun-Long,LI Jun-Qing,LIU Qi-Jing. Response of radial growth to climate change for Larix olgensis along an altitudinal gradient on the eastern slope of Changbai Mountain, Northeast China [J]. Chin J Plan Ecolo, 2016, 40(1): 24-35. |
| [5] | Lin Liu, Bin Yu, Pengyan Huang, Jun Jia, Hua Zhao, Junhua Peng, Peng Chen, Liangcai Peng. Frequency of Callus Induction and Plant Regeneration Among Eight Genotypes in Miscanthus sinensis Species [J]. Chinese Bulletin of Botany, 2013, 48(2): 192-198. |
| [6] | Shuan Chen, Xiaodong Wang, Bing Zhao, Yuchun Wang. Regulating the Cell Growth and Shoot Induction of Crocus sativus Embryogenic Callus by Rare Earth Elements [J]. Chinese Bulletin of Botany, 2010, 45(05): 609-614. |
| [7] | LENG Wen-Fang, HE Hong-Shi, BU Ren-Cang, HU Yuan-Man. SENSITIVITY ANALYSIS OF THE IMPACTS OF CLIMATE CHANGE ON POTENTIAL DISTRIBUTION OF THREE LARCH (LARIX) SPECIES IN NORTHEASTERN CHINA [J]. Chin J Plant Ecol, 2007, 31(5): 825-833. |
| [8] | SUN Zhi-Hu, MU Chang-Cheng. THE ESTIMATE OF FINE ROOT BIOMASS IN UPPER SOIL LAYER OF LARIX OLGENSIS PLANTATION BY GEOSTATISTICS METHOD [J]. Chin J Plant Ecol, 2006, 30(5): 771-779. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||