

植物学报 ›› 2025, Vol. 60 ›› Issue (2): 235-245.DOI: 10.11983/CBB24061 cstr: 32102.14.CBB24061
曹雪敏1,3,†, 包颖2,†, 张悦新1,3, 李瑞杰1,3, 苏健馨1,3, 张蔚1,3,*( )
)
收稿日期:2024-04-24
接受日期:2024-08-20
出版日期:2025-03-10
发布日期:2024-08-22
通讯作者:
张蔚
作者简介:第一联系人:†共同第一作者
基金资助:
Xuemin Cao1,3,†, Ying Bao2,†, Yuexin Zhang1,3, Ruijie Li1,3, Jianxin Su1,3, Wei Zhang1,3,*( )
)
Received:2024-04-24
Accepted:2024-08-20
Online:2025-03-10
Published:2024-08-22
Contact:
Wei Zhang
About author:First author contact:†These authors contributed equally to this paper
摘要: 以野蔷薇(Rosa multiflora)当年生带芽茎段为试材, 建立了其组培快繁体系。结果表明, 最佳外植体是带腋芽茎段, 外植体最佳消毒方法是先用75%乙醇浸泡30秒, 再用10%次氯酸钠溶液浸泡20分钟, 成活率可达96%; 带芽茎段萌芽最佳诱导培养基为MS+1.0 mg∙L-1 6-BA+0.01 mg∙L-1 NAA+0.1 mg∙L-1 GA3, 培养30天, 萌芽率可达98%; 无菌再生苗增殖最佳基础培养基为WPM, 增殖系数为2.87; 无菌再生苗生根最佳培养基为1/2MS+1.0 mg∙L-1 6-BA+0.1 mg∙L-1 NAA, 生根率可达93%; 无菌再生苗移栽成活率达98%。在此基础上, 以野蔷薇无菌再生苗为受体, 建立了野蔷薇瞬时表达体系。结果表明, 瞬时表达最佳转化条件是菌液OD600为0.8, 负压为-0.10 MPa, 真空抽吸2次, 每次15分钟, 瞬时表达效率可达96%。研究结果为建立野蔷薇再生及遗传转化体系奠定了基础, 并为蔷薇属植物基因功能研究提供技术支持。
曹雪敏, 包颖, 张悦新, 李瑞杰, 苏健馨, 张蔚. 野蔷薇组培快繁和高效瞬时表达体系的建立. 植物学报, 2025, 60(2): 235-245.
Xuemin Cao, Ying Bao, Yuexin Zhang, Ruijie Li, Jianxin Su, Wei Zhang. Tissue Culture, Rapid Propagation and Efficient Transient Expression Systems of Rosa multiflora. Chinese Bulletin of Botany, 2025, 60(2): 235-245.
| Reagents | Required solvent dosage |
|---|---|
| 0.2 mol∙L-1 NaH2PO4 | 38 mL |
| 0.2 mol∙L-1 Na2HPO4 | 62 mL |
| 100 mmol∙L-1 K3Fe(CN)6 | 1 mL |
| 100 mmol∙L-1 K4Fe(CN)6 | 1 mL |
| 0.5 mol∙L-1 NaEDTA | 4 mL |
| 0.1% Triton X-100 | 200 μL |
| 20% methanol | 20 mL |
| X-Gluc | 200 mg |
表1 GUS染色液配方
Table 1 GUS staining solution formulation
| Reagents | Required solvent dosage |
|---|---|
| 0.2 mol∙L-1 NaH2PO4 | 38 mL |
| 0.2 mol∙L-1 Na2HPO4 | 62 mL |
| 100 mmol∙L-1 K3Fe(CN)6 | 1 mL |
| 100 mmol∙L-1 K4Fe(CN)6 | 1 mL |
| 0.5 mol∙L-1 NaEDTA | 4 mL |
| 0.1% Triton X-100 | 200 μL |
| 20% methanol | 20 mL |
| X-Gluc | 200 mg |
| Treatments | 75% ethanol immersion time (s) | NaClO (%) | NaClO immersion time (min) |
|---|---|---|---|
| A | 30 | 5 | 10 |
| B | 30 | 5 | 15 |
| C | 30 | 5 | 20 |
| D | 30 | 10 | 10 |
| E | 30 | 10 | 15 |
| F | 30 | 10 | 20 |
| G | 30 | 15 | 10 |
| H | 30 | 15 | 15 |
| I | 30 | 15 | 20 |
表2 外植体消毒方案
Table 2 Explant disinfection scheme
| Treatments | 75% ethanol immersion time (s) | NaClO (%) | NaClO immersion time (min) |
|---|---|---|---|
| A | 30 | 5 | 10 |
| B | 30 | 5 | 15 |
| C | 30 | 5 | 20 |
| D | 30 | 10 | 10 |
| E | 30 | 10 | 15 |
| F | 30 | 10 | 20 |
| G | 30 | 15 | 10 |
| H | 30 | 15 | 15 |
| I | 30 | 15 | 20 |
| Treatments | Basic medium | 6-BA (mg∙L-1) | NAA (mg∙L-1) | GA3 (mg∙L-1) |
|---|---|---|---|---|
| A1 | MS | 0.5 | 0.01 | 0 |
| A2 | MS | 1.0 | 0.01 | 0 |
| A3 | MS | 0.5 | 0.1 | 0 |
| A4 | MS | 1.0 | 0.1 | 0 |
| A5 | MS | 0.5 | 0.01 | 0.1 |
| A6 | MS | 1.0 | 0.01 | 0.1 |
| A7 | MS | 0.5 | 0.1 | 0.1 |
| A8 | MS | 1.0 | 0.1 | 0.1 |
表3 野蔷薇启动培养的处理方案
Table 3 Treatment schemes for the initiation culture of Rosa multiflora
| Treatments | Basic medium | 6-BA (mg∙L-1) | NAA (mg∙L-1) | GA3 (mg∙L-1) |
|---|---|---|---|---|
| A1 | MS | 0.5 | 0.01 | 0 |
| A2 | MS | 1.0 | 0.01 | 0 |
| A3 | MS | 0.5 | 0.1 | 0 |
| A4 | MS | 1.0 | 0.1 | 0 |
| A5 | MS | 0.5 | 0.01 | 0.1 |
| A6 | MS | 1.0 | 0.01 | 0.1 |
| A7 | MS | 0.5 | 0.1 | 0.1 |
| A8 | MS | 1.0 | 0.1 | 0.1 |
| Treatments | Basic medium | Sucrose (%) | Agar (%) |
|---|---|---|---|
| B1 | MS | 3 | 0.75 |
| B2 | 1/2MS | 3 | 0.75 |
| B3 | QL | 3 | 0.75 |
| B4 | 1/2QL | 3 | 0.75 |
| B5 | WPM | 3 | 0.75 |
| B6 | B5 | 3 | 0.75 |
表4 野蔷薇增殖培养处理方案
Table 4 Treatment schemes for the propagation culture of Rosa multiflora
| Treatments | Basic medium | Sucrose (%) | Agar (%) |
|---|---|---|---|
| B1 | MS | 3 | 0.75 |
| B2 | 1/2MS | 3 | 0.75 |
| B3 | QL | 3 | 0.75 |
| B4 | 1/2QL | 3 | 0.75 |
| B5 | WPM | 3 | 0.75 |
| B6 | B5 | 3 | 0.75 |
| Treatments | Basic medium | NAA (mg∙L-1) | IBA (mg∙L-1) |
|---|---|---|---|
| C1 | MS | 0.05 | 0 |
| C2 | MS | 0.1 | 0 |
| C3 | 1/2MS | 0.05 | 0 |
| C4 | 1/2MS | 0.1 | 0 |
| C5 | MS | 0 | 0.5 |
| C6 | MS | 0 | 1.0 |
| C7 | 1/2MS | 0 | 0.5 |
| C8 | 1/2MS | 0 | 1.0 |
表5 野蔷薇生根培养处理方案
Table 5 Treatment schemes for the rooting culture of Rosa multiflora
| Treatments | Basic medium | NAA (mg∙L-1) | IBA (mg∙L-1) |
|---|---|---|---|
| C1 | MS | 0.05 | 0 |
| C2 | MS | 0.1 | 0 |
| C3 | 1/2MS | 0.05 | 0 |
| C4 | 1/2MS | 0.1 | 0 |
| C5 | MS | 0 | 0.5 |
| C6 | MS | 0 | 1.0 |
| C7 | 1/2MS | 0 | 0.5 |
| C8 | 1/2MS | 0 | 1.0 |
| Treatments | Bacterium OD600 | Vacuum pressure (MPa) | Vacuum processing time (min) |
|---|---|---|---|
| D1 | 0.6 | -0.06 | 5 |
| D2 | 0.8 | -0.08 | 5 |
| D3 | 1.0 | -0.10 | 5 |
| D4 | 1.0 | -0.08 | 10 |
| D5 | 0.8 | -0.06 | 10 |
| D6 | 0.6 | -0.10 | 10 |
| D7 | 0.6 | -0.08 | 15 |
| D8 | 0.8 | -0.10 | 15 |
| D9 | 1.0 | -0.06 | 15 |
表6 野蔷薇无菌再生苗瞬时表达实验设计
Table 6 Experimental design for transient expression of sterile re-generated plantlets of Rosa multiflora
| Treatments | Bacterium OD600 | Vacuum pressure (MPa) | Vacuum processing time (min) |
|---|---|---|---|
| D1 | 0.6 | -0.06 | 5 |
| D2 | 0.8 | -0.08 | 5 |
| D3 | 1.0 | -0.10 | 5 |
| D4 | 1.0 | -0.08 | 10 |
| D5 | 0.8 | -0.06 | 10 |
| D6 | 0.6 | -0.10 | 10 |
| D7 | 0.6 | -0.08 | 15 |
| D8 | 0.8 | -0.10 | 15 |
| D9 | 1.0 | -0.06 | 15 |
| Treatments | Contamination rate (%) | Browning rate (%) | Survival rate (%) |
|---|---|---|---|
| A | 11.11±0.00 a | 59.26±6.42 b | 29.60±6.42 a |
| B | 0.00±0.00 b | 92.59±6.42 a | 7.41±6.42 b |
| C | 7.41±6.42 ab | 100.00±0.00 a | 0.00±0.00 b |
| D | 0.00±0.00 b | 92.59±6.42 a | 7.41±6.42 b |
| E | 3.70±6.42 b | 100.00±0.00 a | 0.00±0.00 b |
| F | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
| G | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
| H | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
| I | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
表7 不同消毒方法对野蔷薇带顶芽茎段消毒效果的影响
Table 7 Effects of different disinfection methods on the stem segments with apical buds of Rosa multiflora
| Treatments | Contamination rate (%) | Browning rate (%) | Survival rate (%) |
|---|---|---|---|
| A | 11.11±0.00 a | 59.26±6.42 b | 29.60±6.42 a |
| B | 0.00±0.00 b | 92.59±6.42 a | 7.41±6.42 b |
| C | 7.41±6.42 ab | 100.00±0.00 a | 0.00±0.00 b |
| D | 0.00±0.00 b | 92.59±6.42 a | 7.41±6.42 b |
| E | 3.70±6.42 b | 100.00±0.00 a | 0.00±0.00 b |
| F | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
| G | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
| H | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
| I | 0.00±0.00 b | 100.00±0.00 a | 0.00±0.00 b |
| Treatments | Contamination rate (%) | Browning rate (%) | Survival rate (%) |
|---|---|---|---|
| A | 3.70±6.42 a | 18.52±6.42 ab | 77.78±11.11 a |
| B | 7.41±6.42 a | 25.93±6.42 a | 66.67±0.00 a |
| C | 3.70±6.42 a | 14.81±6.42 ab | 81.48±6.42 a |
| D | 7.41±6.42 a | 11.11±0.00 b | 81.48±6.42 a |
| E | 14.81±16.97 a | 14.81±6.42 ab | 70.37±16.97 a |
| F | 3.70±6.42 a | 0.00±0.00 c | 96.30±6.42 a |
| G | 11.11±11.11 a | 14.81±6.42 ab | 74.07±16.97 a |
| H | 0.00±0.00 a | 25.93±6.42 a | 74.07±6.42 a |
| I | 18.52±12.83 a | 11.11±0.00 b | 70.37±12.83 a |
表8 不同消毒方法对野蔷薇带腋芽茎段消毒效果的影响
Table 8 Effects of different disinfection methods on the stem segments with axillary buds of Rosa multiflora
| Treatments | Contamination rate (%) | Browning rate (%) | Survival rate (%) |
|---|---|---|---|
| A | 3.70±6.42 a | 18.52±6.42 ab | 77.78±11.11 a |
| B | 7.41±6.42 a | 25.93±6.42 a | 66.67±0.00 a |
| C | 3.70±6.42 a | 14.81±6.42 ab | 81.48±6.42 a |
| D | 7.41±6.42 a | 11.11±0.00 b | 81.48±6.42 a |
| E | 14.81±16.97 a | 14.81±6.42 ab | 70.37±16.97 a |
| F | 3.70±6.42 a | 0.00±0.00 c | 96.30±6.42 a |
| G | 11.11±11.11 a | 14.81±6.42 ab | 74.07±16.97 a |
| H | 0.00±0.00 a | 25.93±6.42 a | 74.07±6.42 a |
| I | 18.52±12.83 a | 11.11±0.00 b | 70.37±12.83 a |

图1 野蔷薇在不同启动培养基上的生长状态 A1-A8同表3。每处理接种10瓶, 每瓶接种3个外植体, 每处理重复3次。Bars=1.5 cm
Figure 1 Growth condition of Rosa multiflora on different initiation media A1-A8 are the same as shown in Table 3. Each treatment is inoculated with 10 vials, each vial is inoculated with 3 explants, and each treatment is repeated 3 times. Bars=1.5 cm
| Treatments | Germination rate (%) | Plant height (cm) | Number of compound leaves (slice) |
|---|---|---|---|
| A1 | 92.22±7.70 a | 1.30±0.50 bc | 4.37±1.39 cd |
| A2 | 97.78±1.92 a | 1.52±0.59 a | 4.94±1.72 abc |
| A3 | 96.67±3.34 a | 1.20±0.59 bc | 4.45±1.46 cd |
| A4 | 98.89±1.92 a | 1.25±0.50 bc | 5.17±1.41 a |
| A5 | 98.89±1.92 a | 1.11±0.39 c | 4.74±1.64 abcd |
| A6 | 98.89±1.92 a | 1.35±0.45 b | 5.07±1.61 ab |
| A7 | 97.78±1.92 a | 1.15±0.48 bc | 4.17±1.71 d |
| A8 | 96.67±5.77 a | 1.25±0.41 bc | 4.69±1.77 abcd |
表9 不同植物生长调节剂类型及浓度对野蔷薇萌发效果的影响
Table 9 Effects of different plant growth regulator types and concentrations on the germination of Rosa multiflora
| Treatments | Germination rate (%) | Plant height (cm) | Number of compound leaves (slice) |
|---|---|---|---|
| A1 | 92.22±7.70 a | 1.30±0.50 bc | 4.37±1.39 cd |
| A2 | 97.78±1.92 a | 1.52±0.59 a | 4.94±1.72 abc |
| A3 | 96.67±3.34 a | 1.20±0.59 bc | 4.45±1.46 cd |
| A4 | 98.89±1.92 a | 1.25±0.50 bc | 5.17±1.41 a |
| A5 | 98.89±1.92 a | 1.11±0.39 c | 4.74±1.64 abcd |
| A6 | 98.89±1.92 a | 1.35±0.45 b | 5.07±1.61 ab |
| A7 | 97.78±1.92 a | 1.15±0.48 bc | 4.17±1.71 d |
| A8 | 96.67±5.77 a | 1.25±0.41 bc | 4.69±1.77 abcd |
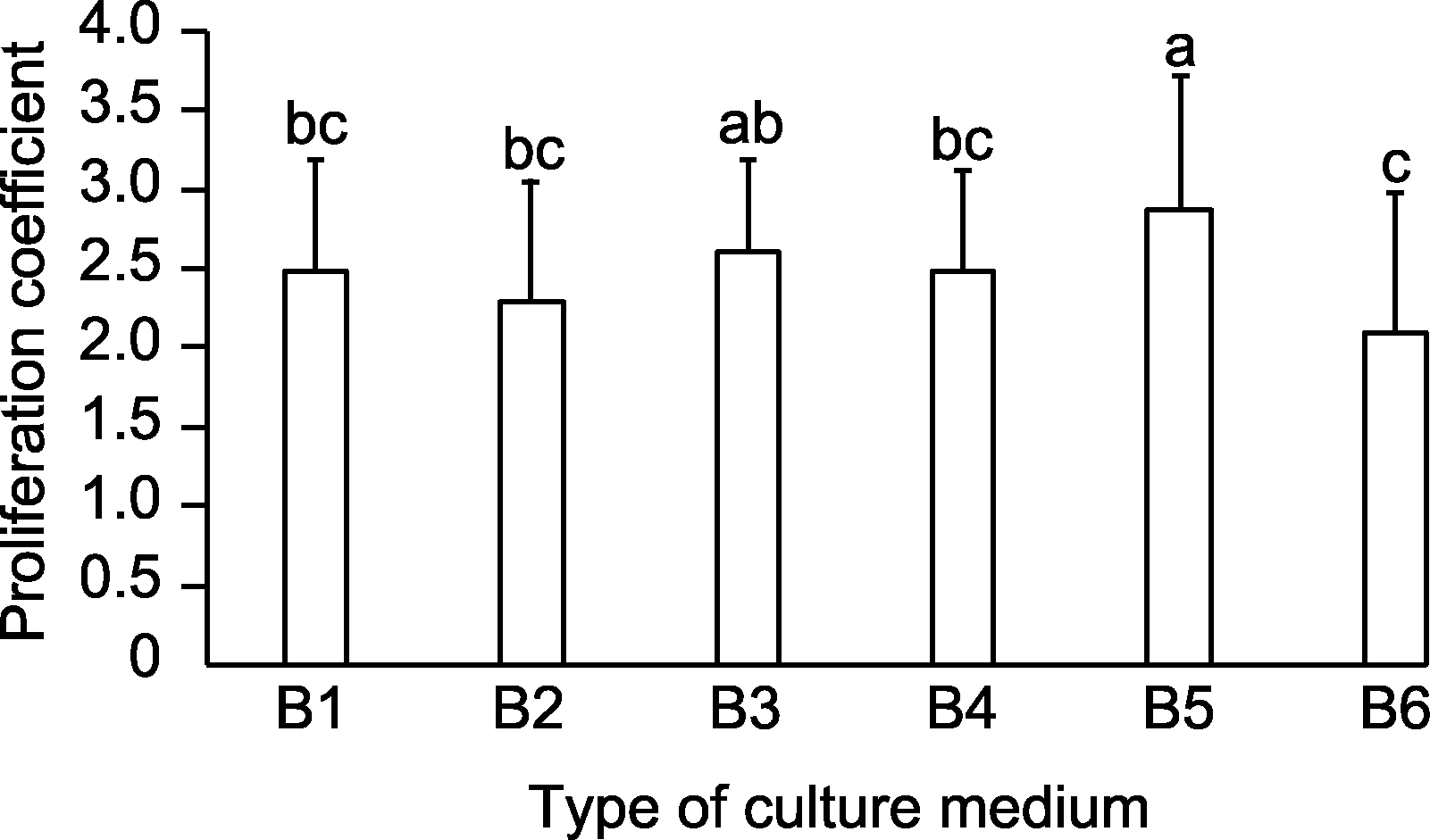
图2 基础培养基类型对野蔷薇增殖系数的影响 B1-B6同表4。不同小写字母表示差异显著(P<0.05)。
Figure 2 Effects of basal medium types on the proliferation coefficient of Rosa multiflora B1-B6 are the same as shown in Table 4. Different lowercase letters indicate significant differences (P<0.05).

图3 野蔷薇在不同增殖培养基上的生长状态 B1-B6同表4。每处理接种5瓶, 每瓶接种3个外植体, 每处理重复3次。Bars=1 cm
Figure 3 Growth condition of Rosa multiflora on different proliferation media B1-B6 are the same as shown in Table 4. Each treatment is inoculated with 5 vials, each vial is inoculated with 3 explants, and each treatment is repeated 3 times. Bars=1 cm

图4 野蔷薇在不同生根培养基上的根部状态 C1-C8同表5。每处理接种5瓶, 每瓶接种3个外植体, 每处理重复3次。Bars=1 cm
Figure 4 Root condition of Rosa multiflora on different rooting media C1-C8 are the same as shown in Table 5. Each treatment is inoculated with 5 vials, each vial is inoculated with 3 explants, and each treatment is repeated 3 times. Bars=1 cm
| Treatments | Rooting rate (%) | Average number of roots (root) | Root status |
|---|---|---|---|
| C1 | 88.33±2.89 ab | 8.22 | Short roots, weak growth |
| C2 | 88.33±2.89 ab | 9.30 | Strong roots, grow well |
| C3 | 93.33±7.64 ab | 9.78 | Short roots, weak growth |
| C4 | 93.33±2.89 a | 8.85 | Strong roots, grow well |
| C5 | 81.67±5.77 ab | 8.25 | Short roots, weak growth |
| C6 | 81.67±2.89 ab | 8.47 | Strong roots, grow well |
| C7 | 78.33±5.77 b | 6.68 | Short roots, weak growth |
| C8 | 86.67±5.77 ab | 7.18 | Short roots, weak growth |
表10 不同培养基配方对野蔷薇生根的影响
Table 10 Effects of different media formulations on the rooting of Rosa multiflora
| Treatments | Rooting rate (%) | Average number of roots (root) | Root status |
|---|---|---|---|
| C1 | 88.33±2.89 ab | 8.22 | Short roots, weak growth |
| C2 | 88.33±2.89 ab | 9.30 | Strong roots, grow well |
| C3 | 93.33±7.64 ab | 9.78 | Short roots, weak growth |
| C4 | 93.33±2.89 a | 8.85 | Strong roots, grow well |
| C5 | 81.67±5.77 ab | 8.25 | Short roots, weak growth |
| C6 | 81.67±2.89 ab | 8.47 | Strong roots, grow well |
| C7 | 78.33±5.77 b | 6.68 | Short roots, weak growth |
| C8 | 86.67±5.77 ab | 7.18 | Short roots, weak growth |

图5 野蔷薇生根苗炼苗移栽过程 (A) 生根的无菌再生苗; (B) 移栽当天的植株; (C) 移栽30天后的植株。每个重复移栽40株生根苗, 重复3次。Bars=1 cm
Figure 5 The transplanting process of rooted plantlets of Rosa multiflora (A) Rooted sterile regenerated plantlets; (B) Plantlets on the first day of transplanting; (C) Plantlets transplanted 30 days later. Transplant 40 rooted plantlets per replicate and repeat 3 times. Bars=1 cm
| Treatments | Transient expression efficiency (%) | Staining effect |
|---|---|---|
| D1 | 14.07±12.24 d | Pale blue, flake area blue |
| D2 | 30.63±2.44 cd | Pale blue, flake area blue |
| D3 | 65.27±17.71 b | Blue, flake area blue |
| D4 | 52.38±10.82 bc | Pale blue, leaf blue |
| D5 | 15.87±5.72 d | Blue, flake area blue |
| D6 | 52.89±17.16 bc | Blue, leaf blue |
| D7 | 4.76±8.25 d | Blue, leaf blue |
| D8 | 96.30±6.42 a | Blue, leaf blue |
| D9 | 31.19±7.84 cd | Blue, flake area blue |
表11 不同处理对野蔷薇瞬时表达效率的影响
Table 11 Effects of different treatments on the transient expression efficiency of Rosa multiflora
| Treatments | Transient expression efficiency (%) | Staining effect |
|---|---|---|
| D1 | 14.07±12.24 d | Pale blue, flake area blue |
| D2 | 30.63±2.44 cd | Pale blue, flake area blue |
| D3 | 65.27±17.71 b | Blue, flake area blue |
| D4 | 52.38±10.82 bc | Pale blue, leaf blue |
| D5 | 15.87±5.72 d | Blue, flake area blue |
| D6 | 52.89±17.16 bc | Blue, leaf blue |
| D7 | 4.76±8.25 d | Blue, leaf blue |
| D8 | 96.30±6.42 a | Blue, leaf blue |
| D9 | 31.19±7.84 cd | Blue, flake area blue |

图6 不同处理下野蔷薇无菌再生苗GUS染色效果 D1-D9同表6。每处理接种3瓶, 每瓶接种3个外植体, 每处理重复3次。Bars=1 cm
Figure 6 Effects of different treatments on GUS staining of sterile regenerated plantlets of Rosa multiflora D1-D9 are the same as shown in Table 6. Each treatment is inoculated with 3 vials, each vial is inoculated with 3 explants, and each treatment is repeated 3 times. Bars=1 cm
| [1] | 丁萌 (2012). 野蔷薇再生体系的建立及其遗传转化的研究. 硕士论文. 武汉: 华中农业大学. pp. 14-38. |
| [2] | 龚维红 (2022). 大马士革玫瑰组培与快繁技术研究. 安徽农学通报 28, 19-21. |
| [3] | 李刚, 宋平丽, 王翔, 马青翠, 张海霞, 张玉星, 许建锋, 亓宝秀 (2021). 农杆菌介导的杜梨叶片瞬时转化方法的建立. 果树学报 38, 2006-2013. |
| [4] |
李惠玲, 罗玉兰, 章漳, 尹丽娟, 李圃锦, 张冬梅 (2020). 细梗蔷薇种子发芽及组织培养技术研究. 中国农学通报 36 (13), 89-93.
DOI |
| [5] |
李佳慧, 叶维雁, 朱鹏锦, 庞新华, 张继, 唐毓玮, 韦俏宇 (2022). 猫须草无菌短枝组织培养与快速繁殖体系的建立. 热带作物学报 43, 2063-2070.
DOI |
| [6] | 李静, 陈敏, 刘现伟, 沈法富, 王鹏 (2006). 莴苣高效瞬时表达体系的建立. 园艺学报 33, 405-407. |
| [7] | 李晓亮, 张军云, 张钟, 董春富, 杨世先, 王文智, 张建康, 张翠萍 (2017). 滇红食用玫瑰生根培养基的试验筛选研究. 西南农业学报 30, 656-663. |
| [8] | 李心悦, 张克闯, 张道远, 李进, 王玉成 (2018). 新疆野苹果瞬时遗传转化方法建立及初步验证. 分子植物育种 16, 7315-7321. |
| [9] | 李增武, 钟玲, 赵梁军 (2011). 野蔷薇种子休眠和萌发整齐度研究. 种子 30(10), 42-44. |
| [10] |
刘雪梅, 王蕾, 文添龙, 何鹏, 俞嘉宁 (2014). 农杆菌介导的棉花子叶瞬时表达系统的建立. 植物学报 49, 587-594.
DOI |
| [11] | 刘亚娟, 杨小艳, 谢树章, 吴红, 高立均 (2018). 芳纯月季组培快繁技术研究. 西南师范大学学报(自然科学版) 43(8),52-56. |
| [12] | 鲁艺, 史文君, 黄淑丹, 肖明 (2022). 苦水玫瑰组培快繁技术研究. 青海大学学报 40(3), 41-46. |
| [13] | 穆建鑫 (2021). ‘中梨一号’梨再生体系建立及秋子梨遗传转化体系探索. 硕士论文. 南京: 南京农业大学. pp. 13-24. |
| [14] | 彭奎莉, 孟繁博, 周志达, 郑关任, 张金柱, 杨涛, 车代弟 (2018). 不同基因型丰花月季快繁体系差异的研究. 见:张启翔. 中国观赏园艺研究进展2018. 北京: 中国林业出版社. pp. 480-486. |
| [15] | 强泽宇 (2015). 中华金叶榆繁殖技术研究. 硕士论文. 保定: 河北农业大学. pp. 6-12. |
| [16] | 任菲宏, 王姣, 李晓松, 石丽姝, 任红艳, 吴茂宏 (2023). 外植体取样时间和消毒方式对沙子空心李诱导培养的影响. 现代农业科技 (12), 77-79. |
| [17] | 宋常美, 文晓鹏 (2015). 4种贵州樱桃的高效离体再生. 西南大学学报(自然科学版) 37(9),19-24. |
| [18] | 王悦, 吴艳菊, 孟大伟, 郎晨婧, 赖薪宇, 刘佳, 葛禹凡, 杨丽萍 (2020). 植物瞬时表达体系真空侵染法的优化. 分子植物育种 18, 6743-6748. |
| [19] | 徐立军, 李志斌, 蒋淑磊, 李振勤 (2015). 大马士革玫瑰组织培养技术研究. 河北林业科技 (2), 19-21. |
| [20] | 闫海霞, 蒋月喜, 黄昌艳, 邓杰玲, 何荆洲, 王晓国, 卜朝阳 (2016). 月季‘卡罗拉’的组培快繁技术. 热带作物学报 37, 1741-1746. |
| [21] | 闫允青, 姜桦韬, 谷超, 吴俊 (2017). ‘雪花梨’扩繁和叶片再生体系的建立. 南京农业大学学报 40, 68-75. |
| [22] | 杨丽萍, 许亚男, 刘雨晴, 孟大伟, 金太成 (2016). 利用根部真空侵染法在烟草中瞬时表达外源蛋白. 分子植物育种 14, 3385-3389. |
| [23] |
张燕红, 黄乐平, 周小云, 王冬梅 (2008). 农杆菌真空渗透法转化棉花花粉的初步研究. 棉花学报 20, 354-358.
DOI |
| [24] | 赵月玲, 王汉海, 程贯召, 杜延飞 (2001). 枣组织培养中外植体取材部位的比较研究. 潍坊学院学报 1(2), 53-54, 43. |
| [25] | 周玉洁, 韦雪芬, 申长青, 李焜钊, 孙朝辉, 黄久香 (2019). 濒危植物四药门花的组培快繁. 植物生理学报 55, 635-641. |
| [26] |
Lu J, Bai MJ, Ren HR, Liu JY, Wang CQ (2017). An efficient transient expression system for gene function analysis in rose. Plant Methods 13, 116.
DOI PMID |
| [27] |
Nakamura N, Hirakawa H, Sato S, Otagaki S, Matsumoto S, Tabata S, Tanaka Y (2018). Genome structure of Rosa multiflora, a wild ancestor of cultivated roses. DNA Res 25, 113-121.
DOI PMID |
| [28] |
Zhang Y, Zhao MJ, Zhu W, Shi CM, Bao MZ, Zhang W (2021). Nonglandular prickle formation is associated with development and secondary metabolism-related genes in Rosa multiflora. Physiol Plant 173, 1147-1162.
DOI PMID |
| [1] | 张尚文, 黄诗宇, 杨天为, 李婷, 张向军, 高曼熔. 基于正交实验的赤苍藤组培快繁体系建立[J]. 植物学报, 2024, 59(1): 99-109. |
| [2] | 吕秀立, 于泽群, 陈香波, 傅仁杰, 缪珊珊, 杜安. 粉美人萱草的快繁技术和大田种植[J]. 植物学报, 2022, 57(3): 350-357. |
| [3] | 杨立文,刘双荣,李玉红,林荣呈. 植物转录因子与DNA互作研究技术[J]. 植物学报, 2020, 55(4): 468-474. |
| [4] | 赵华,邵广达,高文鑫,顾彪. 双管基因枪介导的基因瞬时表达技术在拟南芥中的应用[J]. 植物学报, 2020, 55(2): 182-191. |
| [5] | 赵燕,周俭民. 萤火素酶互补实验检测蛋白互作[J]. 植物学报, 2020, 55(1): 69-75. |
| [6] | 刘利静,赵庆臻,谢旗,于菲菲. 快速高效检测植物体内蛋白泛素化修饰研究方法[J]. 植物学报, 2019, 54(6): 753-763. |
| [7] | 宋爱华, 张文斌, 孙姝兰, 李凌飞, 王小菁. 非洲菊原生质体制备及瞬时转化系统的建立[J]. 植物学报, 2017, 52(4): 511-519. |
| [8] | 李潇, 孙海彦, 阮孟斌, 王霈虹, 彭明. 地衣芽孢杆菌耐高温α-淀粉酶基因的克隆、烟草瞬时表达及转化拟南芥的研究[J]. 植物学报, 2015, 50(3): 354-362. |
| [9] | 刘雪梅, 王蕾, 文添龙, 何鹏, 俞嘉宁. 农杆菌介导的棉花子叶瞬时表达系统的建立[J]. 植物学报, 2014, 49(5): 587-594. |
| [10] | 陈书安, 王晓东, 赵兵, 王玉春. 应用稀土调控藏红花胚性愈伤组织的生长与分化[J]. 植物学报, 2010, 45(05): 609-614. |
| [11] | 王道杰, 杨翠玲, 陆鸣. 真空渗透法转化油菜及转化种子的筛选[J]. 植物学报, 2009, 44(02): 216-222. |
| [12] | 谢伟 乐超银 郭政宏 戴志鹏 刘敏 姚伟. 不同调控序列作用下GUS基因在烟草中瞬时表达活性的研究[J]. 植物学报, 2007, 24(04): 452-458. |
| [13] | 汲逢源, 王戈亮, 许亦农. 抗氧化剂对农杆菌介导的大豆下胚轴GUS基因瞬时表达的影响[J]. 植物生态学报, 2006, 30(2): 330-334. |
| [14] | 冯英 薛庆中. 生姜脱菌快繁研究进展[J]. 植物学报, 2002, 19(04): 439-443. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||