

Chinese Bulletin of Botany ›› 2020, Vol. 55 ›› Issue (2): 182-191.DOI: 10.11983/CBB19169 cstr: 32102.14.CBB19169
• TECHNIQUES AND METHODS • Previous Articles Next Articles
Hua Zhao1,Guangda Shao1,2,Wenxin Gao1,2,Biao Gu1,2,*( )
)
Received:2019-08-28
Accepted:2019-11-28
Online:2020-03-01
Published:2020-02-12
Contact:
Biao Gu
Hua Zhao,Guangda Shao,Wenxin Gao,Biao Gu. The Application of Double-barreled Particle Bombardment for Transient Gene Expression in Arabidopsis[J]. Chinese Bulletin of Botany, 2020, 55(2): 182-191.

Figure 1 Transient expression of GFP and GUS reporter genes in Arabidopsis thaliana and Nicotiana benthamiana leaves via double-barreled particle bombardment (A) The rosette leaves (red arrow pointed) of A. thaliana were selected for transient gene expression (Bar=5 mm); (B) Observation of green fluorescent reporter gene (GFP) with fluorescent microscopy (Bar=50 μm); (C), (D) Transient expression of GUS in A. thaliana and N. benthamiana leaves, respectively. Numbers of GUS positive spots are indicated in black numbers (Bars=5 mm).
| Primer name | Forward primer (5'-3') | Reverse primer (5'-3') |
|---|---|---|
| GFP | CTAGCCCCGGGATGGTGAGCAAGGGCGAG | CTGAGGTACCTTACTTGTACAGCTCGTC |
| GUS | CTAGCCCCGGGATGGTAGATCTGAGGAACC | CTGAGGTACCTCACACGTGATGGTGATGG |
| BAX | CTAGCCCCGGGATGGACGGGTCCGGGGAG | GTGCAGGATCCCTGGAAGAAGATGGGCTGA |
| Avh238 | CTAGCCCCGGGATGGAGGCGAAGCTCTTG | GTGCAGGATCCGTTGGCCAAGCCTGTGTAG |
| Avrblb1 | CTAGCCCCGGGATGGTTTCATCCAATCTCAAC | GTGCAGGTACCCTAGCTAGGGCCAACGTTTTTATC |
| RB | CTAGCCCCGGGATGGCTGAAGCTTTCATTCAAG | GTGCAGGATCCTTAAATATATATATTCACATTAG |
| ATR13 | CTAGCCCCGGGATGAATCTGCTCCACGCCCATG | GTGCAGGATCCTTACTGACTGGCAACGGCAGTC |
| Rpp13 | CTAGCCCCGGGATGGTAGATGCGATCACGGAG | GTGCAGGATCCCTAAACGCTCGCAATCGGTTTG |
| qRB | GAGAAGAGCAATTCCCTGTGC | GTTAGGTGCTGCAATCCCTCTG |
| qUBC9 | CATCGGATAGCCCTTATTCTG | TGGAACACCTTCGTCCTAAAA |
Table 1 Primer sequences of PCR and qRT-PCR
| Primer name | Forward primer (5'-3') | Reverse primer (5'-3') |
|---|---|---|
| GFP | CTAGCCCCGGGATGGTGAGCAAGGGCGAG | CTGAGGTACCTTACTTGTACAGCTCGTC |
| GUS | CTAGCCCCGGGATGGTAGATCTGAGGAACC | CTGAGGTACCTCACACGTGATGGTGATGG |
| BAX | CTAGCCCCGGGATGGACGGGTCCGGGGAG | GTGCAGGATCCCTGGAAGAAGATGGGCTGA |
| Avh238 | CTAGCCCCGGGATGGAGGCGAAGCTCTTG | GTGCAGGATCCGTTGGCCAAGCCTGTGTAG |
| Avrblb1 | CTAGCCCCGGGATGGTTTCATCCAATCTCAAC | GTGCAGGTACCCTAGCTAGGGCCAACGTTTTTATC |
| RB | CTAGCCCCGGGATGGCTGAAGCTTTCATTCAAG | GTGCAGGATCCTTAAATATATATATTCACATTAG |
| ATR13 | CTAGCCCCGGGATGAATCTGCTCCACGCCCATG | GTGCAGGATCCTTACTGACTGGCAACGGCAGTC |
| Rpp13 | CTAGCCCCGGGATGGTAGATGCGATCACGGAG | GTGCAGGATCCCTAAACGCTCGCAATCGGTTTG |
| qRB | GAGAAGAGCAATTCCCTGTGC | GTTAGGTGCTGCAATCCCTCTG |
| qUBC9 | CATCGGATAGCCCTTATTCTG | TGGAACACCTTCGTCCTAAAA |
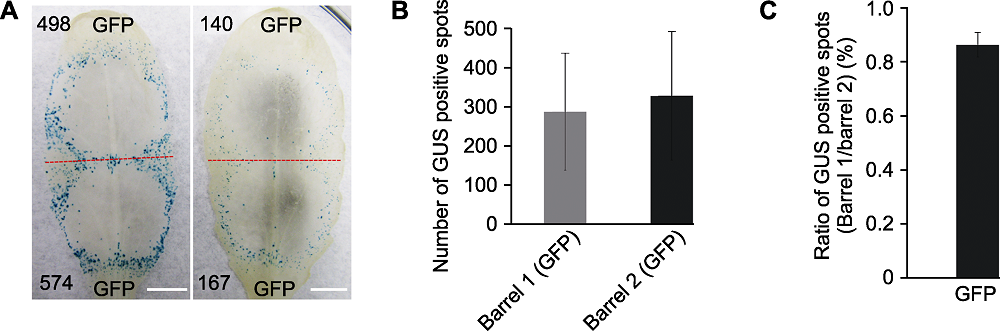
Figure 2 The efficiency of transient expression of GUS gene in different rosette leaves of Arabidopsis thaliana (A) Co-bombardment of GFP/GUS gene mixture in different rosette leaves, the red dotted line separates two positions produced by double-barreled particle bombardment, numbers of GUS blue spots are indicated in black numbers (Bars=5 mm); (B) Statistic analysis of GUS spots counted from the tip of leaf blade (Barrel 1) and petiole (Barrel 2) (error bar represents ± SD, P>0.1); (C) Ratios of GUS spot numbers in Barrel 1 and Barrel 2 of the same leaf (error bar represents ± SD). Ten rosette leaves from two A. thaliana plants were tested and the test was repeated twice. Statistical analysis by Wilcoxon rank sum test using log ratios of GUS spot numbers in Barrel 1 and Barrel 2.
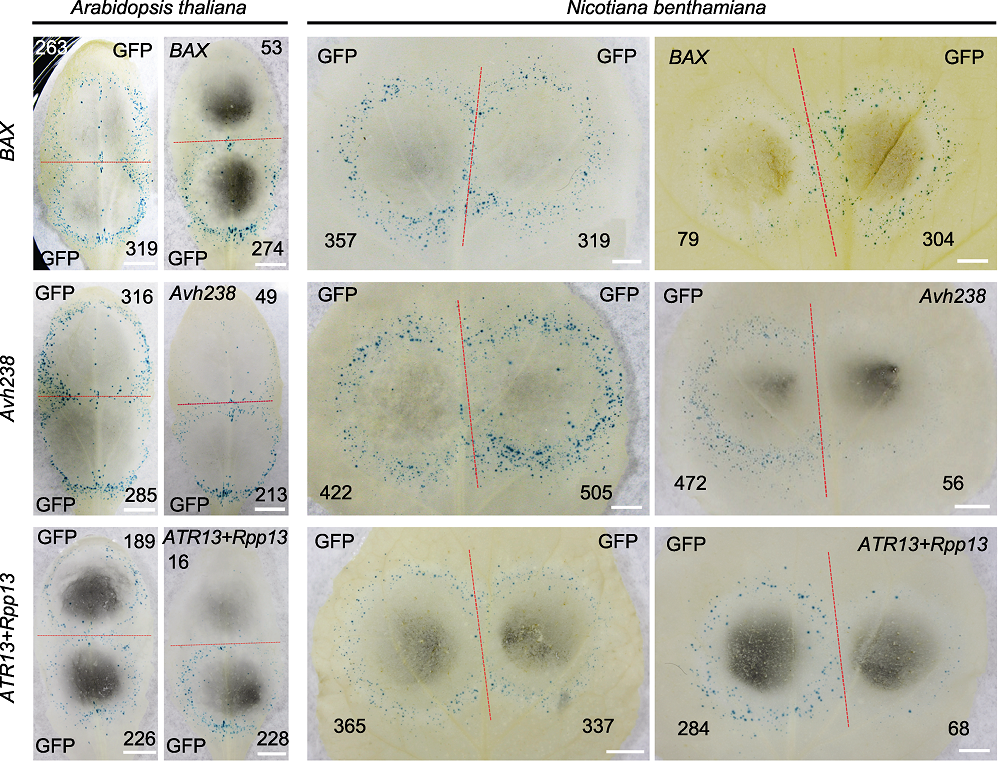
Figure 3 Cell death induction by various pathogen effectors in Arabidopsis thaliana and Nicotiana benthamiana leaves Mammalian apoptosis inducer BAX, RxLR effector gene Avh238 of Phytophthora sojae, effector gene ATR13 of Hyaloperonospora arabidopsidis and disease resistance gene Rpp13 of A. thaliana gene pairs were transiently expressed in leaves of A. thaliana and N. benthamiana by double-barreled particle bombardment, cell necrosis was indicated by GUS blue spots, with GFP as control. The red dotted line separates two positions produced by co-bombardment, numbers of GUS spots are indicated in black numbers (Bars=5 mm).
| BAX ± SD | Avh238 ± SD | ATR13/Rpp13 ± SD | |||
|---|---|---|---|---|---|
| GFP/GFP | BAX/GFP | GFP/GFP | Avh238/GFP | ATR13 + Rpp13/GFP | |
| Arabidopsis thaliana | 0.92±0.13 | 0.23±0.13* | 0.84±0.17 | 0.94±0.15 | 0.22±0.08* |
| Nicotiana benthamiana | 1.05±0.23 | 0.13±0.07* | 0.92±0.21 | 0.94±0.10 | 0.22±0.05* |
Table 2 Measurement of cell death induction activities by BAX, Avh238 and ATR13/Rpp13 in Arabidopsis thaliana and Nicotiana benthamiana
| BAX ± SD | Avh238 ± SD | ATR13/Rpp13 ± SD | |||
|---|---|---|---|---|---|
| GFP/GFP | BAX/GFP | GFP/GFP | Avh238/GFP | ATR13 + Rpp13/GFP | |
| Arabidopsis thaliana | 0.92±0.13 | 0.23±0.13* | 0.84±0.17 | 0.94±0.15 | 0.22±0.08* |
| Nicotiana benthamiana | 1.05±0.23 | 0.13±0.07* | 0.92±0.21 | 0.94±0.10 | 0.22±0.05* |
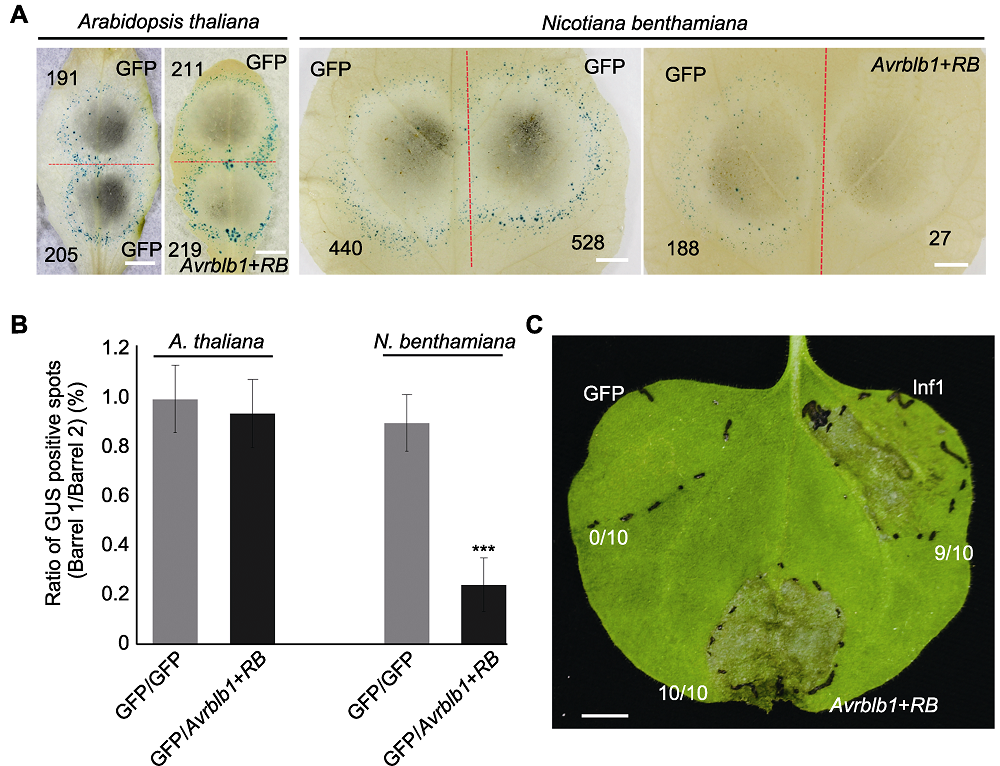
Figure 4 Cell death induction triggered by Avrblb1 of Phytophthora infestans and RB of Solanum tuberosum in Arabidopsis thaliana and Nicotiana benthamiana leaves (A) Co-bombardment mediated transient expression of Avrblb1/Rpp13 gene pair in A. thaliana and N. benthamiana leaves, the red dotted line separates two positions produced by co-bombardment, numbers of GUS spots are indicated in black numbers (Bars=5 mm); (B) Ratio of GUS positive spots (ten leaves from each plant were tested, and the test was repeated twice, error bar represents ± SD). P values for treatments and the control were calculated from the log ratios using the Wilcoxon rank sum test, *** indicates extremely significant differences (P<0.001); (C) Agrobacterium-mediated transient expression of Avrblb1/RB in N. benthamiana leaves, GFP and Inf1 were used as negative and positive control, respectively. Pictures were taken 4 days post infiltration, each treatment contains 10 replicates (Bar=5 mm).
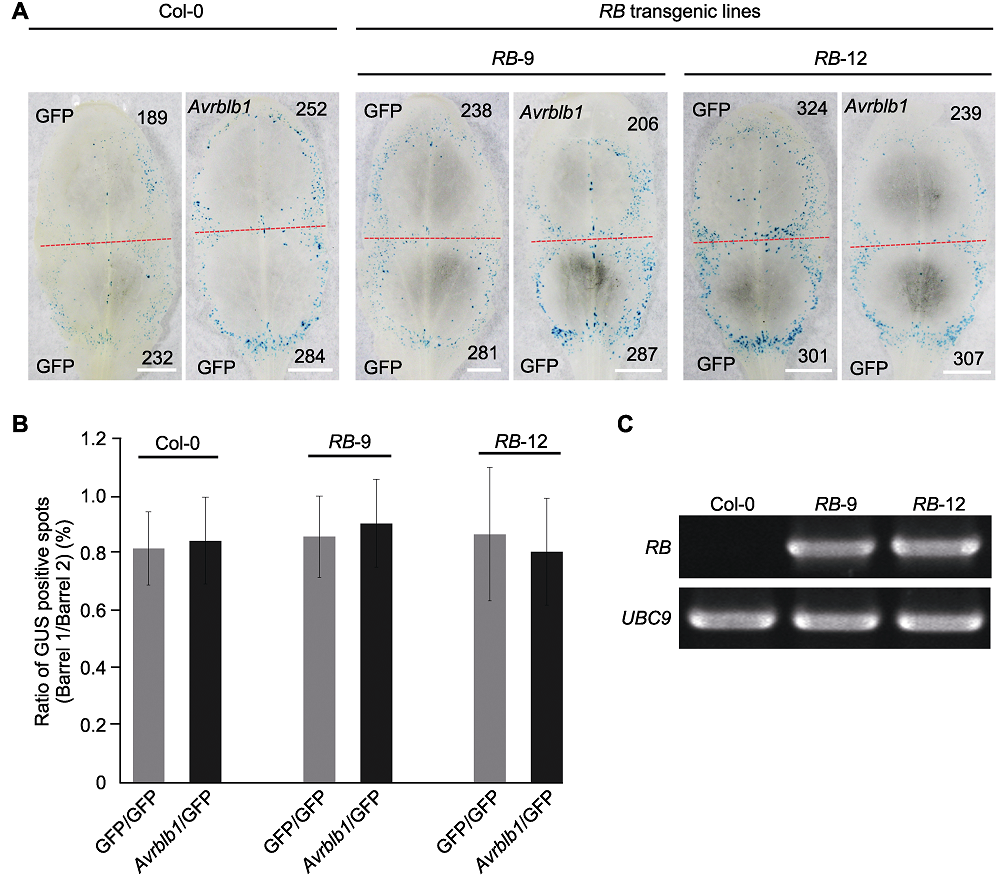
Figure 5 Measurement of cell death induction by effector gene Avrblb1 in Arabidopsis transgenic line RB-9 and RB-12 (A) Co-bombardment mediated transient expression of Avrblb1 in transgenic lines RB-9 and RB-12, with GFP as control (the red dotted line separates two positions produced by co-bombardment, numbers of GUS spots are indicated in black numbers (Bars=5 mm)); (B) The diagram showed that transient expression of Avrblb1 in RB transgenic lines (ten leaves of each plant were tested, and the test was repeated twice, P values for treatments and the control were calculated from the log ratios using the Wilcoxon rank sum test (error bar represents ± SD, P>0.1)); (C) The RT-PCR detection of RB expressed in transgenic line RB-9 and RB-12, UBC9 was used as the reference gene.
| [1] | Champouret N, Bouwmeester K, Rietman H, van der Lee T, Maliepaard C, Heupink A, van de Vondervoort PJI, Jacobsen E, Visser RGF, van der Vossen EAG, Govers F, Vleeshouwers VGAA ( 2009). Phytophthora infestans isolates lacking class I ipiO variants are virulent on Rpi-blb1 potato. Mol Plant Microbe Interact 22, 1535-1545. |
| [2] | Coll NS, Vercammen D, Smidler A, Clover C, Van Breusegem F, Dangl JF, Epple P ( 2010). Arabidopsis type I metacaspases control cell death. Science 330, 1393-1397. |
| [3] | Dong SM, Yin WX, Kong GH, Yang XY, Qutob D, Chen QH, Kale SD, Sui YY, Zhang ZG, Dou DL, Zheng XB, Gijzen M, Tyler BM, Wang YC ( 2011). Phytophthora sojae avirulence effector Avr3b is a secreted NADH and ADP-ribose pyrophosphorylase that modulates plant immunity. PLoS Pathog 7, e1002353. |
| [4] | Dou DL, Kale SD, Wang XL, Chen YB, Wang QQ, Wang X, Jiang RHY, Arredondo FD, Anderson RG, Thakur PB, McDowell JM, Wang YC, Tyler BM ( 2008). Conserved C-terminal motifs required for avirulence and suppression of cell death by Phytophthora sojae effector Avr1b. Plant Cell 20, 1118-1133. |
| [5] | Gu B, Kale SD, Wang QH, Wang DH, Pan QN, Cao H, Meng YL, Kang ZS, Tyler BM, Shan WX ( 2011). Rust secreted protein Ps87 is conserved in diverse fungal pathogens and contains a RXLR-like motif sufficient for translocation into plant cells. PLoS One 6, e27217. |
| [6] | Kale SD, Tyler BM ( 2011). Assaying effector function in Planta using double-barreled particle bombardment. In: McDowell J, ed. Plant Immunity. Methods in Molecular Biology (Methods and Protocols), Vol. 712. New York: Humana Press. pp. 153-172. |
| [7] | Koornneef M, Meinke D ( 2010). The development of Arabidopsis as a model plant. Plant J 61, 909-921. |
| [8] | Mangano S, Gonzalez CD, Petruccelli S ( 2014). Agrobacterium tumefaciens-mediated transient transformation of Arabidopsis thaliana leaves. In: Sanchez-Serrano JJ, Salinas J, eds. Arabidopsis Protocols, Methods in Molecular Biology (Methods and Protocols), Vol. 1062. Totowa, NJ: Humana Press. pp. 165-173. |
| [9] | Sohn KH, Lei R, Nemri A, Jones JDG ( 2007). The downy mildew effector proteins ATR1 and ATR13 promote disease susceptibility in Arabidopsis thaliana. Plant Cell 19, 4077-4090. |
| [10] | Tsuda K, Qi YP, Nguyen LV, Bethke G, Tsuda Y, Glazebrook J, Katagiri F ( 2012). An efficient Agrobacterium-mediated transient transformation of Arabidopsis. Plant J 69, 713-719. |
| [11] | Ueki S, Lacroix B, Krichevsky A, Lazarowitz SG, Citovsky V ( 2009). Functional transient genetic transformation of Arabidopsis leaves by biolistic bombardment. Nat Protoc 4, 71-77. |
| [12] | Wang QQ, Han CZ, Ferreira AO, Yu XL, Ye WW, Tripathy S, Kale SD, Gu B, Sheng YT, Sui YY, Wang XL, Zhang ZG, Cheng BP, Dong SM, Shan WX, Zheng XB, Dou DL, Tyler BM, Wang YC ( 2011a). Transcriptional programming and functional interactions within the Phytophthora sojae RXLR effector repertoire. Plant Cell 23, 2064-2086. |
| [13] | Wang XJ, Tang CL, Zhang HC, Xu JR, Liu B, Lv J, Han DJ, Huang LL, Kang ZS ( 2011b). TaDAD2, a negative regulator of programmed cell death, is important for the interaction between wheat and the stripe rust fungus. Mol Plant Microbe Interact 24, 79-90. |
| [14] | Weigel D, Glazebrook J ( 2011). Arabidopsis: a Laboratory Manual. New York: Cold Spring Harbor Laboratory Press. pp. 119-140. |
| [15] | Yang B, Wang QQ, Jing MF, Guo BD, Wu JW, Wang HN, Wang Y, Lin L, Wang Y, Ye WW, Dong SM, Wang YC ( 2017). Distinct regions of the Phytophthora essential effector Avh238 determine its function in cell death activation and plant immunity suppression. New Phytol 214, 361-375. |
| [16] | Yoo SD, Cho YH, Sheen J ( 2007). Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2, 1565-1572. |
| [1] | Xuemin Cao, Ying Bao, Yuexin Zhang, Ruijie Li, Jianxin Su, Wei Zhang. Tissue Culture, Rapid Propagation and Efficient Transient Expression Systems of Rosa multiflora [J]. Chinese Bulletin of Botany, 2025, 60(2): 235-245. |
| [2] | Yanjun Jing, Rongcheng Lin. Blue Light Receptor CRY2 Transforms into a ‘dark dancer’ [J]. Chinese Bulletin of Botany, 2024, 59(6): 878-882. |
| [3] | Yan Luo, Qiyuan Liu, Yuanbing Lü, Yue Wu, Yaoyu Tian, Tian An, Zhenhua Li. Photothermal Sensitivity of Phytochrome Mutants During Seed Germination in Arabidopsis thaliana [J]. Chinese Bulletin of Botany, 2024, 59(5): 752-762. |
| [4] | Qiuxin Li, Wei Chi, Daili Ji. Research Progress of CURT1 on Regulating Thylakoid Membrane Curvature [J]. Chinese Bulletin of Botany, 2021, 56(4): 462-469. |
| [5] | Fei Du, Yuling Jiao. WUSCHEL-mediated Innate Immunity in Plant Stem Cells Provides a Novel Antiviral Strategy [J]. Chinese Bulletin of Botany, 2020, 55(5): 537-540. |
| [6] | Liang Wu, Yijun Qi. Small RNA, No Small Feat: Plants Deploy 22 nt siRNAs to Cope with Environmental Stress [J]. Chinese Bulletin of Botany, 2020, 55(3): 270-273. |
| [7] | Qingping Zhao,Shifan Ma,Ruixi Li,Tianyu Wang,Xiang Zhao. Advances of NPH3/RPT2-Like (NRL) Family Proteins in Phototropin-mediated Signaling in Arabidopsis thaliana [J]. Chinese Bulletin of Botany, 2020, 55(2): 240-253. |
| [8] | Gaoping Qu,Jingbo Jin. Detection of SUMOylation in Plants [J]. Chinese Bulletin of Botany, 2020, 55(1): 83-89. |
| [9] | Yan Zhao,Jianmin Zhou. Luciferase Complementation Assay for Detecting Protein Interactions [J]. Chinese Bulletin of Botany, 2020, 55(1): 69-75. |
| [10] | Lijing Liu,Qingzhen Zhao,Qi Xie,Feifei Yu. An Quick and Efficient Assay for In Vivo Protein Ubiquitination [J]. Chinese Bulletin of Botany, 2019, 54(6): 753-763. |
| [11] | Aihua Song, Wenbin Zhang, Shulan Sun, Lingfei Li, Xiaojing Wang. Preparation of Protoplast and Establishment of Transient Expression System in Gerbera hybrida [J]. Chinese Bulletin of Botany, 2017, 52(4): 511-519. |
| [12] | Xiao Li, Haiyan Sun, Mengbin Ruan, Peihong Wang, Ming Peng. Studies of the Transient Expression and Transformation of Cloned Thermostable α-Amylase Genes from Bacillus licheniformis in Tobacco and Arabidopsis [J]. Chinese Bulletin of Botany, 2015, 50(3): 354-362. |
| [13] | Xuemei Liu, Lei Wang, Tianlong Wen, Peng He, Jianing Yu. The Establishment of Agrobacterium-mediated Transient Expression System in Cotton Cotyledons [J]. Chinese Bulletin of Botany, 2014, 49(5): 587-594. |
| [14] | Wei Xie Chaoyin Yue Zhenghong Guo Zhipeng Dai Min Liu Wei Yao. Transient Expression of GUS Gene Controlled by Different Regulator Sequences of Tobacco [J]. Chinese Bulletin of Botany, 2007, 24(04): 452-458. |
| [15] | JI Feng_Yuan, WANG Ge_Liang, XU Yi_Nong. THE EFFECTS OF ANTIOXIDANTS ON THE TRANSIENT EXPRESSION OF GUS GENE IN SOYBEAN HYPOCOTYLS MEDIATED BY AGROBACTERIUM TUMEFACIENS [J]. Chin J Plant Ecol, 2006, 30(2): 330-334. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||