

Chinese Bulletin of Botany ›› 2022, Vol. 57 ›› Issue (3): 320-326.DOI: 10.11983/CBB22043 cstr: 32102.14.CBB22043
• TECHNIQUES AND METHODS • Previous Articles Next Articles
Haitao Hu, Tingting Qian, Ling Yang( )
)
Received:2022-03-10
Accepted:2022-05-11
Online:2022-05-01
Published:2022-05-18
Contact:
Ling Yang
Haitao Hu, Tingting Qian, Ling Yang. Detection of Reactive Oxygen Species Using H2DCFDA Probe in Plant[J]. Chinese Bulletin of Botany, 2022, 57(3): 320-326.
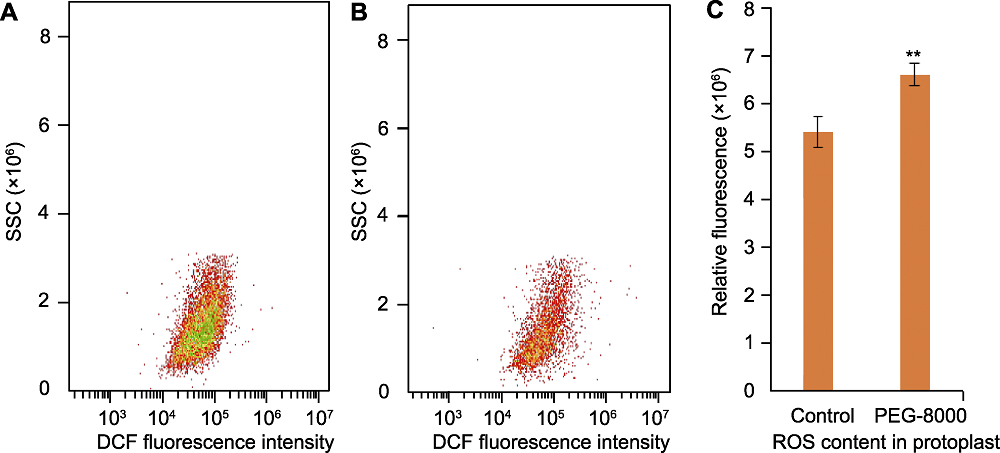
Figure 1 Reactive oxygen species (ROS) evaluation in rice protoplasts using 2′,7′-dichlorofluorescein (DCF)-based flow cytometry (A) ROS fluorescence intensity in rice leaves under normal growth conditions; (B) ROS fluorescence intensity in rice leaves under PEG-8000 treatment conditions; (C) The relative fluorescence intensity in treatment and control groups was determined to assess the ROS content. n=3; ** indicates significant difference at P<0.01 level by Student’s t test. SSC: Side scatter
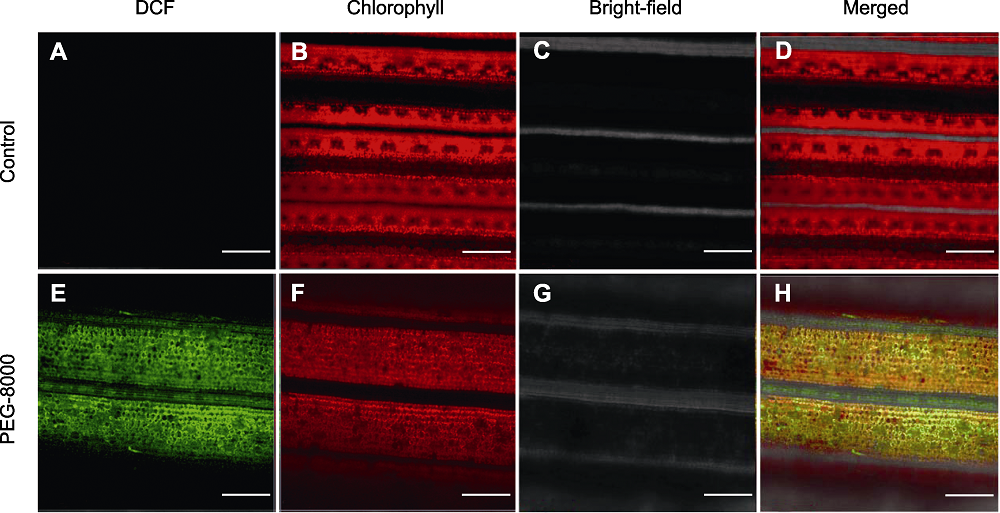
Figure 2 Confocal imaging analysis of rice leaves under normal growth (A)-(D) and PEG-8000 treatment (E)-(H) Red is the spontaneous fluorescence of chlorophyll, and green is the fluorescence of 2′,7′-dichlorofluorescein (DCF) generated by 2′,7′-dichlorodi-hydrofluorescein diacetate (H2DCFDA) oxidation. Bars=100 µm
| [1] |
Akter S, Khan MS, Smith EN, Flashman E (2021). Measuring ROS and redox markers in plant cells. RSC Chem Biol 2, 1384-1401.
DOI URL |
| [2] |
Anjum NA, Amreen N, Tantray AY, Khan NA, Ahmad A (2020). Reactive oxygen species detection-approaches in plants: insights into genetically encoded FRET-based sen- sors. J Biotechnol 308, 108-117.
DOI URL |
| [3] |
Apel K, Hirt H (2004). REACTIVE OXYGEN SPECIES: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55, 373-399.
DOI URL |
| [4] |
Castro B, Citterico M, Kimura S, Stevens DM, Wrzaczek M, Coaker G (2021). Stress-induced reactive oxygen species compartmentalization, perception and signaling. Nat Plants 7, 403-412.
DOI PMID |
| [5] | Chan ZL, Yokawa K, Kim WY, Song CP (2016). Editorial: ROS regulation during plant abiotic stress responses. Front Plant Sci 7, 1536. |
| [6] |
Choudhury FK, Rivero RM, Blumwald E, Mittler R (2017). Reactive oxygen species, abiotic stress and stress combination. Plant J 90, 856-867.
DOI URL |
| [7] |
Considine MJ, Foyer CH (2021a). Oxygen and reactive oxygen species-dependent regulation of plant growth and development. Plant Physiol 186, 79-92.
DOI URL |
| [8] |
Considine MJ, Foyer CH (2021b). Stress effects on the reactive oxygen species-dependent regulation of plant growth and development. J Exp Bot 72, 5795-5806.
DOI URL |
| [9] |
Duanghathaipornsuk S, Farrell EJ, Alba-Rubio AC, Zelenay P, Kim DS (2021). Detection technologies for reactive oxygen species: fluorescence and electrochemical methods and their applications. Biosensors 11, 30.
DOI URL |
| [10] |
Eruslanov E, Kusmartsev S (2010). Identification of ROS using oxidized DCFDA and flow-cytometry. Methods Mol Biol 594, 57-72.
DOI PMID |
| [11] |
Fichman Y, Miller G, Mittler R (2019). Whole-plant live imaging of reactive oxygen species. Mol Plant 12, 1203- 1210.
DOI PMID |
| [12] |
Gomes A, Fernandes E, Lima JLFC (2005). Fluorescence probes used for detection of reactive oxygen species. J Biochem Biophys Methods 65, 45-80.
PMID |
| [13] |
Hasanuzzaman M, Bhuyan MHMB, Zulfiqar F, Raza A, Mohsin SM, Mahmud JA, Fujita M, Fotopoulos V (2020). Reactive oxygen species and antioxidant defense in plants under abiotic stress: revisiting the crucial role of a universal defense regulator. Antioxidants 9, 681.
DOI URL |
| [14] |
Hu HT, Ren DY, Hu J, Jiang HZ, Chen P, Zeng DL, Qian Q, Guo LB (2021). WHITE AND LESION-MIMIC LEAF1, encoding a lumazine synthase, affects reactive oxygen species balance and chloroplast development in rice. Plant J 108, 1690-1703.
DOI URL |
| [15] |
Kristiansen KA, Jensen PE, Møller IM, Schulz A (2009). Monitoring reactive oxygen species formation and localisation in living cells by use of the fluorescent probe CM- H2DCFDA and confocal laser microscopy. Physiol Plant 136, 369-383.
DOI PMID |
| [16] |
Li HX, Liu Y, Qin HH, Lin XL, Tang D, Wu ZJ, Luo W, Shen Y, Dong FQ, Wang YL, Feng TT, Wang LL, Li LY, Chen DD, Zhang Y, Murray JD, Chao DY, Chong K, Cheng ZK, Meng Z (2020). A rice chloroplast-localized ABC transporter ARG1 modulates cobalt and nickel homeostasis and contributes to photosynthetic capacity. New Phytol 228, 163-178.
DOI URL |
| [17] |
Liu XY, Zhang ZG (2022). A double-edged sword: reactive oxygen species (ROS) during the rice blast fungus and host interaction. FEBS J doi:10.1111/febs.16171
DOI |
| [18] |
Maulucci G, Bačić G, Bridal L, Schmidt HH, Tavitian B, Viel T, Utsumi H, Yalçın AS, De Spirito M (2016). Imaging reactive oxygen species-induced modifications in living systems. Antioxid Redox Signal 24, 939-958.
DOI URL |
| [19] | Mhamdi A, van Breusegem F (2018). Reactive oxygen species in plant development. Development 145, dev164376. |
| [20] |
Oparka M, Walczak J, Malinska D, van Oppen LMPE, Szczepanowska J, Koopman WJH, Wieckowski MR (2016). Quantifying ROS levels using CM-H2DCFDA and HyPer. Methods 109, 3-11.
DOI URL |
| [21] |
Ortega-Villasante C, Burén S, Barón-Sola Á, Martínez F, Hernández LE (2016). In vivo ROS and redox potential fluorescent detection in plants: present approaches and future perspectives. Methods 109, 92-104.
DOI PMID |
| [22] |
Ortega-Villasante C, Burén S, Blázquez-Castro A, Barón- Sola Á, Hernández LE (2018). Fluorescent in vivo imaging of reactive oxygen species and redox potential in plants. Free Radic Biol Med 122, 202-220.
DOI URL |
| [23] |
Qi JS, Song CP, Wang BS, Zhou JM, Kangasjärvi J, Zhu JK, Gong ZZ (2018). Reactive oxygen species signaling and stomatal movement in plant responses to drought stress and pathogen attack. J Integr Plant Biol 60, 805- 826.
DOI URL |
| [24] |
Rajneesh, Pathak J, Chatterjee A, Singh SP, Sinha RP (2017). Detection of reactive oxygen species (ROS) in cyanobacteria using the oxidant-sensing probe 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA). Bio Protoc 7, e2545.
DOI PMID |
| [25] | Robles V, Riesco MF, Martínez-Vázquez JM, Valcarce DG (2021). Flow cytometry and confocal microscopy for ROS evaluation in fish and human spermatozoa. Methods Mol Biol 2202, 93-102. |
| [26] |
Tripathy BC, Oelmüller R (2012). Reactive oxygen species generation and signaling in plants. Plant Signal Behav 7, 1621-1633.
DOI URL |
| [27] |
Waszczak C, Carmody M, Kangasjärvi J (2018). Reactive oxygen species in plant signaling. Annu Rev Plant Biol 69, 209-236.
DOI PMID |
| [28] |
Xia SS, Liu H, Cui YJ, Yu HP, Rao YC, Yan YP, Zeng DL, Hu J, Zhang GH, Gao ZY, Zhu L, Shen L, Zhang Q, Li Q, Dong GJ, Guo LB, Qian Q, Ren DY (2022). UDP-N-acetylglucosamine pyrophosphorylase enhances rice survival at high temperature. New Phytol 233, 344- 359.
DOI URL |
| [29] |
Xiong HY, Yu JP, Miao JL, Li JJ, Zhang HL, Wang X, Liu PL, Zhao Y, Jiang CH, Yin ZG, Li Y, Guo Y, Fu BY, Wang WS, Li ZK, Ali J, Li ZC (2018). Natural variation in OsLG3 increases drought tolerance in rice by inducing ROS scavenging. Plant Physiol 178, 451-467.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||