

Chinese Bulletin of Botany ›› 2018, Vol. 53 ›› Issue (5): 661-670.DOI: 10.11983/CBB17141 cstr: 32102.14.CBB17141
• TECHNIQUES AND METHODS • Previous Articles Next Articles
Zeng Yinwei, Cao Yuman, Sha Xuyang, Li Shuxia, Yang Peizhi, Hu Tianming, Liu Jinlong*( )
)
Received:2017-07-30
Accepted:2017-12-05
Online:2018-09-01
Published:2018-11-29
Contact:
Liu Jinlong
About author:† These authors contributed equally to this paper
Zeng Yinwei, Cao Yuman, Sha Xuyang, Li Shuxia, Yang Peizhi, Hu Tianming, Liu Jinlong. An Observation Method of Nodule and Root Morphology without Damage in Real-time[J]. Chinese Bulletin of Botany, 2018, 53(5): 661-670.
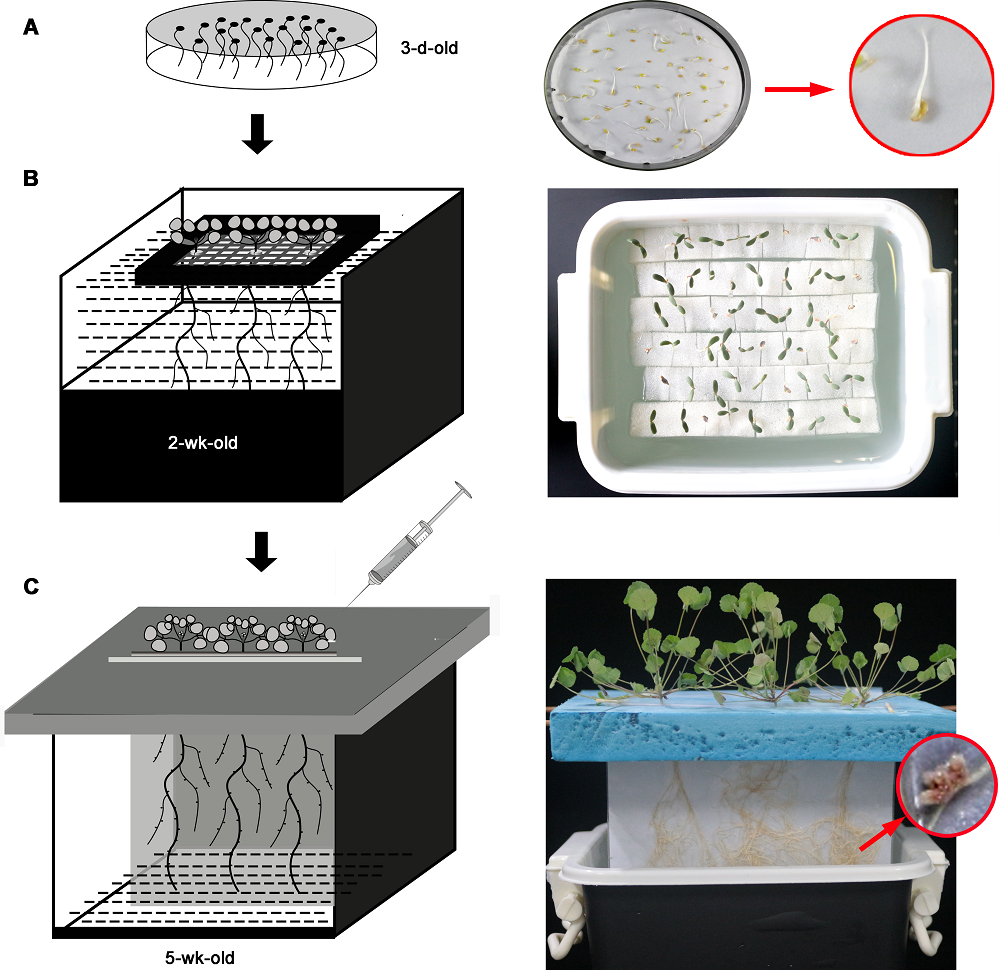
Figure 1 Diagram of paper-based Ziplock bag hydroponics system (right side is the picture of the actual object)(A) Seed germination; (B) Water culture seedlings; (C) Paper-based Ziplock bag hydroponics device
| Treatment | Number of nodule per plant | Lateral root length (cm) | Number of lateral root per plant | Angle of lateral root with primary root (°) | Secondary lateral root |
|---|---|---|---|---|---|
| Control | 70.7±2.5 | 4.1±0.2 | 86.3±4.2 | 52.3±2.1 | YES |
| Melatonin (50 μmol·mL-1) | 21.3±1.5*** | 1.7±0.2*** | 143.3±7.1*** | 85.7±3.1*** | NO |
Table 1 Effects of melatonin (50 μmol·mL-1) treatment on lateral root development and the inoculation of nodules in Medicago truncatula (means±SD, n=12)
| Treatment | Number of nodule per plant | Lateral root length (cm) | Number of lateral root per plant | Angle of lateral root with primary root (°) | Secondary lateral root |
|---|---|---|---|---|---|
| Control | 70.7±2.5 | 4.1±0.2 | 86.3±4.2 | 52.3±2.1 | YES |
| Melatonin (50 μmol·mL-1) | 21.3±1.5*** | 1.7±0.2*** | 143.3±7.1*** | 85.7±3.1*** | NO |

Figure 2 Effects of different culture methods on inoculation of nodules and root morphology of Medicago truncatula(A)-(C) Sand culture method, (A) Sand culture device; (B) Root morphology and nodules; (C) The lost roots; (D)-(F) Hydroponics, (D) Hydroponics; (E) Root morphology; (F) The enlarged image of local roots and nodules of Figure E; (G)-(I) Aeroponics, (G) Aeroponics device; (H) Root morphology; (I) The enlarged image of local roots and nodules of Figure H; (J)-(M) Paper-based Ziplock bag hydroponics method, (J) Above-ground parts of M. truncatula (the left side is R108, the right is A17); (K) Root morphology; (L) The whole plants of M. truncatula; (M) The enlarged image of local roots and nodules of Figure L. Red arrows for the nodules, green arrows for the quartz sand; The M. truncatula is 6 weeks old except Figure J and K which is 7 weeks old.
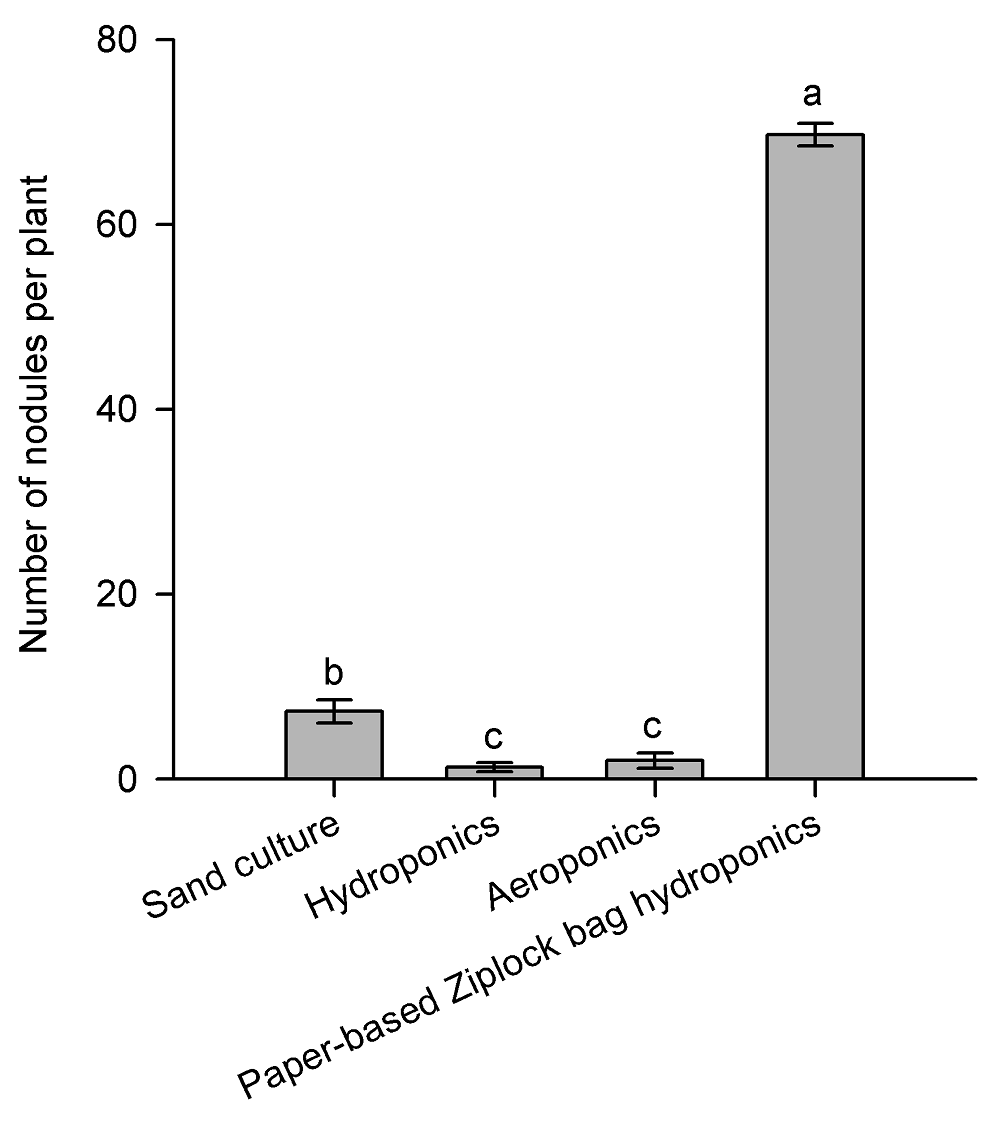
Figure 3 Effects of different culture methods on the number of inoculated nodules in Medicago truncatula (means±SD, n=12)Different lowercase letters represent significant difference among different methods (P<0.05).
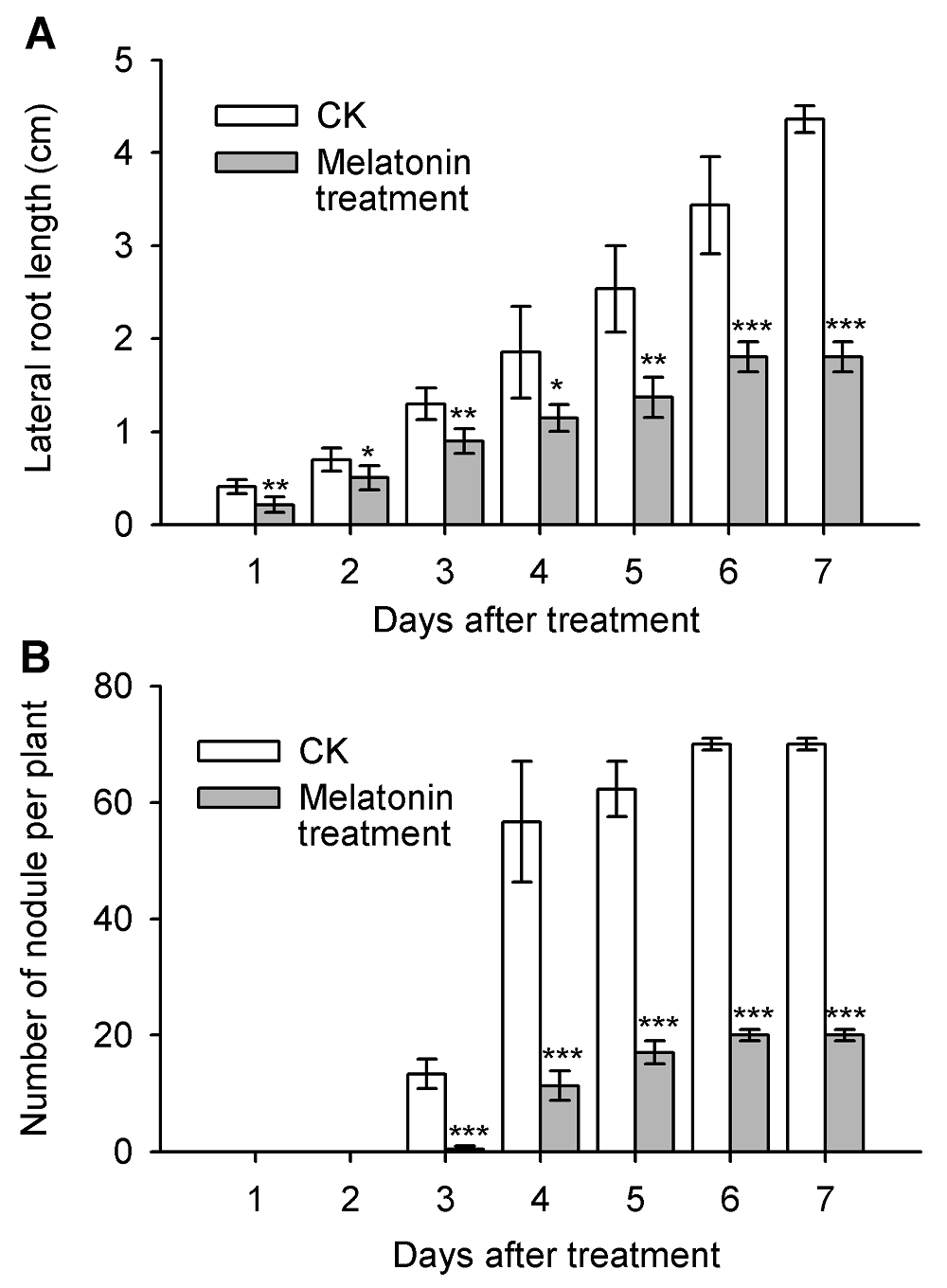
Figure 5 Effects of melatonin (50 μmol·mL-1) on nodule formation and lateral root development of Medicago truncatula(A) Dynamic changes of lateral root growth; (B) The formation of nodules. * Represent significant differences at P<0.05; ** Represent highly significant differences at P<0.01; *** Represent extremely significant differences at P<0.001.
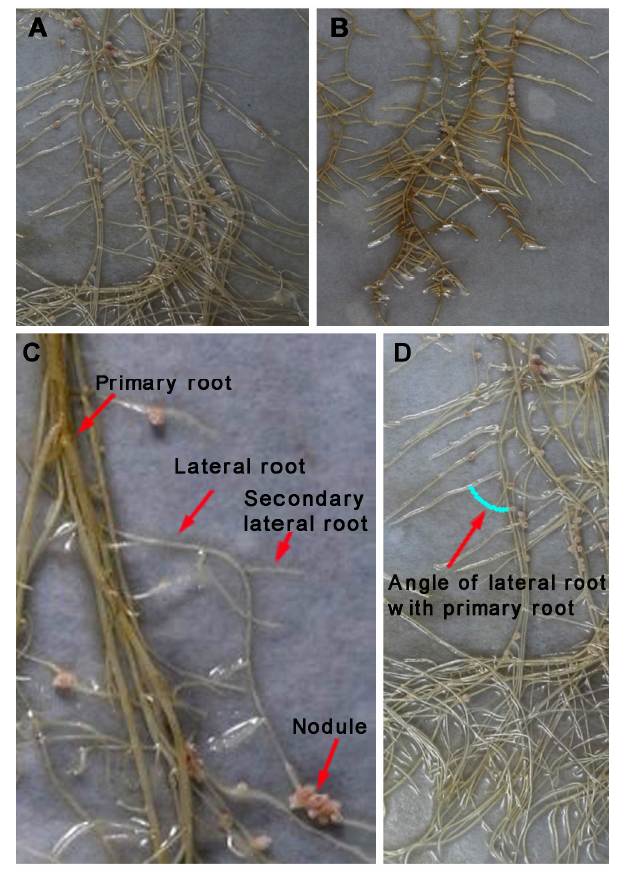
Figure 6 Effect of melatonin (50 μmol?mL–1) on the root system of Medicago truncatula (6 weeks old)(A) Control; (B) Melatonin treatment; (C) Primary root, lateral root, secondary lateral root and nodules; (D) Angle of lateral root with primary root
| 1 | 陈文新, 陈文峰 (2004). 发挥生物固氮作用减少化学氮肥用量. 中国农业科技导报 6(6), 3-6. |
| 2 | 褚贵新, 沈其荣, 曹金留, 茆泽圣, 钟增涛, 赵龙 (2003). 旱作水稻与花生间作系统中的氮素固定与转移及其对土壤肥力的影响. 土壤学报 40, 717-723. |
| 3 | 梁如玉, 李登煜 (1984). 用试管水培法鉴定豆科作物的结瘤性能. 土壤肥料 (3), 37-38. |
| 4 | 章家恩, 高爱霞, 徐华勤, 罗明珠 (2009). 玉米/花生间作对土壤微生物和土壤养分状况的影响. 应用生态学报 20, 1597-1602. |
| 5 | Arnao MB, Hernández-Ruiz J (2007). Melatonin promotes adventitious-and lateral root regeneration in etiolated hypocotyls of Lupinus albus L. J Pineal Res 42, 147-152. |
| 6 | Arnao MB, Hernández-Ruiz J (2015). Functions of melatonin in plants: a review.J Pineal Res 59, 133-150. |
| 7 | Bergersen FJ (1965). Ammonia? An early stable product of nitrogen fixation by soybean root nodules F.J. Bergersen.Aust J Biol Sci 18, 1-9. |
| 8 | Clark RT, Famoso AN, Zhao KY, Shaff JE, Craft EJ, Bustamante CD, McCouch SR, Aneshansley DJ, Kochian LV (2013). High-throughput two-dimensional root system phenotyping platform facilitates genetic analysis of root growth and development.Plant Cell Environ 36, 454-466. |
| 9 | Danso SKA, Alexander M (1974). Survival of two strains of Rhizobium in soil. Soil Sci Soc Am J 38, 86-89. |
| 10 | De M Rangel W, De Oliveira Longatti SM, Ferreira PAA, Bonaldi DS, Guimarães AA, Thijs S, Weyens N, Vangronsveld J, Moreira FMS (2017). Leguminosae native nodulating bacteria from a gold mine As-contaminated soil: multi-resistance to trace elements, and possible role in plant growth and mineral nutrition.Int J Phytoremediation 19, 925-936. |
| 11 | Di Giacomo E, Laffont C, Sciarra F, Iannelli MA, Frugier F, Frugis G (2017). KNAT3/4/5-like class 2 KNOX transcription factors are involved in Medicago truncatula symbiotic nodule organ development. New Phytol 213, 822-837. |
| 12 | Dickstein R, Hu XJ, Yang J, Ba L, Coque L, Kim DJ, Cook DR, Yeung AT (2002). Differential expression of tandemly duplicated Enod8 genes in Medicago. Plant Sci 163, 333-343. |
| 13 | Famoso AN, Clark RT, Shaff JE, Craft E, McCouch SR, Kochian LV (2010). Development of a novel aluminum tolerance phenotyping platform used for comparisons of cereal aluminum tolerance and investigations into rice aluminum tolerance mechanisms.Plant Physiol 153, 1678-1691. |
| 14 | Farías-Rodríguez R, Mellor RB, Arias C, Peña-Cabriales JJ (1998). The accumulation of trehalose in nodules of several cultivars of common bean (Phaseolus vulgaris) and its correlation with resistance to drought stress. Phy- siol Plantarum 102, 353-359. |
| 15 | Hund A, Trachsel S, Stamp P (2009). Growth of axile and lateral roots of maize: I development of a phenotying platform.Plant Soil 325, 335-349. |
| 16 | Jahnke S, Menzel MI, Van Dusschoten D, Roeb GW, Bühler J, Minwuyelet S, Blümler P, Temperton VM, Hombach T, Streun M, Beer S, Khodaverdi M, Ziemons K, Coenen HH, Schurr U (2009). Combined MRI-PET dissects dynamic changes in plant structures and functions.Plant J 59, 634-644. |
| 17 | Jeudy C, Adrian M, Baussard C, Bernard C, Bernaud E, Bourion V, Busset H, Cabrera-Bosquet L, Cointault F, Han SM, Lamboeuf M, Moreau D, Pivato B, Prudent M, Trouvelot S, Truong HN, Vernoud V, Voisin AS, Wipf D, Salon C (2016). RhizoTubes as a new tool for high throughput imaging of plant root development and architecture: test, comparison with pot grown plants and validation.Plant Methods 12, 31. |
| 18 | Jeudy C, Ruffel S, Freixes S, Tillard P, Santoni AL, Morel S, Journet EP, Duc G, Gojon A, Lepetit M, Salon C (2010). Adaptation of Medicago truncatula to nitrogen limi- tation is modulated via local and systemic nodule developmental responses. New Phytol 185, 817-828. |
| 19 | Johnson HW, Means UM, Weber CR (1965). Competition for nodule sites between strains of Rhizobium japonicum applied as inoculum and strains in the soil. Agron J 57, 179-185. |
| 20 | Keyser HH, Bohlool BB, Hu TS, Weber DF (1982). Fast- growing rhizobia isolated from root nodules of soybean.Science 215, 1631-1632. |
| 21 | Kiers ET, Rousseau RA, West SA, Denison RF (2003). Host sanctions and the legume-rhizobium mutualism.Nature 425, 78-81. |
| 22 | Kistner C, Parniske M (2002). Evolution of signal transduction in intracellular symbiosis.Trends Plant Sci 7, 511-518. |
| 23 | Kuchenbuch RO, Ingram KT, Buczko U (2006). Effects of decreasing soil water content on seminal lateral roots of young maize plants.J Plant Nutr Soil Sci 169, 841-848. |
| 24 | Labidi N, Mahmoudi H, Dorsaf M, Slama I, Abdelly C (2009). Assessment of intervarietal differences in drought tolerance in chickpea using both nodule and plant traits as indicators.J Plant Breed Crop Sci 1(4), 80-86. |
| 25 | Le Marié C, Kirchgessner N, Marschall D, Walter A, Hund A (2014). Rhizoslides: paper-based growth system for non-destructive, high throughput phenotyping of root development by means of image analysis.Plant Methods 10, 13. |
| 26 | Liang CZ, Li AF, Yu H, Li WZ, Liang CZ, Guo SD, Zhang R, Chu CC (2017). Melatonin regulates root architecture by modulating auxin response in rice.Front Plant Sci 8, 134. |
| 27 | Mooney SJ, Pridmore TP, Helliwell J, Bennett MJ (2012). Developing X-ray computed tomography to non-invasively image 3-D root systems architecture in soil.Plant Soil 352, 1-22. |
| 28 | Nagel KA, Putz A, Gilmer F, Heinz K, Fischbach A, Pfeifer J, Faget M, Blossfeld S, Ernst M, Dimaki C, Kastenholz B, Kleinert AK, Galinski A, Scharr H, Fiorani F, Schurr U (2012). GROWSCREEN-Rhizo is a novel phenotyping robot enabling simultaneous measurements of root and shoot growth for plants grown in soil-filled rhizotrons.Funct Plant Biol 39, 891-904. |
| 29 | Nap JP, Bisseling T (1990). Developmental biology of a plant-prokaryote symbiosis: the legume root nodule.Science 250, 948-954. |
| 30 | O'Gara F, Shanmugam KT (1976). Regulation of nitrogen fixation by rhizobia export of fixed N2 as NH4+.Biochim Biophys Acta 437, 313-321. |
| 31 | Patterson III WA, Olson JJ (1983). Effects of heavy metals on radicle growth of selected woody species germinated on filter paper, mineral and organic soil substrates.Can J Forest Res 13, 233-238. |
| 32 | Pelagio-Flores R, Muñoz-Parra E, Ortiz-Castro R, López- Bucio J (2012). Melatonin regulates Arabidopsis root system architecture likely acting independently of auxin signaling.J Pineal Res 53, 279-288. |
| 33 | Planchamp C, Balmer D, Hund A, Mauch-Mani B (2013). A soil-free root observation system for the study of root-micro- organism interactions in maize.Plant Soil 367, 605-614. |
| 34 | Popp C, Ott T (2011). Regulation of signal transduction and bacterial infection during root nodule symbiosis.Curr Opin Plant Biol 14, 458-467. |
| 35 | Reddy CS, Pattanaik C, Mohapatra A, Biswal AK (2007). Phytosociological observations on tree diversity of tropical forest of Similipal Biosphere Reserve, Orissa, India.TAIWANIA 52, 352-359. |
| 36 | Reddy PM, Rendón-Anaya M, de los Dolores del Río M, Khandual S (2007). Flavonoids as signaling molecules and regulators of root nodule development.Dyn Soil Dyn Plant 1, 83-94. |
| 37 | Reimer R, Stich B, Melchinger AE, Schrag TA, Sørensen AP, Stamp P, Hund A (2013). Root response to temperature extremes: association mapping of temperate maize (Zea mays L). Maydica 58, 156-168. |
| 38 | Rispail N, Dita MA, González-Verdejo C, Pérez-de-Luque A, Castillejo MA, Prats E, Román B, Jorrín J, Rubiales D (2007). Plant resistance to parasitic plants: molecular approaches to an old foe.New Phytol 173, 703-712. |
| 39 | Robinson D, Hodge A, Griffiths BS, Fitter AH (1999). Plant root proliferation in nitrogen-rich patches confers competitive advantage.Proc Roy Soc B-Biol Sci 266, 431-435. |
| 40 | Ruta N, Stamp P, Liedgens M, Fracheboud Y, Hund A (2010). Collocations of QTLs for seedling traits and yield components of tropical maize under water stress conditions.Crop Sci 50, 1385-1392. |
| 41 | Ryle GJA, Powell CE, Gordon AJ (1978). Effect of source of nitrogen on the growth of Fiskeby soya bean: the carbon economy of whole plants.Ann Bot 42, 637-648. |
| 42 | Smit AL, Groenwold J (2005). Root characteristics of selected field crops: data from the Wageningen Rhizolab (1990-2002).Plant Soil 272, 365-384. |
| 43 | Smith S, De Smet I (2012). Root system architecture: insights from Arabidopsis and cereal crops.Philos Trans Roy Soc B Biol Sci 367,1441-1452. |
| 44 | Sparks DL (2003). Environmental Soil Chemistry, 2nd edn. London: Academic Press. pp. 75-133. |
| 45 | Stracke S, Kistner C, Yoshida S, Mulder L, Sato S, Kaneko T, Tabata S, Sandal N, Stougaard J, Szczyglowski K, Parniske M (2002). A plant receptor-like kinase required for both bacterial and fungal symbiosis.Nature 417, 959-962. |
| 46 | Streeter JG, Salminen SO (1992). Evidence supporting a non-phloem source of water for export of solutes in the xylem of soybean root nodules.Plant Cell Environ 15, 735-741. |
| 47 | Tilman D, Cassman KG, Matson PA, Naylor R, Polasky S (2002). Agricultural sustainability and intensive production practices.Nature 418, 671-677. |
| 48 | Truchet G, Barker DG, Camut S, De Billy F, Vasse J, Huguet T (1989). Alfalfa nodulation in the absence of Rhizobium. Mol Gen Genet 219, 65-68. |
| 49 | Udvardi MK, Day DA (1997). Metabolite transport across symbiotic membranes of legume nodules.Annu Rev Plant Biol 48, 493-523. |
| 50 | Zahran HH (1999). Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol Mol Biol Rev 63, 968-989. |
| 51 | Zhu JM, Kaeppler SM, Lynch JP (2005). Mapping of QTLs for lateral root branching and length in maize (Zea mays L.) under differential phosphorus supply. Theor Appl Genet 111, 688-695. |
| 52 | Zipfel C, Oldroyd GED (2017). Plant signaling in symbiosis and immunity.Nature 543, 328-336. |
| [1] | QU Ze-Kun, ZHU Li-Qin, JIANG Qi, WANG Xiao-Hong, YAO Xiao-Dong, CAI Shi-Feng, LUO Su-Zhen, sCHEN Guang-Shui. Nutrient foraging strategies of arbuscular mycorrhizal tree species in a subtropical evergreen broadleaf forest and their relationship with fine root morphology [J]. Chin J Plant Ecol, 2024, 48(4): 416-427. |
| [2] | Jinyu Du, Zhen Sun, Yanlong Su, Heping Wang, Yaling Liu, Zhenying Wu, Feng He, Yan Zhao, Chunxiang Fu. Identification and Functional Analysis of an Agropyron mongolicum Caffeic Acid 3-O-methyltransferase Gene AmCOMT1 [J]. Chinese Bulletin of Botany, 2024, 59(3): 383-396. |
| [3] | LI Lin, SUN Yi, YANG Xiao-Qiong, FANG Hai-Dong, YAN Bang-Guo. Response of endophytes in root nodules of Arachis hypogaea ‘Qicai’ to nitrogen addition and its relationship with plant stoichiometry characteristics [J]. Chin J Plant Ecol, 2024, 48(10): 1374-1384. |
| [4] | Cai Shuyu, Liu Jianxin, Wang Guofu, Wu Liyuan, Song Jiangping. Regulatory Mechanism of Melatonin on Tomato Seed Germination Under Cd2+ Stress [J]. Chinese Bulletin of Botany, 2023, 58(5): 720-732. |
| [5] | Qi Zhang, Wenjing Zhang, Xiankai Yuan, Ming Li, Qiang Zhao, Yanli Du, Jidao Du. The Regulatory Mechanism of Melatonin on Nucleic Acid Repairing of Common Bean (Phaseolus vulgaris) at the Sprout Stage Under Salt Stress [J]. Chinese Bulletin of Botany, 2023, 58(1): 108-121. |
| [6] | Jinxiang Ai, Jiayi Song, Zhenan Yan, Zhichao Wang, Wenqian Chen, Yuhuan Wu, Yanyan Wang, Leilei Pan, Yutao Xu, Peng Liu. Effects of Exogenous Melatonin on Physiological Response and DNA Damage of Ardisia mamillata and A. crenata Under Lead Stress [J]. Chinese Bulletin of Botany, 2022, 57(2): 171-181. |
| [7] | Deshuai Liu, Lei Yao, Weirong Xu, Mei Feng, Wenkong Yao. Research Progress of Melatonin in Plant Stress Resistance [J]. Chinese Bulletin of Botany, 2022, 57(1): 111-126. |
| [8] | MA Ju-Feng, XIN Min, XU Chen-Chao, ZHU Wan-Ying, MAO Chuan-Zao, CHEN Xin, CHENG Lei. Effects of arbuscular mycorrhizal fungi and nitrogen addition on nitrogen uptake of rice genotypes with different root morphologies [J]. Chin J Plant Ecol, 2021, 45(7): 728-737. |
| [9] | LI Qiang, HUANG Ying-Xin, ZHOU Dao-Wei, CONG Shan. Mechanism of the trade-off between biological nitrogen fixation and phosphorus acquisition strategies of herbaceous legumes under nitrogen and phosphorus addition [J]. Chin J Plant Ecol, 2021, 45(3): 286-297. |
| [10] | Chengwu Liu, Zhong Zhao. The Legume SHR-SCR Module Predetermines Nodule Founder Cell Identity [J]. Chinese Bulletin of Botany, 2020, 55(6): 661-665. |
| [11] | ZOU Xian-Hua, HU Ya-Nan, WEI Dan, CHEN Si-Tong, WU Peng-Fei, MA Xiang-Qing. Correlation between endogenous hormone and the adaptability of Chinese fir with high phosphorus-use efficiency to low phosphorus stress [J]. Chin J Plant Ecol, 2019, 43(2): 139-151. |
| [12] | Guan-Tao CHEN, Yong PENG, Jun ZHENG, Shun LI, Tian-Chi PENG, Xi-Rong QIU, Li-Hua TU. Effects of short-term nitrogen addition on fine root biomass, lifespan and morphology of Castanopsis platyacantha in a subtropical secondary evergreen broad-leaved forest [J]. Chin J Plant Ecol, 2017, 41(10): 1041-1050. |
| [13] | ZHOU Hong-Hua,LI Wei-Hong. Responses and adaptation of xylem hydraulic conductivity to salt stress in Populus euphratica [J]. Chin J Plan Ecolo, 2015, 39(1): 81-91. |
| [14] | ZHENG Ya-Ping,XIN Cai-Yun,WANG Cai-Bin,SUN Xiu-Shan,YANG Wei-Qiang,WAN Shu-Bo,ZHENG Yong-Mei,FENG Hao,CHEN Dian-Xu,SUN Xue-Wu,WU Zheng-Feng. Effects of phosphorus fertilizer on root morphology, physiological characteristics and yield in peanut (Arachis hypogaea) [J]. Chin J Plant Ecol, 2013, 37(8): 777-785. |
| [15] | MA Xiao-Dong, ZHU Cheng-Gang, LI Wei-Hong. Response of root morphology and biomass of Tamarix ramosissima seedlings to different water irrigations [J]. Chin J Plant Ecol, 2012, 36(10): 1024-1032. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||