

植物学报 ›› 2018, Vol. 53 ›› Issue (6): 773-781.DOI: 10.11983/CBB17195 cstr: 32102.14.CBB17195
孙建飞, 翟建云, 马元丹*( ), 傅卢成, 卜柯丽, 王柯杨, 高岩, 张汝民
), 傅卢成, 卜柯丽, 王柯杨, 高岩, 张汝民
收稿日期:2017-10-22
出版日期:2018-11-01
发布日期:2018-12-05
通讯作者:
马元丹
作者简介:作者简介:白克智, 1959年开始在中国科学院植物研究所工作, 先后任助理研究员、研究员, 长期从事植物生长发育及其调控的研究。1986年,其主持的“满江红生物学特性研究”荣获中国科学院科技进步二等奖。曾任《植物生理学报》编委、《植物学报》常务编委、中国植物生长调节剂协会主任等职。
基金资助:
Sun Jianfei, Zhai Jianyun, Ma Yuandan*( ), , Fu Lucheng, Bu Keli, Wang Keyang, Gao Yan, Zhang Rumin
), , Fu Lucheng, Bu Keli, Wang Keyang, Gao Yan, Zhang Rumin
Received:2017-10-22
Online:2018-11-01
Published:2018-12-05
Contact:
Ma Yuandan
摘要: 为揭示毛竹(Phyllostachys edulis)快速生长期茎秆中的光合碳同化特征及其在不同节间的变化规律, 以毛竹笋竹茎秆为材料, 测定不同节间光合色素含量、核酮糖-1,5-二磷酸羧化酶/加氧酶(Rubisco)、磷酸烯醇式丙酮酸羧化酶(PEPC)、苹果酸脱氢酶(NADP-MDH)、NADP-苹果酸酶(NADP-ME)、磷酸烯醇式丙酮酸羧激酶(PEPCK)以及丙酮酸磷酸双激酶(PPDK)活性。结果显示, 茎秆中叶绿素a、叶绿素b以及类胡萝卜素含量随节间升高均呈下降趋势, 叶绿素a/b比值呈逐渐上升趋势; 随着节间的升高, 茎秆中Rubisco、PEPC和PPDK活性在第1-10节间显著下降, 之后酶活性降幅逐渐减缓; NADP-ME活性在第1-13节间呈显著下降趋势, 之后酶活性趋于平稳; NADP-MDH活性在第1-25节间显著下降。PEPC/Rubisco活性比值随节间升高而不断增加, 其范围介于18.37-65.09之间, 明显大于典型C3植物中的活性比值。上述结果表明, 茎秆不同节间的光合碳同化能力存在明显差异, 中、下部节间生长相对较快; 茎秆中存在多种C4酶且活性较高, 这为此时期茎秆中存在C4光合途径提供了有力证据。
孙建飞, 翟建云, 马元丹, 傅卢成, 卜柯丽, 王柯杨, 高岩, 张汝民. 毛竹快速生长期茎秆不同节间光合色素和光合酶活性的差异. 植物学报, 2018, 53(6): 773-781.
Sun Jianfei, Zhai Jianyun, Ma Yuandan, , Fu Lucheng, Bu Keli, Wang Keyang, Gao Yan, Zhang Rumin. Differences in Photosynthetic Pigments and Photosynthetic Enzyme Activities in Different Internodes of Phyllostachys edulis During Rapid Growth Stage. Chinese Bulletin of Botany, 2018, 53(6): 773-781.
| Internode number | Chlorophyll a (μg·g -1 FW) | Chlorophyll b (μg·g -1 FW) | Total chlorophyll (μg·g -1 FW) | Carotenoids (μg·g -1 FW) | Chlorophyll a/b |
|---|---|---|---|---|---|
| 1 | 15.19±0.28 a | 5.07±0.04 a | 20.26±0.29 a | 6.37±0.27 a | 3.00±0.24 c |
| 4 | 16.22±0.27 a | 5.27±0.40 a | 21.49±0.39 a | 7.00±0.28 a | 3.10±0.27 c |
| 7 | 15.80±1.09 a | 5.21±0.20 a | 21.01±1.28 a | 6.66±0.48 a | 3.03±0.10 c |
| 10 | 13.98±0.42 b | 3.91±0.11 b | 17.89±0.53 b | 6.27±0.32 a | 3.58±0.02 c |
| 13 | 9.51±0.63 c | 2.71±0.03 c | 12.22±0.61 c | 4.27±0.19 b | 3.50±0.25 c |
| 16 | 6.49±0.29 d | 1.70±0.09 d | 8.19±0.23 d | 2.89±0.17 c | 3.83±0.35 bc |
| 19 | 2.43±0.07 e | 0.53±0.02 e | 2.96±0.09 e | 1.23±0.09 d | 4.59±0.1 ab |
| 22 | 1.83±0.15 e | 0.38±0.03 e | 2.22±0.16 e | 0.97±0.05 d | 4.82±0.55 a |
| 25 | 1.25±0.01 e | 0.25±0.01 e | 1.50±0.01 e | 0.63±0.09 d | 5.07±0.23 a |
表 1 毛竹茎秆不同节间内光合色素含量的差异(平均值±标准差)
Table 1 Differences of pigment contents in the Phyllostachys edulis stems of different internodes (means±SD)
| Internode number | Chlorophyll a (μg·g -1 FW) | Chlorophyll b (μg·g -1 FW) | Total chlorophyll (μg·g -1 FW) | Carotenoids (μg·g -1 FW) | Chlorophyll a/b |
|---|---|---|---|---|---|
| 1 | 15.19±0.28 a | 5.07±0.04 a | 20.26±0.29 a | 6.37±0.27 a | 3.00±0.24 c |
| 4 | 16.22±0.27 a | 5.27±0.40 a | 21.49±0.39 a | 7.00±0.28 a | 3.10±0.27 c |
| 7 | 15.80±1.09 a | 5.21±0.20 a | 21.01±1.28 a | 6.66±0.48 a | 3.03±0.10 c |
| 10 | 13.98±0.42 b | 3.91±0.11 b | 17.89±0.53 b | 6.27±0.32 a | 3.58±0.02 c |
| 13 | 9.51±0.63 c | 2.71±0.03 c | 12.22±0.61 c | 4.27±0.19 b | 3.50±0.25 c |
| 16 | 6.49±0.29 d | 1.70±0.09 d | 8.19±0.23 d | 2.89±0.17 c | 3.83±0.35 bc |
| 19 | 2.43±0.07 e | 0.53±0.02 e | 2.96±0.09 e | 1.23±0.09 d | 4.59±0.1 ab |
| 22 | 1.83±0.15 e | 0.38±0.03 e | 2.22±0.16 e | 0.97±0.05 d | 4.82±0.55 a |
| 25 | 1.25±0.01 e | 0.25±0.01 e | 1.50±0.01 e | 0.63±0.09 d | 5.07±0.23 a |
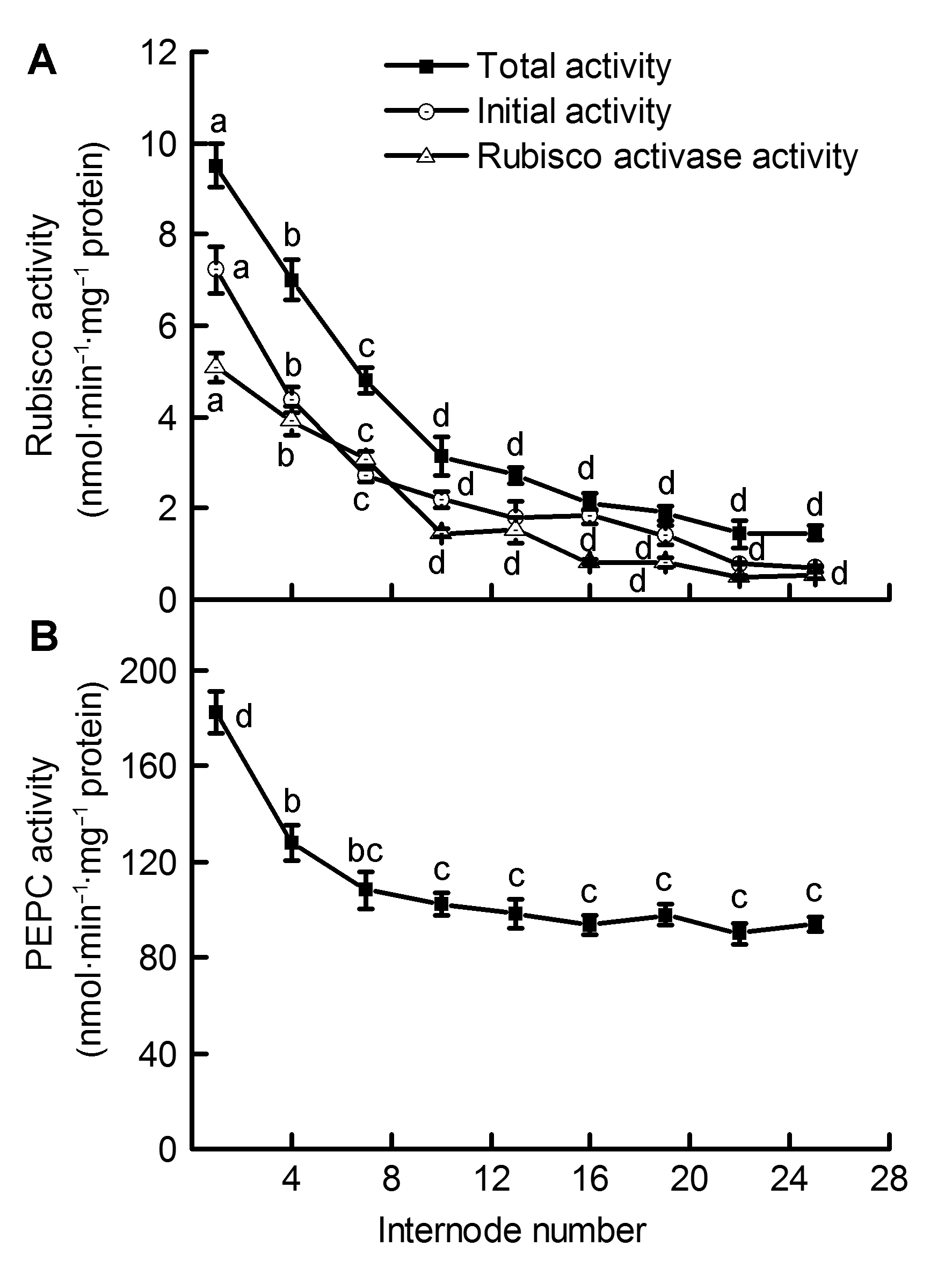
图 1 毛竹茎秆不同节间内核酮糖-1,5-二磷酸羧化酶/加氧酶(Rubisco)活性(A)和磷酸烯醇式丙酮酸羧化酶(PEPC)活性(B)的差异(平均值±标准差)
Figure 1 Differences of Ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) activity (A) and Phosphoenolpyruvate carboxylase (PEPC) activity (B) in the Phyllostachys edulis stems of different internodes (means±SD)
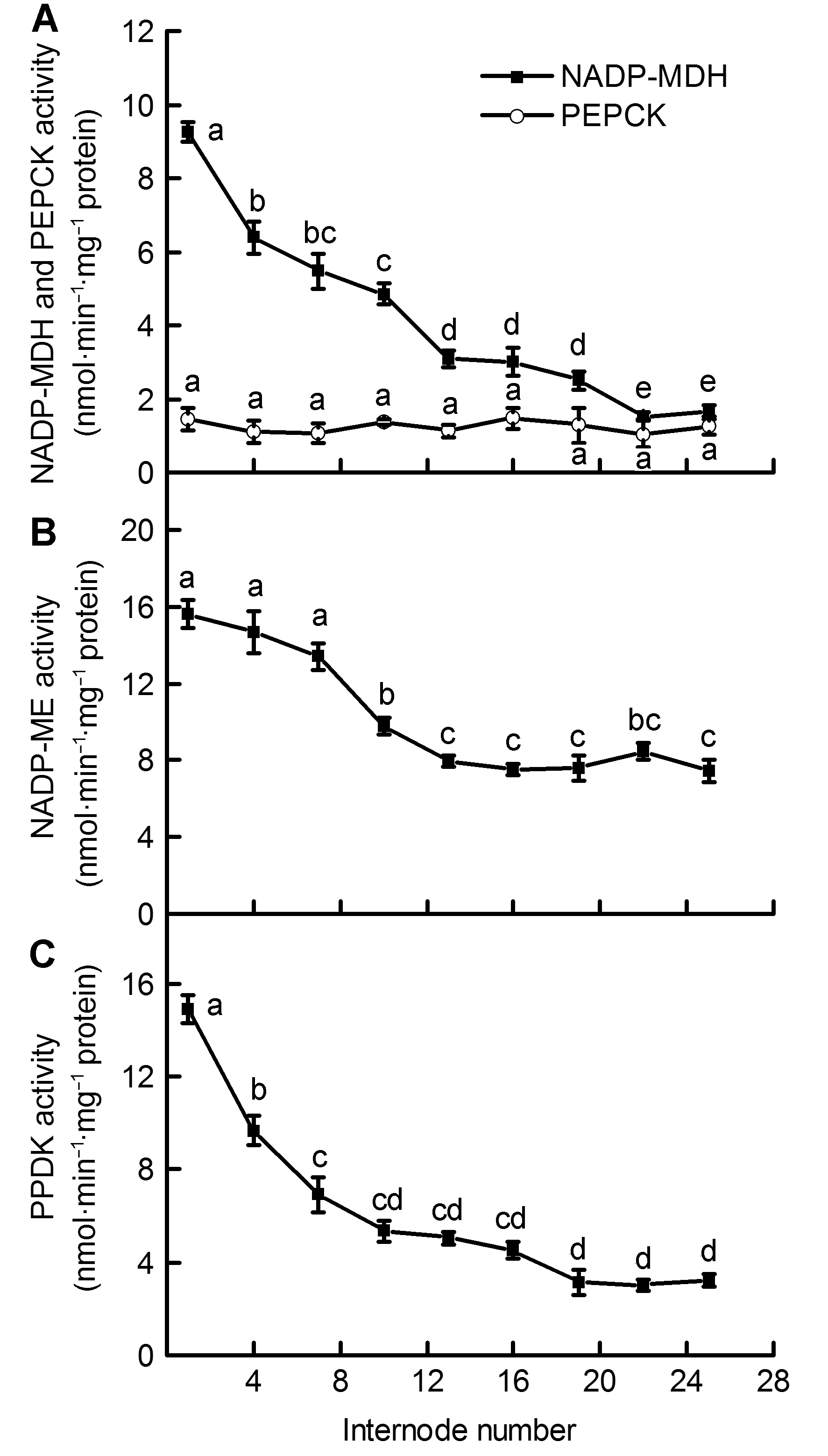
图 2 毛竹茎秆不同节间内4种C4酶活性的差异(平均值±标准差) (A) NADP-苹果酸脱氢酶(NADP-MDH)和磷酸烯醇式丙酮酸羧激酶(PEPCK)活性; (B) NADP-苹果酸酶(NADP-ME)活性; (C) 丙酮酸磷酸双激酶(PPDK)活性
Figure 2 Differences of activities of 4 C4 enzymes in the Phyllostachys edulis stems of different internodes (means± SD) (A) NADP-malate dehydrogenase (NADP-MDH) activity and Phosphoenolpyruvate carboxy-kinase (PEPCK) activity; (B) NADP-malic enzyme (NADP-ME) activity; (C) Pyruvate, orthophosphate dikinase (PPDK) activity
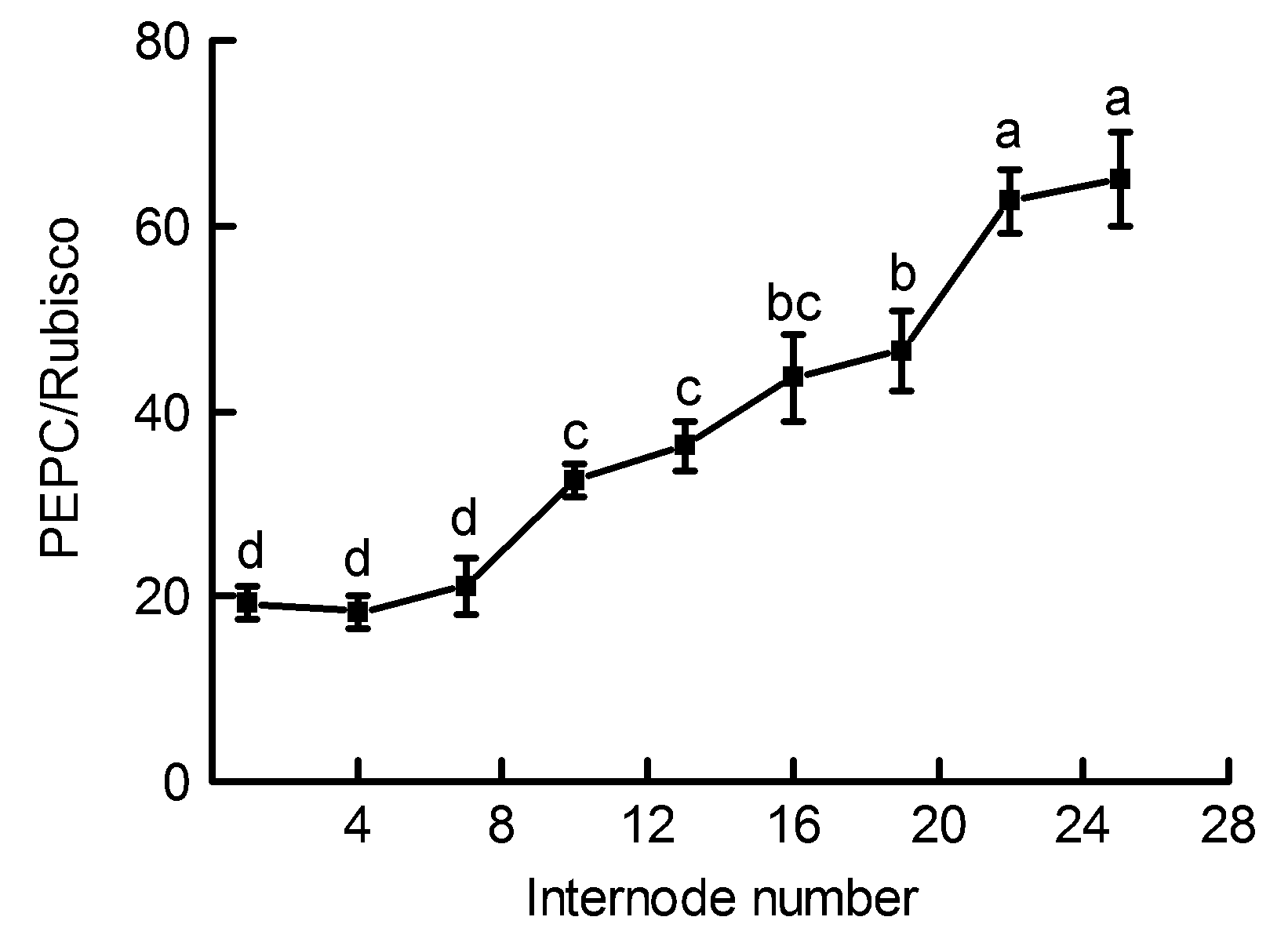
图 3 毛竹茎秆不同节间内磷酸烯醇式丙酮酸羧化酶和核酮糖-1,5-二磷酸羧化酶/加氧酶(PEPC/Rubisco)比值的差异(平均值± 标准差)
Figure 3 Changes of the ratios of Phosphoenolpyruvate carboxylase/Ribulose-1,5-bisphosphate carboxylase/oxygen- ase (PEPC/Rubisco) in the Phyllostachys edulis stems of different internodes (means±SD)
| [1] |
陈登举, 高培军, 吴兴波, 高岩, 温国胜, 王玉魁, 高荣孚, 张汝民 (2013). 毛竹茎秆叶绿体超微结构及其发射荧光光谱特征. 植物学报 48, 635-642.
DOI URL |
| [2] |
高苏娟, 谢修志, 陈兆平, 黄志刚, 赵琦, 王小菁 (2009). 蓝光调节高粱突变体har1幼苗的去黄化反应. 植物学报 44, 69-78.
DOI URL |
| [3] |
龚春梅, 宁蓬勃, 王根轩, 梁宗锁 (2009). C3和C4植物光合途径的适应性变化和进化. 植物生态学报 33, 206-221.
DOI URL |
| [4] | 郝迺斌, 谭克辉, 那松青, 贾志旺, 戈巧英, 张玉竹, 杜维广 (1991). C3植物绿色器官PEP羧化酶活性的比较研究. 植物学报 33, 692-697. |
| [5] | 姜振升, 孙晓琦, 艾希珍, 王美玲, 毕焕改, 王洪涛 (2010). 低温弱光对黄瓜幼苗Rubisco与Rubisco活化酶的影响. 应用生态学报 21, 2045-2050. |
| [6] |
孙建磊, 王崇启, 肖守华, 高超, 李利斌, 曹齐卫, 王晓, 董玉梅, 焦自高 (2017). 弱光对黄瓜幼苗光合特性及Rubisco酶的影响. 核农学报 31, 1200-1209.
DOI URL |
| [7] | 王文杰, 许慧男, 王莹, 于兴洋, 郑广宇, 祖元刚 (2010). 盐碱地土壤改良对银中杨叶片、树枝和树皮绿色组织色素和C4光合酶的影响. 植物研究 30, 299-304. |
| [8] |
王文杰, 祖元刚, 王慧梅 (2007). 林木非同化器官树枝(干)光合功能研究进展. 生态学报 27, 1583-1595.
DOI URL |
| [9] |
王星星, 刘琳, 张洁, 王玉魁, 温国胜, 高荣孚, 高岩, 张汝民 (2012). 毛竹出笋后快速生长期内茎秆中光合色素和光合酶活性的变化. 植物生态学报 36, 456-462.
DOI URL |
| [10] | 王莹, 王文杰, 许慧男, 郑广宇, 孙伟, 祖元刚 (2011). 3种C3木本植物绿色组织C4酶活性、色素含量及叶绿素荧光参数的比较. 植物研究 31, 461-466. |
| [11] |
徐超华, 李军营, 崔明昆, 马二登, 黄国宾, 龚明 (2013). 延长光照时间对烟草叶片生长发育及光合特性的影响. 西北植物学报 33, 763-770.
DOI URL |
| [12] |
杨春菊, 陈永刚, 汤孟平, 施拥军, 侯建花, 孙燕飞 (2016). 不同管理模式下毛竹幼竹的生长规律. 植物学报 51, 774-781.
DOI URL |
| [13] |
张金尧, 刘俊祥, 巨关升, 韩蕾, 孙振元 (2014). 旱柳非叶光合组织(皮层)叶绿体光合特性. 林业科学 50, 30-35.
DOI URL |
| [14] | 张汝民 (2005). 绿豆幼苗脱黄化初期质体发育生理生化机制的研究. 博士论文. 北京: 北京林业大学. pp. 45-47. |
| [15] |
庄明浩, 李迎春, 郭子武, 杨清平, 顾李俭, 陈双林, 邓宗付 (2013). 大气CO2与O3浓度升高对毛竹叶片膜脂过氧化和抗氧化系统的影响. 西北植物学报 33, 322-328.
DOI URL |
| [16] | 祖元刚, 张衷华, 王文杰, 杨逢建, 贺海升 (2006). 薇甘菊叶和茎的光合特性. 植物生态学报 30, 998-1004. |
| [17] |
Aschan G, Pfanz H (2003). Non-foliar photosynthesis—a strategy of additional carbon acquisition.Flora 198, 81-97.
DOI URL |
| [18] |
Aschan G, Wittmann C, Pfanz H (2001). Age-dependent bark photosynthesis of aspen twigs.Trees 15, 431-437.
DOI URL |
| [19] |
ávila E, Herrera A, Tezara W (2014). Contribution of stem CO2 fixation to whole-plant carbon balance in nonsucculent species.Photosynthetica 52, 3-15.
DOI URL |
| [20] |
Berveiller D, Damesin C (2008). Carbon assimilation by tree stems: potential involvement of phosphoenolpyruvate car- boxylase.Trees 22, 149-157.
DOI URL |
| [21] |
Berveiller D, Kierzkowski D, Damesin C (2007). Interspecific variability of stem photosynthesis among tree species.Tree Physiol 27, 53-61.
DOI URL PMID |
| [22] |
Bloemen J, Vergeynst LL, Overlaet-Michiels L, Steppe K (2016). How important is woody tissue photosynthesis in poplar during drought stress? Trees 30, 63-72.
DOI URL |
| [23] |
Bradford MM (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding.Anal Biochem 72, 248-254.
DOI URL PMID |
| [24] |
Burnell JN (1986). Purification and properties of phosphoenolpyruvate carboxykinase from C4 plants.Aust J Plant Physiol 13, 577-587.
DOI URL |
| [25] |
Burnell JN, Chastain CJ (2006). Cloning and expression of maize-leaf pyruvate, Pi dikinase regulatory protein gene.Biochem Biophys Res Commun 345, 675-680.
DOI URL PMID |
| [26] |
Carnal NW, Agostino A, Hatch MD (1993). Photosynthesis in phosphoenolpyruvate carboxykinase-type C4 plants: mechanism and regulation of C4 acid decarboxylation in bundle sheath cells.Arch Biochem Biophys 306, 360-367.
DOI URL |
| [27] |
Cen YP, Sage RF (2005). The regulation of rubisco activity in response to variation in temperature and atmospheric CO2 partial pressure in sweet potato.Plant Physiol 139, 979-990.
DOI URL PMID |
| [28] |
Chao Q, Liu XY, Mei YC, Gao ZF, Chen YB, Qian CR, Hao YB, Wan BC (2014). Light-regulated phosphorylation of maize phosphoenolpyruvate carboxykinase plays a vital role in its activity.Plant Mol Biol 85, 95-105.
DOI URL PMID |
| [29] |
Chastain CJ, Failing CJ, Manandhar L, Zimmerman MA, Lakner MM, Nguyen THT (2011). Functional evolution of C4 pyruvate, orthophosphate dikinase.J Exp Bot 62, 3083-3091.
DOI URL PMID |
| [30] |
Chen YB, Lu TC, Wang HX, Shen J, Bu TT, Chao Q, Gao ZF, Zhu XG, Wang YF, Wang BC (2014). Posttranslational modification of maize chloroplast pyruvate orthophosphate dikinase reveals the precise regulatory mechanism of its enzymatic activity.Plant Physiol 165, 534-549.
DOI URL PMID |
| [31] |
Cui K, He CY, Zhang JG, Duan AG, Zeng YF (2012). Temporal and spatial profiling of internode elongation-associated protein expression in rapidly growing culms of bamboo.J Proteome Res 11, 2492-2507.
DOI URL PMID |
| [32] |
Damesin C (2003). Respiration and photosynthesis characteristics of current-year stems of Fagus sylvatica: from the seasonal pattern to an annual balance. New Phytol 158, 465-475.
DOI URL |
| [33] |
Dima E, Manetas Y, Psaras GK (2006). Chlorophyll distribution pattern in inner stem tissues: evidence from epifluorescence microscopy and reflectance measurements in 20 woody species.Trees 20, 515-521.
DOI URL |
| [34] |
Hatch MD, Slack CR (1975). Pyruvate, Pi dikinase from leaves.Methods Enzymol 42, 212-219.
DOI URL |
| [35] |
Hibberd JM, Quick WP (2002). Characteristics of C4 photosynthesis in stems and petioles of C3 flowering plants.Nature 415, 451-454.
DOI URL |
| [36] | Hrstka M, Urban O, Babák L (2012). Seasonal changes of rubisco content and activity in Fagus sylvatica and Picea abies affected by elevated CO2 concentration. Chem Pap 66, 836-841. |
| [37] |
Johnson HS, Hatch MD (1970). Properties and regulation of leaf nicotinamide-adenine dinucleotide phosphate-malate dehydrogenase and ‘malic’ enzyme in plants with the C4-dicarboxylic acid pathway of photosynthesis.Biochem J 119, 273-280.
DOI URL |
| [38] |
Kocurek M, Pilarski J (2011). Activity of C4 enzymes in C3-type herbaceous plants.Photosynthetica 49, 473-477.
DOI URL |
| [39] | Kosvancová M, Urban O, Šprtová M, Hrstka M, Kalina J, Tomáskova I, Špunda V, Marek MV (2009). Photosynthetic induction in broadleaved Fagus sylvatica and conife- rous Picea abies cultivated under ambient and elevated CO2 concentrations. Plant Sci 177, 123-130. |
| [40] |
Leegood RC (2013). Strategies for engineering C4 photosynthesis.J Plant Physiol 170, 378-388.
DOI URL PMID |
| [41] | Leegood RC, Osmond CB (1990). The flux of metabolites in C4 and CAM plants. In: Dennis DT, Turpin DH, eds. Plant Physiology, Biochemistry and Molecular Biology. Essex: Longman Scientific & Technical. pp. 274-298. |
| [42] |
Lichtenthaler HK (1987). Chlorophylls and carotenoids: pigments of photosynthetic biomembranes.Methods Enzymol 148, 350-382.
DOI URL |
| [43] |
Muhaidat R, Sage RF, Dengler NG (2007). Diversity of Kranz anatomy and biochemistry in C4 eudicots.Am J Bot 94, 362-381.
DOI URL PMID |
| [44] |
Ocampo G, Columbus JT (2010). Molecular phylogenetics of suborder cactineae (caryophyllales), including insights into photosynthetic diversification and historical biogeography.Am J Bot 97, 1827-1847.
DOI URL PMID |
| [45] |
Parry MAJ, Andralojc PJ, Scales JC, Salvucci ME, Carmo-Silva AE, Alonso H, Whitney SM (2013). Rubisco activity and regulation as targets for crop improvement.J Exp Bot 64, 717-730.
DOI URL PMID |
| [46] | Peng ZH, Lu Y, Li LB, Zhao Q, Feng Q, Gao ZM, Lu HY, Hu T, Yao N, Liu KY, Li Y, Fan DL, Guo YL, Li WJ, Lu YQ, Weng QJ, Zhou CC, Zhang L, Huang T, Zhao Y, Zhu CR, Liu XE, Yang XW, Wang T, Miao K, Zhuang CY, Cao XL, Tang WL, Liu GS, Liu YL, Chen J, Liu ZJ, Yuan LC, Liu ZH, Huang XH, Lu TT, Fei BH, Ning ZM, Han B, Jiang ZH (2013). The draft genome of the fast-growing non-timber forest species moso bamboo (Phyllostachys heterocycla). Nat Genet 45, 456-461. |
| [47] |
Pfanz H (2008). Bark photosynthesis.Trees 22, 137-138.
DOI URL |
| [48] |
Portis Jr AR (2003). Rubisco activase-Rubisco’s catalytic chaperone.Photosynth Res 75, 11-27.
DOI URL |
| [49] |
Rangan P, Furtado A, Henry RJ (2016). New evidence for grain specific C4 photosynthesis in wheat.Sci Rep 6, 31721.
DOI URL |
| [50] |
Sage RF, Sage TL, Kocacinar F (2012). Photorespiration and the evolution of C4 photosynthesis.Annu Rev Plant Biol 63, 19-47.
DOI URL |
| [51] |
Sage RF, Sage TL, Pearcy RW, Borsch T (2007). The taxonomic distribution of C4 photosynthesis in Amaranthaceae sensu stricto.Am J Bot 94, 1992-2003.
DOI URL |
| [52] |
Saveyn A, Steppe K, Ubierna N, Dawson TE (2010). Woody tissue photosynthesis and its contribution to trunk growth and bud development in young plants.Plant Cell Environ 33, 1949-1958.
DOI URL PMID |
| [53] | Sheth BP, Thaker VS (2014). In silico analyses of Rubisco enzymes from different classes of Algae. Int Res J Biol Sci 3, 11-17. |
| [54] | Sun JL, Sui XL, Wang SH, Wei YX, Huang HY, Hu LP, Zhang ZX (2014). The response of rbcL, rbcS and rca genes in cucumber(Cucumis sativus L.) to growth and induction light intensity. Acta Physiol Plant 36, 2779-2791. |
| [55] |
Suzuki Y, Makino A (2012). Availability of Rubisco small subunit up-regulates the transcript levels of large subunit for stoichiometric assembly of its holoenzyme in rice.Plant Physiol 160, 533-540.
DOI URL PMID |
| [56] |
Teskey RO, Saveyn A, Steppe K, McGuire MA (2008). Origin, fate and significance of CO2 in tree stems.New Phytol 177, 17-32.
DOI URL |
| [57] |
Tikhonov KG, Khristin MS, Klimov VV, Sundireva MA, Kreslavski VD, Sidorov RA, Tsidendambayev VD, Savchenko TV (2017). Structural and functional characteristics of photosynthetic apparatus of chlorophyllcon- taining grape vine tissue.Russian J Plant Physiol 64, 73-82.
DOI URL |
| [58] |
Wang QM, Hou FY, Dong SX, Xie BT, Li AX, Zhang HY, Zhang LM (2014). Effects of shading on the photosynthetic capacity, endogenous hormones and root yield in purple-fleshed sweetpotato ( Ipomoea batatas (L.) Lam). Plant Growth Regul 72, 113-122.
DOI URL |
| [59] |
Wittmann C, Pfanz H (2014). Bark and woody tissue photosynthesis: a means to avoid hypoxia or anoxia in deve- loping stem tissues.Funct Plant Biol 41, 940-953.
DOI URL |
| [1] | 王贝贝, 吴苏, 王苗苗, 胡锦涛. 日光诱导叶绿素荧光不同组分在作物总初级生产力估算中的贡献比例: 多时间尺度分析[J]. 植物生态学报, 2025, 49(4): 562-572. |
| [2] | 闫小莉, 刘贵梅, 李小玉, 江宇翔, 全小强, 王燕茹, 曲鲁平, 汤行昊. 不同氮添加水平和铵硝态氮配比环境下木荷幼苗光合及叶绿素荧光特性[J]. 植物生态学报, 2025, 49(4): 624-637. |
| [3] | 刘柯言, 韩璐, 宋午椰, 张初蕊, 胡旭, 许行, 陈立欣. 基于日光诱导叶绿素荧光探测干旱对黄土高原植被光合稳定性的影响[J]. 植物生态学报, 2025, 49(3): 415-431. |
| [4] | 全小强, 王燕茹, 李小玉, 梁海燕, 王立冬, 闫小莉. 氮添加和铵硝态氮配比对杉木幼苗光合特性及叶绿素荧光参数的影响[J]. 植物生态学报, 2024, 48(8): 1050-1064. |
| [5] | 黄佳慧, 杨惠敏, 陈欣雨, 朱超宇, 江亚楠, 胡程翔, 连锦瑾, 芦涛, 路梅, 张维林, 饶玉春. 水稻突变体pe-1对弱光胁迫的响应机制[J]. 植物学报, 2024, 59(4): 574-584. |
| [6] | 毛莹儿, 周秀梅, 王楠, 李秀秀, 尤育克, 白尚斌. 毛竹扩张对杉木林土壤细菌群落的影响[J]. 生物多样性, 2023, 31(6): 22659-. |
| [7] | 师生波, 周党卫, 李天才, 德科加, 杲秀珍, 马家麟, 孙涛, 王方琳. 青藏高原高山嵩草光合功能对模拟夜间低温的响应[J]. 植物生态学报, 2023, 47(3): 361-373. |
| [8] | 任培鑫, 李鹏, 彭长辉, 周晓路, 杨铭霞. 洞庭湖流域植被光合物候的时空变化及其对气候变化的响应[J]. 植物生态学报, 2023, 47(3): 319-330. |
| [9] | 金佳怡, 罗怿婷, 杨惠敏, 芦涛, 叶涵斐, 谢继毅, 王珂欣, 陈芊羽, 方媛, 王跃星, 饶玉春. 水稻叶绿素含量QTL定位与候选基因表达分析[J]. 植物学报, 2023, 58(3): 394-403. |
| [10] | 余玉蓉, 吴浩, 高娅菲, 赵媛博, 李小玲, 卜贵军, 薛丹, 刘正祥, 武海雯, 吴林. 模拟氮沉降对鄂西南湿地泥炭藓生理及形态特征的影响[J]. 植物生态学报, 2023, 47(11): 1493-1506. |
| [11] | 师生波, 师瑞, 周党卫, 张雯. 低温对高山嵩草叶片光化学和非光化学能量耗散特征的影响[J]. 植物生态学报, 2023, 47(10): 1441-1452. |
| [12] | 郑宁, 李素英, 王鑫厅, 吕世海, 赵鹏程, 臧琛, 许玉珑, 何静, 秦文昊, 高恒睿. 基于环境因子对叶绿素影响的典型草原植物生活型优势研究[J]. 植物生态学报, 2022, 46(8): 951-960. |
| [13] | 王浩, 王明, 梁婷, 姚玉新, 杜远鹏, 高振. 气温和根区温度对葡萄叶片光合荧光特性的影响[J]. 植物学报, 2022, 57(2): 209-216. |
| [14] | 薛金儒, 吕肖良. 黄土高原生态工程实施下基于日光诱导叶绿素荧光的植被恢复生产力效益评价[J]. 植物生态学报, 2022, 46(10): 1289-1304. |
| [15] | 吴霖升, 张永光, 章钊颖, 张小康, 吴云飞. 日光诱导叶绿素荧光遥感及其在陆地生态系统监测中的应用[J]. 植物生态学报, 2022, 46(10): 1167-1199. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||