

植物学报 ›› 2019, Vol. 54 ›› Issue (2): 168-184.DOI: 10.11983/CBB18219 cstr: 32102.14.CBB18219
所属专题: 逆境生物学专辑 (2019年54卷2期)
收稿日期:2018-10-20
接受日期:2018-12-29
出版日期:2019-03-01
发布日期:2019-09-01
通讯作者:
侯岁稳
基金资助:Received:2018-10-20
Accepted:2018-12-29
Online:2019-03-01
Published:2019-09-01
Contact:
Suiwen Hou
摘要: 蛋白磷酸化修饰是植物细胞信号调控的普遍机制。植物-病原微生物互作过程中, 关键调控蛋白的磷酸化状态影响免疫信号的激活。多种病原微生物通过干扰宿主蛋白的磷酸化状态攻击免疫系统, 以提高致病性。该文对植物免疫调控过程中关键元件的磷酸化修饰及其在免疫信号中的调控作用进行了综述。研究植物-病原菌互作过程中关键蛋白的磷酸化修饰, 有助于深入探讨植物-病原微生物互作的分子机理。该文将为寻找广谱抗病的新途径提供理论依据。
刘雅琼,侯岁稳. 蛋白磷酸化修饰在植物-病原微生物互作中的作用研究进展. 植物学报, 2019, 54(2): 168-184.
Yaqiong Liu,Suiwen Hou. Research Progress in Protein Phosphorylation in Plant-pathogen Interactions. Chinese Bulletin of Botany, 2019, 54(2): 168-184.
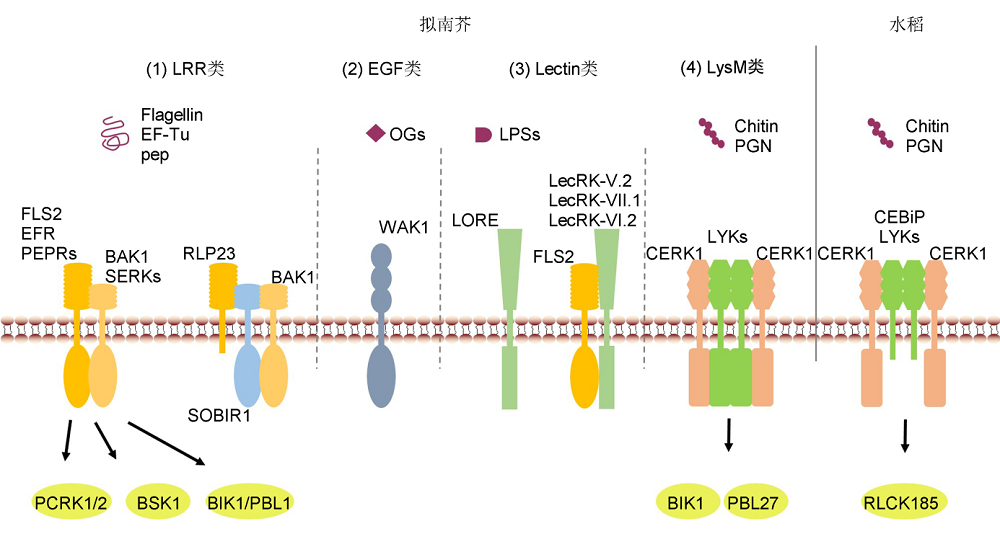
图1 受体激酶和受体蛋白对免疫信号的识别(1) 拟南芥中, LRR类受体激酶和受体蛋白识别病原微生物蛋白或多肽序列; (2) 拟南芥EGF类受体激酶WAK1识别寡聚半乳糖醛酸; (3) Lectin类受体激酶参与免疫信号识别; (4) 拟南芥和水稻LysM类受体激酶或受体蛋白识别几丁质和肽聚糖。
Figure 1 Recognition of the immune signals by receptor kinases and receptor proteins(1) In Arabidopsis, LRR-type receptor kinase and receptor protein recognize pathogenic protein or polypeptide; (2) EGF-type receptor WAK1 recognize oligogalacturonides (OGs); (3) Lectin-type receptor kinase participate in immune signaling recognition; (4) LysM-type receptor kinase or receptor protein recognize chitin and peptidoglycan (PGN) signal in Arabidopsis and rice.
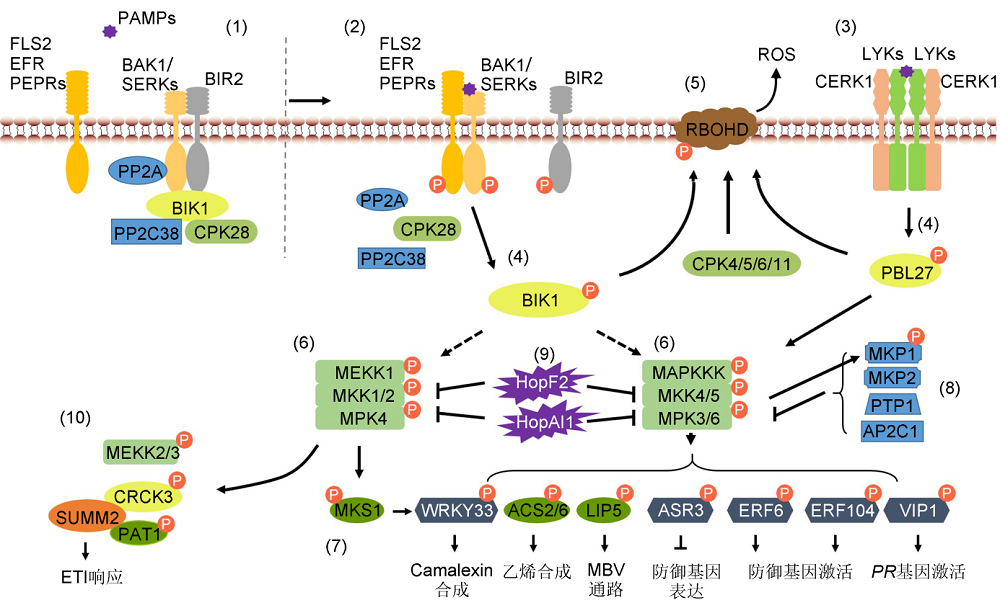
图2 植物中防御相关蛋白的磷酸化修饰调控免疫信号途径(1) 拟南芥中, 未接受病原信号时, PP2A负调控BAK1的激活, 同时BIK1的活性受到PP2C38和CPK28的共同抑制。(2), (3) 病原菌侵染时, LRR类PRRs识别PAMPs, 结合BAK1后被磷酸化激活, LysM类受体激酶CERK1结合LYKs激活免疫信号。(4) 激活的PRRs磷酸化激活胞质内受体激酶(BIK1和PBL27)等。(5) BIK1与CPKs共同磷酸化激活RBOHD, 促进ROS的产生。(6), (7) 激活的RLCKs将信号传递至MAPK信号级联, 导致MAPK激酶被激活。激活的MAPK磷酸化修饰不同的下游底物, 调控不同的防御应答。(8) MKP1、MKP2、PTP1以及AP2C1等磷酸酶负调控MAPKs的活性。(9) 病原菌分泌的效应蛋白HopF2和HopAI1抑制MAPK信号级联途径。(10) 抗病蛋白SUMM2通过监测MPK4对底物MEKK2、CRCK3以及PAT1的磷酸化修饰, 适时地激活ETI途径。
Figure 2 Phosphorylation of defense related proteins in plant immune signaling pathway(1) In Arabidopsis, PP2A negatively regulates the activation of BAK1, meanwhile PP2C38 and CPK28 negatively regulate the phosphorylation of BIK1 without pathogen infection. (2), (3) LRR-type PRRs combine with BAK1 and LysM-type RLK CERK1 combine with LYKs to activate immune signaling after perception of pathogen attack. (4) Activated PRRs phosphorylate BIK1 or PBL27. (5) BIK1 and CPKs phosphorylate RBOHD to promote ROS generation. (6), (7) MAPK cascades are activated and then phosphorylate different substrates to regulate different defense responses. (8) MKP1, MKP2, PTP1 and AP2C1 negatively regulate the activity of MAPKs. (9) Bacterial pathogens inject effector proteins HopF2 and HopAI1 to inhibit the activation of MAPK cascades. (10) Resistance protein SUMM2 activate ETI pathway in time by monitoring the phosphorylation statue of MEKK2, CRCK3 and PAT1, the substrates of MPK4.
| [1] |
毕国志, 周俭民 ( 2017). 厚积薄发: 我国植物-微生物互作研究取得突破. 植物学报 52, 685-688.
DOI URL |
| [2] | 闫佳, 刘雅琼, 侯岁稳 ( 2018). 植物抗病蛋白研究进展. 植物学报 53, 250-263. |
| [3] |
Albert I, Böhm H, Albert M, Feiler CE, Imkampe J, Wallmeroth N, Brancato C, Raaymakers TM, Oome S, Zhang HQ, Krol E, Grefen C, Gust AA, Chai JJ, Hedrich R, Van den Ackerveken G, Nürnberger T ( 2015). An RLP23-SOBIR1-BAK1 complex mediates NLP-triggered immunity. Nat Plants 1, 15140.
DOI |
| [4] |
Anderson JC, Bartels S, González Besteiro MA, Sha- hollari B, Ulm R, Peck SC ( 2011). Arabidopsis MAP Kinase Phosphatase 1 (AtMKP1) negatively regulates MPK6-mediated PAMP responses and resistance against bacteria. Plant J 67, 258-268.
DOI URL PMID |
| [5] |
Andreasson E, Jenkins T, Brodersen P, Thorgrimsen S, Petersen NHT, Zhu SJ, Qiu JL, Micheelsen P, Rocher A, Petersen M, Newman MA, Bjørn Nielsen H, Hirt H, Somssich I, Mattsson O, Mundy J ( 2005). The MAP kinase substrate MKS1 is a regulator of plant defense responses. EMBO J 24, 2579-2589.
DOI URL PMID |
| [6] |
Ao Y, Li ZQ, Feng DR, Xiong F, Liu J, Li JF, Wang ML, Wang JF, Liu B, Wang HB ( 2014). OsCERK1 and OsRLCK176 play important roles in peptidoglycan and chitin signaling in rice innate immunity. Plant J 80, 1072-1084.
DOI URL PMID |
| [7] |
Axtell MJ, Staskawicz BJ ( 2003). Initiation of RPS2- specified disease resistance in Arabidopsis is coupled to the AvrRpt2-directed elimination of RIN4. Cell 112, 369-377.
DOI URL PMID |
| [8] | Bethke G, Unthan T, Uhrig JF, Poschl Y, Gust AA, Scheel D, Lee J ( 2009). Flg22 regulates the release of an ethy- lene response factor substrate from MAP kinase 6 in Arabidopsis thaliana via ethylene signaling. Proc Natl Acad Sci USA 106, 8067-8072. |
| [9] |
Bi GZ, Zhou JM ( 2017). MAP kinase signaling pathways: a hub of plant-microbe interactions. Cell Host Microbe 21, 270-273.
DOI URL PMID |
| [10] | Bi GZ, Zhou ZY, Wang WB, Li L, Rao SF, Wu Y, Zhang XJ, Menke FLH, Chen S, Zhou JM ( 2018). Receptor-like cytoplasmic kinases directly link diverse pattern recog- nition receptors to the activation of mitogen-activated protein kinase cascades in Arabidopsis. Plant Cell 30, 1543-1561. |
| [11] |
Bigeard J, Colcombet J, Hirt H ( 2015). Signaling mech- anisms in pattern-triggered immunity (PTI). Mol Plant 8, 521-539.
DOI URL PMID |
| [12] |
Bigeard J, Rayapuram N, Pflieger D, Hirt H ( 2014). Phosphorylation-dependent regulation of plant chromatin and chromatin-associated proteins. Proteomics 14, 2127-2140.
DOI URL PMID |
| [13] |
Bouwmeester K, de Sain M, Weide R, Gouget A, Klamer S, Canut H, Govers F ( 2011). The lectin receptor kinase LecRK-I.9 is a novel Phytophthora resistance component and a potential host target for a RXLR effector. PLoS Pathog 7, e1001327.
DOI URL PMID |
| [14] |
Bouwmeester K, Govers F ( 2009). Arabidopsis L-type lectin receptor kinases: phylogeny, classification, and expression profiles. J Exp Bot 60, 4383-4396.
DOI URL PMID |
| [15] |
Brutus A, Sicilia F, Macone A, Cervone F, De Lorenzo G ( 2010). A domain swap approach reveals a role of the plant wall-associated kinase 1 (WAK1) as a receptor of oligogalacturonides. Proc Natl Acad Sci USA 107, 9452-9457.
DOI URL PMID |
| [16] |
Cao YR, Liang Y, Tanaka K, Nguyen CT, Jedrzejczak RP, Joachimiak A, Stacey G ( 2014). The kinase LYK5 is a major chitin receptor in Arabidopsis and forms a chitin- induced complex with related kinase CERK1. eLife 3, e03766.
DOI URL PMID |
| [17] |
Chen XW, Zuo SM, Schwessinger B, Chern M, Canlas PE, Ruan DL, Zhou XG, Wang J, Daudi A, Petzold CJ, Heazlewood JL, Ronald PC ( 2014). An XA21-associated kinase (OsSERK2) regulates immunity mediated by the XA21 and XA3 immune receptors. Mol Plant 7, 874-892.
DOI URL PMID |
| [18] |
Cheng SH, Willmann MR, Chen HC, Sheen J ( 2002). Calcium signaling through protein kinases. The Arabi- dopsis calcium-dependent protein kinase gene family. Plant Physiol 129, 469-485.
DOI URL PMID |
| [19] |
Chinchilla D, Bauer Z, Regenass M, Boller T, Felix G ( 2006). The Arabidopsis receptor kinase FLS2 binds flg22 and determines the specificity of flagellin perception. Plant Cell 18, 465-476.
DOI URL |
| [20] |
Chung EH, El-Kasmi F, He YJ, Loehr A, Dangl JL ( 2014). A plant phosphoswitch platform repeatedly targeted by type III effector proteins regulates the output of both tiers of plant immune receptors. Cell Host Microbe 16, 484-494.
DOI URL PMID |
| [21] |
Couto D, Niebergall R, Liang XX, Bucherl CA, Sklenar J, Macho AP, Ntoukakis V, Derbyshire P, Altenbach D, Maclean D, Robatzek S, Uhrig J, Menke F, Zhou JM, Zipfel C ( 2016). The Arabidopsis protein phosphatase PP2C38 negatively regulates the central immune kinase BIK1. PLoS Pathog 12, e1005811.
DOI URL PMID |
| [22] |
Couto D, Zipfel C ( 2016). Regulation of pattern recognition receptor signaling in plants. Nat Rev Immunol 16, 537-552.
DOI |
| [23] |
Dangl JL, Jones JDG ( 2001). Plant pathogens and integ- rated defence responses to infection. Nature 411, 826-833.
DOI |
| [24] |
del Pozo O, Pedley KF, Martin GB ( 2004). MAPKKKα is a positive regulator of cell death associated with both plant immunity and disease. EMBO J 23, 3072-3082.
DOI URL |
| [25] | Desclos-Theveniau M, Arnaud D, Huang TY, Lin GJC, Chen WY, Lin YC, Zimmerli L ( 2012). The Arabidopsis lectin receptor kinase LecRK-V.5 represses stomatal immunity induced by Pseudomonas syringae pv. tomato DC3000. PLoS Pathog 8, e1002513. |
| [26] | Djamei A, Pitzschke A, Nakagami H, Rajh I, Hirt H ( 2007). Trojan horse strategy in Agrobacterium transformation: abusing MAPK defense signaling. Science 318, 453-456. |
| [27] | Dodds PN, Rathjen JP ( 2010). Plant immunity: towards an integrated view of plant-pathogen interactions. Nat Rev Genet 11, 539-548. |
| [28] | Du J, Verzaux E, Chaparro-Garcia A, Bijsterbosch G, Keizer LCP, Zhou J, Liebrand TWH, Xie CH, Govers F, Robatzek S, van der Vossen EAG, Jacobsen E, Visser RGF, Kamoun S, Vleeshouwers VGAA ( 2015). Elicitin recognition confers enhanced resistance to Phytophthora infestans in potato. Nat Plants 1, 15034. |
| [29] |
Dubiella U, Seybold H, Durian G, Komander E, Lassig R, Witte CP, Schulze WX, Romeis T ( 2013). Calcium- dependent protein kinase/NADPH oxidase activation cir- cuit is required for rapid defense signal propagation. Proc Natl Acad Sci USA 110, 8744-8749.
DOI URL |
| [30] |
Dyson HJ, Wright PE ( 2005). Intrinsically unstructured proteins and their functions. Nat Rev Mol Cell Biol 6, 197-208.
DOI |
| [31] |
Eschen-Lippold L, Jiang XY, Elmore JM, Mackey D, Shan LB, Coaker G, Scheel D, Lee J ( 2016). Bacterial AvrRpt2- like cysteine proteases block activation of the Arabidopsis mitogen-activated protein kinases, MPK4 and MPK11. Plant Physiol 171, 2223-2238.
DOI URL |
| [32] |
Fuchs S, Grill E, Meskiene I, Schweighofer A ( 2013). Type 2C protein phosphatases in plants. FEBS J 280, 681-693.
DOI URL |
| [33] |
Furlan G, Nakagami H, Eschen-Lippold L, Jiang XY, Majovsky P, Kowarschik K, Hoehenwarter W, Lee J, Trujillo M ( 2017). Changes in PUB22 ubiquitination modes triggered by MITOGEN-ACTIVATED PROTEIN KINASE3 dampen the immune response. Plant Cell 29, 726-745.
DOI URL |
| [34] | Galletti R, Ferrari S, De Lorenzo G ( 2011). Arabidopsis MPK3 and MPK6 play different roles in basal and oligo- galacturonide- or flagellin-induced resistance against Bot- rytis cinerea . Plant Physiol 157, 804-814. |
| [35] |
Gao XG, Chen X, Lin WW, Chen SX, Lu DP, Niu YJ, Li L, Cheng C, McCormack M, Sheen J, Shan LB, He P ( 2013). Bifurcation of Arabidopsis NLR immune signaling via Ca 2+-dependent protein kinases . PLoS Pathog 9, e1003127.
DOI URL |
| [36] |
Gómez-Gómez L, Boller T ( 2000). FLS2: an LRR receptor- like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol Cell 5, 1003-1011.
DOI URL |
| [37] | Gravino M, Savatin DV, Macone A, De Lorenzo G ( 2015). Ethylene production in Botrytis cinerea - and oligogala- cturonide-induced immunity requires calcium-dependent protein kinases. Plant J 84, 1073-1086. |
| [38] |
Gust AA, Felix G ( 2014). Receptor like proteins associate with SOBIR1-type of adaptors to form bimolecular receptor kinases. Curr Opin Plant Biol 21, 104-111.
DOI URL |
| [39] |
Halter T, Imkampe J, Mazzotta S, Wierzba M, Postel S, Bücherl C, Kiefer C, Stahl M, Chinchilla D, Wang XF, Nürnberger T, Zipfel C, Clouse S, Borst JW, Boeren S, de Vries SC, Tax F, Kemmerling B ( 2014). The leucine- rich repeat receptor kinase BIR2 is a negative regulator of BAK1 in plant immunity. Curr Biol 24, 134-143.
DOI URL |
| [40] | Han L, Li GJ, Yang KY, Mao GH, Wang RG, Liu YD, Zhang SQ ( 2010). Mitogen-activated protein kinase 3 and 6 regulate Botrytis cinerea -induced ethylene production in Arabidopsis. Plant J 64, 114-127. |
| [41] |
Hind SR, Strickler SR, Boyle PC, Dunham DM, Bao ZL, O'Doherty IM, Baccile JA, Hoki JS, Viox EG, Clarke CR, Vinatzer BA, Schroeder FC, Martin GB ( 2016). Tomato receptor FLAGELLIN-SENSING 3 binds flgII-28 and acti- vates the plant immune system. Nat Plants 2, 16128.
DOI |
| [42] | Huang PY, Yeh YH, Liu AC, Cheng CP, Zimmerli L ( 2014). The Arabidopsis LecRK-VI.2 associates with the pattern- recognition receptor FLS2 and primes Nicotiana bent- hamiana pattern-triggered immunity. Plant J 79, 243-255. |
| [43] | Ishihama N, Yamada R, Yoshioka M, Katou S, Yoshioka H ( 2011). Phosphorylation of the Nicotiana benthamiana WRKY8 transcription factor by MAPK functions in the defense response. Plant Cell 23, 1153-1170. |
| [44] | Jiang LY, Anderson JC, Besteiro MAG, Peck SC ( 2017). Phosphorylation of Arabidopsis MAP Kinase Phosphatase 1 (MKP1) is required for PAMP responses and resistance against Bacteria . Plant Physiol 175, 1839-1852. |
| [45] |
Jones JDG, Dangl JL ( 2006). The plant immune system. Nature 444, 323-329.
DOI |
| [46] | Joo S, Liu YD, Lueth A, Zhang SQ ( 2008). MAPK phos- phorylation-induced stabilization of ACS6 protein is me- diated by the non-catalytic C-terminal domain, which also contains the cis -determinant for rapid degradation by the 26S proteasome pathway. Plant J 54, 129-140. |
| [47] |
Kadota Y, Sklenar J, Derbyshire P, Stransfeld L, Asai S, Ntoukakis V, Jones JD, Shirasu K, Menke F, Jones A, Zipfel C ( 2014). Direct regulation of the NADPH oxidase RBOHD by the PRR-associated kinase BIK1 during plant immunity. Mol Cell 54, 43-55.
DOI URL |
| [48] |
Kaku H, Nishizawa Y, Ishii-Minami N, Akimoto-Tomiyama C, Dohmae N, Takio K, Minami E, Shibuya N ( 2006). Plant cells recognize chitin fragments for defense signaling through a plasma membrane receptor. Proc Natl Acad Sci USA 103, 11086-11091.
DOI URL |
| [49] | Kong Q, Sun TJ, Qu N, Ma JL, Li M, Cheng YT, Zhang Q, Wu D, Zhang ZB, Zhang YL ( 2016). Two redundant receptor-like cytoplasmic kinases function downstream of pattern recognition receptors to regulate activation of SA biosynthesis. Plant Physiol 171, 1344-1354. |
| [50] |
Lal NK, Nagalakshmi U, Hurlburt NK, Flores R, Bak A, Sone P, Ma XY, Song GY, Walley J, Shan LB, He P, Casteel C, Fisher AJ, Dinesh-Kumar SP ( 2018). The receptor-like cytoplasmic kinase BIK1 localizes to the nucleus and regulates defense hormone expression during plant innate immunity. Cell Host Microbe 23, 485-497.
DOI URL |
| [51] |
Lee D, Bourdais G, Yu G, Robatzek S, Coaker G ( 2015). Phosphorylation of the plant immune regulator RPM1- INTERACTING PROTEIN4 enhances plant plasma mem- brane H +-ATPase activity and inhibits flagellin-triggered immune responses in Arabidopsis . Plant Cell 27, 2042-2056.
DOI URL |
| [52] |
Lee JS, Ellis BE ( 2007). Arabidopsis MAPK phosphatase 2 (MKP2) positively regulates oxidative stress tolerance and inactivates the MPK3 and MPK6 MAPKs. J Biol Chem 282, 25020-25029.
DOI URL |
| [53] |
Lenzoni G, Liu JL, Knight MR ( 2018). Predicting plant immunity gene expression by identifying the decoding mechanism of calcium signatures. New Phytol 217, 1598-1609.
DOI URL |
| [54] |
Li B, Jiang S, Yu X, Cheng C, Chen SX, Cheng YB, Yuan JS, Jiang DH, He P, Shan LB ( 2015). Phosphorylation of trihelix transcriptional repressor ASR3 by MAP KINASE4 negatively regulates Arabidopsis immunity. Plant Cell 27, 839-856.
DOI URL |
| [55] |
Li FJ, Cheng C, Cui FH, de Oliveira MVV, Yu X, Meng XZ, Intorne AC, Babilonia K, Li MY, Li B, Chen SX, Ma XF, Xiao SY, Zheng Y, Fei ZJ, Metz RP, Johnson CD, Koiwa H, Sun WX, Li ZH, de Souza GA, Shan LB, He P ( 2014a). Modulation of RNA polymerase II phosphorylation downstream of pathogen perception orchestrates plant immunity. Cell Host Microbe 16, 748-758.
DOI URL |
| [56] |
Li GJ, Meng XZ, Wang RG, Mao GH, Han L, Liu YD, Zhang SQ ( 2012). Dual-level regulation of ACC synthase activity by MPK3/MPK6 cascade and its downstream WRKY transcription factor during ethylene induction in Arabi- dopsis. PLoS Genet 8, e1002767.
DOI URL |
| [57] | Li H, Zhou SY, Zhao WS, Su SC, Peng YL ( 2009). A novel wall-associated receptor-like protein kinase gene, Os- WAK1 , plays important roles in rice blast disease resis- tance. Plant Mol Biol 69, 337-346. |
| [58] |
Li J, Wen JQ, Lease KA, Doke JT, Tax FE, Walker JC ( 2002). BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 110, 213-222.
DOI URL |
| [59] |
Li L, Li M, Yu LP, Zhou ZY, Liang XX, Liu ZX, Cai GH, Gao LY, Zhang XJ, Wang YC, Chen S, Zhou JM ( 2014b). The FLS2-associated kinase BIK1 directly phosphorylates the NADPH oxidase RbohD to control plant immunity. Cell Host Microbe 15, 329-338.
DOI URL |
| [60] |
Liang XX, Ding PT, Lian KH, Wang JL, Ma MM, Li L, Li L, Li M, Zhang SJ, Chen S, Zhang YL, Zhou JM ( 2016). Arabidopsis heterotrimeric G proteins regulate immunity by directly coupling to the FLS2 receptor. eLife 5, e13568.
DOI URL |
| [61] |
Liang XX, Ma MM, Zhou ZY, Wang JL, Yang XR, Rao SF, Bi GZ, Li L, Zhang XJ, Chai JJ, Chen S, Zhou JM ( 2018). Ligand-triggered de-repression of Arabidopsis heterotrimeric G proteins coupled to immune receptor kinases. Cell Res 28, 529-543.
DOI |
| [62] |
Liebrand TW, van den Berg GCM, Zhang Z, Smit P, Cordewener JHG, America AHP, Sklenar J, Jones AME, Tameling WIL, Robatzek S, Thomma BPHJ, Joosten MHAJ ( 2013). Receptor-like kinase SOBIR1/EVR interacts with receptor-like proteins in plant immunity against fungal infection. Proc Natl Acad Sci USA 110, 10010-10015.
DOI URL |
| [63] |
Lin WW, Li B, Lu DP, Chen SX, Zhu N, He P, Shan LB ( 2014). Tyrosine phosphorylation of protein kinase com- plex BAK1/BIK1 mediates Arabidopsis innate immunity. Proc Natl Acad Sci USA 111, 3632-3637.
DOI URL |
| [64] |
Lin WW, Lu DP, Gao XQ, Jiang S, Ma XY, Wang ZH, Mengiste T, He P, Shan LB ( 2013). Inverse modulation of plant immune and brassinosteroid signaling pathways by the receptor-like cytoplasmic kinase BIK1. Proc Natl Acad Sci USA 110, 12114-12119.
DOI URL |
| [65] | Lin ZJD, Liebrand TWH, Yadeta KA, Coaker G ( 2015). PBL13 is a serine/threonine protein kinase that negatively regulates Arabidopsis immune responses. Plant Physiol 169, 2950-2962. |
| [66] |
Liu J, Liu B, Chen SF, Gong BQ, Chen LJ, Zhou Q, Xiong F, Wang ML, Feng DR, Li JF, Wang HB, Wang JF ( 2018). A tyrosine phosphorylation cycle regulates fungal activation of a plant receptor Ser/Thr kinase. Cell Host Microbe 23, 241-253.
DOI URL |
| [67] |
Liu JY, Yang HB, Bao F, Ao K, Zhang XY, Zhang YL, Yang SH ( 2015). IBR5 modulates temperature-dependent, R protein CHS3-mediated defense responses in Arabidopsis. PLoS Genet 11, e1005584.
DOI URL |
| [68] | Liu N, Hake K, Wang W, Zhao T, Romeis T, Tang DZ ( 2017). CALCIUM-DEPENDENT PROTEIN KINASE5 as- sociates with the truncated NLR protein TIR-NBS2 to con- tribute to exo70B1 -mediated immunity. Plant Cell 29, 746-759. |
| [69] |
Liu ZX, Wu Y, Yang F, Zhang YY, Chen S, Xie Q, Tian XJ, Zhou JM ( 2013). BIK1 interacts with PEPRs to mediate ethylene-induced immunity. Proc Natl Acad Sci USA 110, 6205-6210.
DOI URL |
| [70] |
Lu DP, Wu SJ, Gao XQ, Zhang YL, Shan LB, He P ( 2010). A receptor-like cytoplasmic kinase, BIK1, associates with a flagellin receptor complex to initiate plant innate immunity. Proc Natl Acad Sci USA 107, 496-501.
DOI URL |
| [71] |
Luan S ( 2003). Protein phosphatases in plants. Annu Rev Plant Biol 54, 63-92.
DOI URL |
| [72] |
Lumbreras V, Vilela B, Irar S, Solé M, Capellades M, Valls M, Coca M, Pagès M ( 2010). MAPK phosphatase MKP2 mediates disease responses in Arabidopsis and func- tionally interacts with MPK3 and MPK6. Plant J 63, 1017-1030.
DOI URL |
| [73] |
Luo XM, Xu N, Huang JK, Gao F, Zou HS, Boudsocq M, Coaker G, Liu J ( 2017). A lectin receptor-like kinase mediates pattern-triggered salicylic acid signaling. Plant Physiol 174, 2501-2514.
DOI URL |
| [74] | Luo Y, Caldwell KS, Wroblewski T, Wright ME, Mich- elmore RW ( 2009). Proteolysis of a negative regulator of innate immunity is dependent on resistance genes in tomato and Nicotiana benthamiana and induced by mul- tiple bacterial effectors. Plant Cell 21, 2458-2472. |
| [75] |
Macho AP, Schwessinger B, Ntoukakis V, Brutus A, Segonzac C, Roy S, Kadota Y, Oh MH, Sklenar J, Derbyshire P, Lozano-Durán R, Malinovsky FG, Monaghan J, Menke FL, Huber SC, He SY, Zipfel C ( 2014). A bacterial tyrosine phosphatase inhibits plant pa- ttern recognition receptor activation. Science 343, 1509-1512.
DOI URL |
| [76] |
Mackey D, Belkhadir Y, Alonso JM, Ecker JR, Dangl JL ( 2003). Arabidopsis RIN4 is a target of the type III virulence effector AvrRpt2 and modulates RPS2-mediated resistance. Cell 112, 379-389.
DOI URL |
| [77] | Mackey D, Holt BF 3rd, Wiig A, Dangl JL ( 2002). RIN4 interacts with Pseudomonas syringae type III effector molecules and is required for RPM1-mediated resistance in Arabidopsis. Cell 108, 743-754. |
| [78] |
Mao GH, Meng XZ, Liu YD, Zheng ZY, Chen ZX, Zhang SQ ( 2011). Phosphorylation of a WRKY transcription factor by two pathogen-responsive MAPKs drives phytoalexin bio- synthesis in Arabidopsis. Plant Cell 23, 1639-1653.
DOI URL |
| [79] |
Meng XZ, Xu J, He YX, Yang KY, Mordorski B, Liu YD, Zhang SQ ( 2013). Phosphorylation of an ERF trans- cription factor by Arabidopsis MPK3/MPK6 regulates plant defense gene induction and fungal resistance. Plant Cell 25, 1126-1142.
DOI URL |
| [80] |
Meng XZ, Zhang SQ ( 2013). MAPK cascades in plant disease resistance signaling. Annu Rev Phytopathol 51, 245-266.
DOI URL |
| [81] |
Mine A, Berens ML, Nobori T, Anver S, Fukumoto K, Winkelmüller TM, Takeda A, Becker D, Tsuda K ( 2017). Pathogen exploitation of an abscisic acid- and jasmo- nate-inducible MAPK phosphatase and its interception by Arabidopsis immunity. Proc Natl Acad Sci USA 114, 7456-7461.
DOI URL |
| [82] |
Mithoe SC, Ludwig C, Pel MJC, Cucinotta M, Casartelli A, Mbengue M, Sklenar J, Derbyshire P, Robatzek S, Pieterse CMJ, Aebersold R, Menke FLH ( 2016). Attenuation of pattern recognition receptor signaling is mediated by a MAP kinase kinase kinase. EMBO Rep 17, 441-454.
DOI URL |
| [83] |
Miya A, Albert P, Shinya T, Desaki Y, Ichimura K, Shirasu K, Narusaka Y, Kawakami N, Kaku H, Shibuya N ( 2007). CERK1, a LysM receptor kinase, is essential for chitin elicitor signaling in Arabidopsis. Proc Natl Acad Sci USA 104, 19613-19618.
DOI URL |
| [84] |
Monaghan J, Matschi S, Shorinola O, Rovenich H, Matei A, Segonzac C, Malinovsky FG, Rathjen JP, MacLean D, Romeis T, Zipfel C ( 2014). The calcium-dependent protein kinase CPK28 buffers plant immunity and regulates BIK1 turnover. Cell Host Microbe 16, 605-615.
DOI URL |
| [85] |
Nam KH, Li JM ( 2002). BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling. Cell 110, 203-212.
DOI URL |
| [86] |
Nühse TS, Bottrill AR, Jones AME, Peck SC ( 2007). Quantitative phosphoproteomic analysis of plasma mem- brane proteins reveals regulatory mechanisms of plant innate immune responses. Plant J 51, 931-940.
DOI URL |
| [87] |
Nühse TS, Stensballe A, Jensen ON, Peck SC ( 2004). Phosphoproteomics of the Arabidopsis plasma membrane and a new phosphorylation site database. Plant Cell 16, 2394-2405.
DOI URL |
| [88] |
Oh CS, Martin GB ( 2011). Effector-triggered immunity mediated by the Pto kinase. Trends Plant Sci 16, 132-140.
DOI URL |
| [89] |
Park CJ, Peng Y, Chen XW, Dardick C, Ruan DL, Bart R, Canlas PE, Ronald PC ( 2008). Rice XB15, a protein phosphatase 2C, negatively regulates cell death and XA21-mediated innate immunity. PLoS Biol 6, e231.
DOI URL |
| [90] |
Pedley KF, Martin GB ( 2005). Role of mitogen-activated protein kinases in plant immunity. Curr Opin Plant Biol 8, 541-547.
DOI URL |
| [91] |
Perraki A, DeFalco TA, Derbyshire P, Avila J, Séré D, Sklenar J, Qi XY, Stransfeld L, Schwessinger B, Kadota Y, Macho AP, Jiang SS, Couto D, Torii KU, Menke FLH, Zipfel C ( 2018). Phosphocode-dependent functional dichotomy of a common co-receptor in plant signaling. Nature 561, 248-252.
DOI |
| [92] |
Pruitt RN, Schwessinger B, Joe A, Thomas N, Liu FR, Albert M, Robinson MR, Chan LJG, Luu DD, Chen HM, Bahar O, Daudi A, De Vleesschauwer D, Caddell D, Zhang WG, Zhao XX, Li X, Heazlewood JL, Ruan DL, Majumder D, Chern M, Kalbacher H, Midha S, Patil PB, Sonti RV, Petzold CJ, Liu CC, Brodbelt JS, Felix G, Ronald PC ( 2015). The rice immune receptor XA21 recognizes a tyrosine-sulfated protein from a Gramne- gative bacterium. Sci Adv 1, e1500245.
DOI URL |
| [93] |
Qiu JL, Fiil BK, Petersen K, Nielsen HB, Botanga CJ, Thorgrimsen S, Palma K, Suarez-Rodriguez MC, Sandbech-Clausen S, Lichota J, Brodersen P, Grasser KD, Mattsson O, Glazebrook J, Mundy J, Petersen M ( 2008). Arabidopsis MAP kinase 4 regulates gene ex- pression through transcription factor release in the nu- cleus. EMBO J 27, 2214-2221.
DOI URL |
| [94] | Ranf S, Gisch N, Schäffer M, Illig T, Westphal L, Knirel YA, Sánchez-Carballo PM, Zähringer U, Hückelhoven R, Lee J, Scheel D ( 2015). A lectin S-domain receptor kinase mediates lipopolysaccharide sensing in Arabidopsis thaliana . Nat Immunol 16, 426-433. |
| [95] | Romeis T, Piedras P, Zhang S, Klessig DF, Hirt H, Jones JD ( 1999). Rapid Avr9- and Cf-9-dependent activation of MAP kinases in tobacco cell cultures and leaves: con- vergence of resistance gene, elicitor, wound, and salicy- late responses. Plant Cell 11, 273-287. |
| [96] |
Roux ME, Rasmussen MW, Palma K, Lolle S, Regué AM, Bethke G, Glazebrook J, Zhang WP, Sieburth L, Larsen MR, Mundy J, Petersen M ( 2015). The mRNA decay factor PAT1 functions in a pathway including MAP kinase 4 and immune receptor SUMM2. EMBO J 34, 593-608.
DOI URL |
| [97] |
Saleh A, Withers J, Mohan R, Marqués J, Gu YN, Yan SP, Zavaliev R, Nomoto M, Tada Y, Dong XN ( 2015). Posttranslational modifications of the master transcrip- tional regulator NPR1 enable dynamic but tight control of plant immune responses. Cell Host Microbe 18, 169-182.
DOI URL |
| [98] |
Schweighofer A, Kazanaviciute V, Scheikl E, Teige M, Doczi R, Hirt H, Schwanninger M, Kant M, Schuurink R, Mauch F, Buchala A, Cardinale F, Meskiene I ( 2007). The PP2C-type phosphatase AP2C1, which negatively regulates MPK4 and MPK6, modulates innate immunity, jasmonic acid, and ethylene levels in Arabidopsis. Plant Cell 19, 2213-2224.
DOI URL |
| [99] | Schweighofer A, Meskiene I ( 2015). Phosphatases in plants. In: Schulze W, ed. Plant Phosphoproteomics. Methods in Molecular Biology, Vol. 1 306. New York: Humana Press. pp. 25-46. |
| [100] |
Segonzac C, Macho AP, Sanmartín M, Ntoukakis V, Sánchez-Serrano JJ, Zipfel C ( 2014). Negative control of BAK1 by protein phosphatase 2A during plant innate immunity. EMBO J 33, 2069-2079.
DOI URL |
| [101] | Sheikh AH, Eschen-Lippold L, Pecher P, Hoehenwarter W, Sinha AK, Scheel D, Lee J ( 2016). Regulation of WRK- Y46 transcription factor function by mitogen-activated pro- tein kinases in Arabidopsis thaliana . Front Plant Sci 7, 61. |
| [102] |
Shi H, Shen QJ, Qi YP, Yan HJ, Nie HZ, Chen YF, Zhao T, Katagiri F, Tang DZ ( 2013). BR-SIGNALING KINASE 1 physically associates with FLAGELLIN SENSING 2 and regulates plant innate immunity in Arabidopsis. Plant Cell 25, 1143-1157.
DOI URL |
| [103] |
Shinya T, Nakagawa T, Kaku H, Shibuya N ( 2015). Chitin- mediated plant-fungal interactions: catching, hiding and handshaking. Curr Opin Plant Biol 26, 64-71.
DOI URL |
| [104] |
Shinya T, Yamaguchi K, Desaki Y, Yamada K, Narisawa T, Kobayashi Y, Maeda K, Suzuki M, Tanimoto T, Takeda J, Nakashima M, Funama R, Narusaka M, Narusaka Y, Kaku H, Kawasaki T, Shibuya N ( 2014). Selective regu- lation of the chitin-induced defense response by the Arabi- dopsis receptor-like cytoplasmic kinase PBL27. Plant J 79, 56-66.
DOI URL |
| [105] | Shubchynskyy V, Boniecka J, Schweighofer A, Simulis J, Kvederaviciute K, Stumpe M, Mauch F, Balazadeh S, Mueller-Roeber B, Boutrot F, Zipfel C, Meskiene I ( 2017). Protein phosphatase AP2C1 negatively regulates basal resistance and defense responses to Pseudomonas syringae . J Exp Bot 68, 1169-1183. |
| [106] | Sidonskaya E, Schweighofer A, Shubchynskyy V, Kam- merhofer N, Hofmann J, Wieczorek K, Meskiene I ( 2016). Plant resistance against the parasitic nematode Heterodera schachtii is mediated by MPK3 and MPK6 kinases, which are controlled by the MAPK phosphatase AP2C1 in Arabidopsis. J Exp Bot 67, 107-118. |
| [107] |
Singh P, Kuo YC, Mishra S, Tsai CH, Chien CC, Chen CW, Desclos-Theveniau M, Chu PW, Schulze B, Chinchilla D, Boller T, Zimmerli L ( 2012). The lectin receptor kinase-VI.2 is required for priming and positively regulates Arabidopsis pattern-triggered immunity. Plant Cell 24, 1256-1270.
DOI URL |
| [108] |
Spoel SH, Mou ZL, Tada Y, Spivey NW, Genschik P, Dong XN ( 2009). Proteasome-mediated turnover of the trans- cription coactivator NPR1 plays dual roles in regulating plant immunity. Cell 137, 860-872.
DOI URL |
| [109] |
Tang DZ, Wang GX, Zhou JM ( 2017). Receptor kinases in plant-pathogen interactions: more than pattern recognition. Plant Cell 29, 618-637.
DOI URL |
| [110] |
Wan JR, Tanaka K, Zhang XC, Son GH, Brechenmacher L, Tran HNN, Stacey G ( 2012). LYK4, a lysin motif receptor-like kinase, is important for chitin signaling and plant innate immunity in Arabidopsis. Plant Physiol 160, 396-406.
DOI URL |
| [111] |
Wang F, Shang YF, Fan BF, Yu JQ, Chen ZX ( 2014a). Arabidopsis LIP5, a positive regulator of multivesicular body biogenesis, is a critical target of pathogen-responsive MAPK cascade in plant basal defense. PLoS Pathog 10, e1004243.
DOI URL |
| [112] |
Wang J, Zhou L, Shi H, Chern M, Yu H, Yi H, He M, Yin JJ, Zhu XB, Li Y, Li WT, Liu JL, Wang JC, Chen XQ, Qing H, Wang YP, Liu GF, Wang WM, Li P, Wu XJ, Zhu LH, Zhou JM, Ronald PC, Li SG, Li JY, Chen XW ( 2018a). A single transcription factor promotes both yield and im- munity in rice. Science 361, 1026-1028.
DOI URL |
| [113] |
Wang JL, Grubb LE, Wang JY, Liang XX, Li L, Gao CL, Ma MM, Feng F, Li M, Li L, Zhang XJ, Yu FF, Xie Q, Chen S, Zipfel C, Monaghan J, Zhou JM ( 2018b). A regulatory module controlling homeostasis of a plant immune kinase. Mol Cell 69, 493-504.
DOI URL |
| [114] |
Wang XF, Kota U, He K, Blackburn K, Li J, Goshe MB, Huber SC, Clouse SD ( 2008). Sequential transphosph orylation of the BRI1/BAK1 receptor kinase complex im- pacts early events in brassinosteroid signaling. Dev Cell 15, 220-235.
DOI URL |
| [115] |
Wang Y, Bouwmeester K, Beseh P, Shan WX, Govers F ( 2014b). Phenotypic analyses of Arabidopsis T-DNA insertion lines and expression profiling reveal that multiple L-type lectin receptor kinases are involved in plant imm- unity. Mol Plant Microbe Interact 27, 1390-1402.
DOI URL |
| [116] | Wang YJ, Li JF, Hou SG, Wang XW, Li YA, Ren DT, Chen S, Tang XY, Zhou JM ( 2010). A Pseudomonas syringae ADP-ribosyltransferase inhibits Arabidopsis mitogen-acti- vated protein kinase kinases. Plant Cell 22, 2033-2044. |
| [117] | Widjaja I, Lassowskat I, Bethke G, Eschen-Lippold L, Long HH, Naumann K, Dangl JL, Scheel D, Lee J ( 2010). A protein phosphatase 2C, responsive to the bacterial effector AvrRpm1 but not to the AvrB effector, regulates defense responses in Arabidopsis. Plant J 61, 249-258. |
| [118] |
Willmann R, Lajunen HM, Erbs G, Newman MA, Kolb D, Tsuda K, Katagiri F, Fliegmann J, Bono JJ, Cullimore JV, Jehle AK, Götz F, Kulik A, Molinaro A, Lipka V, Gust AA, Nürnberger T ( 2011). Arabidopsis lysin-motif proteins LYM1 LYM3 CERK1 mediate bacterial pepti- doglycan sensing and immunity to bacterial infection. Proc Natl Acad Sci USA 108, 19824-19829.
DOI URL |
| [119] | Wilton M, Subramaniam R, Elmore J, Felsensteiner C, Coaker G, Desveaux D ( 2010). The type III effector HopF2 Pto targets Arabidopsis RIN4 protein to promote Pseudomonas syringae virulence. Proc Natl Acad Sci USA 107, 2349-2354. |
| [120] | Xie CG, Zhou XN, Deng XW, Guo Y ( 2010). PKS5, a SNF1- related kinase, interacts with and phosphorylates NPR1, and modulates expression of WRKY38 and WRKY62. J Genet Genomics 37, 359-369. |
| [121] |
Yamada K, Yamaguchi K, Shirakawa T, Nakagami H, Mine A, Ishikawa K, Fujiwara M, Narusaka M, Narusaka Y, Ichimura K, Kobayashi Y, Matsui H, Nomura Y, Nomoto M, Tada Y, Fukao Y, Fukamizo T, Tsuda K, Shirasu K, Shibuya N, Kawasaki T ( 2016a). The Arabidopsis CERK1- associated kinase PBL27 connects chitin perception to MAPK activation. EMBO J 35, 2468-2483.
DOI URL |
| [122] |
Yamada K, Yamashita-Yamada M, Hirase T, Fujiwara T, Tsuda K, Hiruma K, Saijo Y ( 2016b). Danger peptide receptor signaling in plants ensures basal immunity upon pathogen-induced depletion of BAK1. EMBO J 35, 46-61.
DOI URL |
| [123] |
Yamaguchi K, Yamada K, Ishikawa K, Yoshimura S, Hayashi N, Uchihashi K, Ishihama N, Kishi-Kaboshi M, Takahashi A, Tsuge S, Ochiai H, Tada Y, Shimamoto K, Yoshioka H, Kawasaki T ( 2013). A receptor-like cyto- plasmic kinase targeted by a plant pathogen effector is directly phosphorylated by the chitin receptor and medi- ates rice immunity. Cell Host Microbe 13, 347-357.
DOI URL |
| [124] |
Yamaguchi Y, Huffaker A, Bryan AC, Tax FE, Ryan CA ( 2010). PEPR2 is a second receptor for the Pep1 and Pep2 peptides and contributes to defense responses in Arabidopsis. Plant Cell 22, 508-522.
DOI URL |
| [125] | Yamaguchi Y, Pearce G, Ryan CA ( 2006). The cell surface leucine-rich repeat receptor for At Pep1, an endogenous peptide elicitor in Arabidopsis, is functional in transgenic tobacco cells. Proc Natl Acad Sci USA 103, 10104-10109. |
| [126] |
Yan HJ, Zhao YF, Shi H, Li J, Wang YC, Tang DZ ( 2018). BRASSINOSTEROID-SIGNALING KINASE 1 phosphory- lates MAPKKK5 to regulate immunity in Arabidopsis. Plant Physiol 176, 2991-3002.
DOI URL |
| [127] |
Yan LM, Ma YY, Liu D, Wei XC, Sun Y, Chen XY, Zhao HD, Zhou JW, Wang ZY, Shui WQ, Lou ZY ( 2012). Structural basis for the impact of phosphorylation on the activation of plant receptor-like kinase BAK1. Cell Res 22, 1304-1308.
DOI |
| [128] |
Yang HB, Shi YT, Liu JY, Guo L, Zhang XY, Yang SH ( 2010). A mutant CHS3 protein with TIR-NB-LRR-LIM domains modulates growth, cell death and freezing tole- rance in a temperature-dependent manner in Arabi- dopsis. Plant J 63, 283-296.
DOI URL |
| [129] |
Yekondi S, Liang FC, Okuma E, Radziejwoski A, Mai HW, Swain S, Singh P, Gauthier M, Chien HC, Murata Y, Zimmerli L ( 2018). Nonredundant functions of Arabidopsis LecRK-V.2 and LecRK-VII.1 in controlling stomatal imm- unity and jasmonate-mediated stomatal closure. New Phytol 218, 253-268.
DOI URL |
| [130] | Zhang J, Shao F, Li Y, Cui HT, Chen LJ, Li HT, Zou Y, Long CZ, Lan LF, Chai JJ, Chen S, Tang XY, Zhou JM ( 2007). A Pseudomonas syringae effector inactivates MAPKs to suppress PAMP-induced immunity in plants. Cell Host Microbe 1, 175-185. |
| [131] | Zhang SQ, Klessig DF ( 1998). Resistance gene N-mediated de novo synthesis and activation of a tobacco mitogen- activated protein kinase by tobacco mosaic virus infection. Proc Natl Acad Sci USA 95, 7433-7438. |
| [132] |
Zhang WG, Fraiture M, Kolb D, Löffelhardt B, Desaki Y, Boutrot FFG, Tör M, Zipfel C, Gust AA, Brunner F ( 2013). Arabidopsis receptor-like protein30 and receptor- like kinase suppressor of BIR1-1/EVERSHED mediate innate immunity to necrotrophic fungi. Plant Cell 25, 4227-4241.
DOI URL |
| [133] |
Zhang ZB, Liu Y, Huang H, Gao MH, Wu D, Kong Q, Zhang YL ( 2017). The NLR protein SUMM2 senses the disruption of an immune signaling MAP kinase cascade via CRCK3. EMBO Rep 18, 292-302.
DOI URL |
| [134] | Zhang ZB, Wu YL, Gao MH, Zhang J, Kong Q, Liu Y, Ba HP, Zhou JM, Zhang YL ( 2012). Disruption of PAMP- induced MAP kinase cascade by a Pseudomonas syringae effector activates plant immunity mediated by the NB-LRR protein SUMM2. Cell Host Microbe 11, 253-263. |
| [135] | Zipfel C, Kunze G, Chinchilla D, Caniard A, Jones JDG, Boller T, Felix G ( 2006). Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium -medi- ated transformation. Cell 125, 749-760. |
| [1] | 王研, 贾博为, 孙明哲, 孙晓丽. 野生大豆耐逆分子调控机制研究进展[J]. 植物学报, 2021, 56(1): 104-115. |
| [2] | 闫晨阳,陈赢男. 4种模式植物LRR VIII-2亚家族基因的鉴定和进化历史分析[J]. 植物学报, 2020, 55(4): 442-456. |
| [3] | 王梦龙,彭小群,陈竹锋,唐晓艳. 植物凝集素类受体蛋白激酶研究进展[J]. 植物学报, 2020, 55(1): 96-105. |
| [4] | 朱丹,曹汉威,李媛,任东涛. 植物蛋白磷酸化的检测方法[J]. 植物学报, 2020, 55(1): 76-82. |
| [5] | 崔胜男,张艺函,徐凡. 异源过表达水稻OsSAPP3基因促进拟南芥叶片衰老[J]. 植物学报, 2019, 54(1): 46-57. |
| [6] | 王鹤飞, 李雪, 董玲丽, 张俊成, 赵茂林, 邢国珍, 王道文, 郑文明. 小麦受体样蛋白激酶及其衍生蛋白的研究进展[J]. 植物学报, 2015, 50(2): 255-262. |
| [7] | 胡江 孙淑斌 徐国华. 植物中丛枝菌根形成的信号途径研究进展[J]. 植物学报, 2007, 24(06): 703-713. |
| [8] | 赖辉煌 王宏斌 王金发. PTP 及其在植物MAPK 途径中的作用[J]. 植物学报, 2007, 24(05): 677-685. |
| [9] | 翁华 冉亮 魏群. 植物蛋白磷酸酶及其在植物抗逆中的作用[J]. 植物学报, 2003, 20(05): 609-615. |
| [10] | 刘贯山 陈珈. 钙依赖蛋白激酶(CDPKs)在植物钙信号转导中的作用[J]. 植物学报, 2003, 20(02): 160-167. |
| [11] | 陈硕 陈珈. 植物中钙依赖蛋白激酶(CDPKs)的结构与功能[J]. 植物学报, 2001, 18(02): 143-148. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
