

内源激素对高粱主茎与分蘖株高差异的影响
收稿日期: 2025-04-08
录用日期: 2025-07-01
网络出版日期: 2025-07-07
基金资助
国家自然科学基金(32272164);山西省回国留学人员科研资助项目(2024-071);山西省博士研究生、博士后研究人员来晋工作奖励经费科研项目(SXBYKY2024118);山西省现代农业产业技术体系建设专项(2025CYJSTX10-5)
Effects of Endogenous Hormones on Height Difference Between Main Stem and Tiller of Sorghum bicolor
Received date: 2025-04-08
Accepted date: 2025-07-01
Online published: 2025-07-07
分蘖高于主茎是影响高粱(Sorghum bicolor)品种整齐度和机械化生产的重要因素之一, 内源激素及其互作效应在植物株高建成过程中发挥关键调控作用。为探索内源激素对高粱主茎和分蘖株高差异的影响, 以主茎和分蘖高度一致的高粱品系K35-Y5以及分蘖高于主茎的高粱品系1383为材料, 测定了4个时期(主茎孕穗期、分蘖孕穗期、主茎开花期和分蘖开花期)主茎与分蘖的株高性状和内源激素含量, 并分析了二者的变化特征及其相关性。不同时期主茎与分蘖株高差异变化分析发现, 在前3个时期K35-Y5的主茎均高于分蘖, 至分蘖开花期时主茎与分蘖株高基本一致; 在主茎孕穗期, 1383的主茎高于分蘖, 分蘖孕穗期时主茎与分蘖株高基本一致, 后2个时期则分蘖高于主茎。不同时期内源激素含量变化特征分析发现, GA3含量在主茎和分蘖中的变化趋势与株高基本一致; ABA含量在K35-Y5主茎和分蘖间变化趋势基本一致, 主茎开花期时1383的分蘖明显高于主茎。IAA、ICA、tZR、IPA、Dx和JA含量随着生育时期总体呈下降趋势; ACC、SA、JA和H2JA无明显变化规律。相关性分析表明, 分蘖和主茎的株高差异在分蘖孕穗期、主茎开花期和分蘖开花期, 与GA3相对含量呈显著正相关; 且在主茎开花期和分蘖开花期, 与GA3/ABA相对比值呈显著正相关。综上所述, 在分蘖孕穗期、主茎开花期和分蘖开花期, GA3相对含量与主茎和分蘖株高差异密切相关。通过GA3处理和石蜡切片, 我们发现外施GA3影响细胞的伸长, 可实现对株高整齐一致株型的改良, 对于选育适宜机械化生产的高粱品种具有重要意义。

王瑞 , 赵威军 , 白洋 , 程庆军 , 张海燕 , 闫凤霞 , 凌亮 . 内源激素对高粱主茎与分蘖株高差异的影响[J]. 植物学报, 2025 , 60(6) : 901 -913 . DOI: 10.11983/CBB25060
INTRODUCTION: Tiller height higher than main stem is one of the important factors affecting the uniformity and mechanized production of sorghum. Sorghum with tiller height equal to that of the main stem can be densely planted and harvested mechanically. Endogenous hormones play an important regulatory role in the process of plant height development. Understanding the impact of endogenous hormones on the height difference between main stem and tiller in sorghum is of great significance for the improvement of sorghum varieties with tiller higher than the main stem.
RATIONALE: To further explore the effects of endogenous hormones on height difference between main stem and tiller of sorghum, Sorghum K35-Y5 with the similar height of main stem and tiller and 1383 with higher tiller than main stem were used to measure the plant height and endogenous hormone contents in main stem booting stage, tiller booting stage, main stem flowering stage and tiller flowering stage, and analyze the dynamic change and correlation, laying a foundation for studying the regulatory mechanisms of main stem and tiller height in sorghum and for improving plant architecture.
RESULTS: The height difference of main stem and tiller in different stages showed that the main stem of K35-Y5 was higher than that of tiller in the first three periods, and the height of main stem and tiller was basically the same at tiller flowering stage. The main stem height of 1383 was higher than that of tiller in main stem booting stage, the height of main stem and tiller was basically the same in tiller booting stage, and the height of tiller was higher than that of main stem in the latter two stages. Analysis of dynamic changes of endogenous hormone in different stages showed that GA3 content in main stem and tiller was basically consistent with the change of plant height. The dynamic change of ABA in K35-Y5 main stem and tiller is basically the same, which was significantly higher than main stem of 1383 tiller in main stem flowering stage. The contents of IAA, ICA, tZR, IPA, Dx and JA showed a decreasing trend. ACC, SA, JA and H2JA had no obvious characteristics. The correlation analysis showed that the difference of tiller and main stem was positively correlated with the relative content of GA3 at tiller booting stage, main stem flowering stage and tiller flowering stage, and positively correlated with the relative ratio of GA3/ABA at main stem flowering stage and tiller flowering stage.
CONCLUSION: The height difference of main stem and tiller was closely related to the relative content of GA3 at tiller booting stage, main stem flowering stage and tiller flowering stage. Through the treatment of GA3 and subsequent paraffin section analysis, we found that spraying GA3 affects cell elongation to achieve the improvement of plant height uniformity. This will be of great significance for the breeding of sorghum varieties suitable for mechanized production.
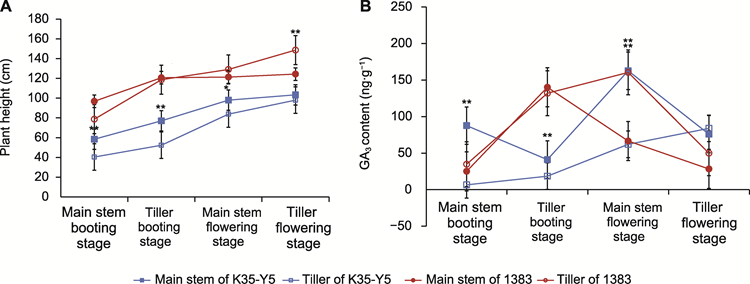
Differences in growth, height (A), and the GA3 contents (B) of main stem and tiller of K35-Y5 and 1383 at different stages. * and ** indicate significant differences at the 0.05 and 0.01 levels, respectively.

Key words: Sorghum bicolor; main stem; tiller; height difference; endogenous hormones
| [1] | Aeschbacher RA, Hauser MT, Feldmann KA, Benfey PN (1995). The SABRE gene is required for normal cell expansion in Arabidopsis. Genes Dev 9, 330-340. |
| [2] | Agehara S, Leskovar DI (2014). Age-dependent effectiveness of exogenous abscisic acid in height control of bell pepper and jalape?o transplants. Sci Hortic 175, 193-200. |
| [3] | Bai YC, Cai MM, Dou YP, Xie YL, Zheng HF, Gao J (2023). Phytohormone crosstalk of cytokinin biosynthesis and signaling family genes in moso bamboo (Phyllostachys edulis). Int J Mol Sci 24, 10863. |
| [4] | Barbier FF, Dun EA, Kerr SC, Chabikwa TG, Beveridge CA (2019). An update on the signals controlling shoot branching. Trends Plant Sci 24, 220-236. |
| [5] | Chen PL, Yang RX, Bartels D, Dong TY, Duan HY (2022). Roles of abscisic acid and gibberellins in stem/root tuber development. Int J Mol Sci 23, 4955. |
| [6] | Chen XS, Di JC, Xu NY, Xiao SH, Liu JG (2007). The inheritance of an ultra-dwarf plant mutant from upland cotton. Hereditas 29, 471-474. (in Chinese). |
| 陈旭升, 狄佳春, 许乃银, 肖松华, 刘剑光 (2007). 陆地棉超矮秆突变性状质量遗传规律分析. 遗传 29, 471-474. | |
| [7] | Chen Y, Hou MM, Liu LJ, Wu S, Shen Y, Ishiyama K, Kobayashi M, Mccarty DR, Tan BC (2014). The maize DWARF1 encodes a gibberellin 3-oxidase and is dual localized to the nucleus and cytosol. Plant Physiol 166, 2028-2039. |
| [8] | Chen YN, Fan XR, Song WJ, Zhang YL, Xu GH (2012). Over-expression of OsPIN2 leads to increased tiller numbers, angle and shorter plant height through suppression of OsLAZY1. Plant Biotechnol J 10, 139-149. |
| [9] | Davies PJ (2012). Plant hormones and their role in plant growth and development. Dordrecht: Springer. pp. 3-10. |
| [10] | Domagalska MA, Leyser O (2011). Signal integration in the control of shoot branching. Nat Rev Mol Cell Biol 12, 211-221. |
| [11] | Du H, Chang Y, Huang F, Xiong LZ (2015). GID1 modulates stomatal response and submergence tolerance involving abscisic acid and gibberellic acid signaling in rice. J Integr Plant Biol 57, 954-968. |
| [12] | Han Y, Teng KC, Nawaz G, Feng X, Usman B, Wang X, Luo L, Zhao N, Liu YG, Li RB (2019). Generation of semi-dwarf rice (Oryza sativa L.) lines by CRISPR/Cas9- directed mutagenesis of OsGA20ox2 and proteomic analysis of unveiled changes caused by mutations. 3 Biotech 9, 387. |
| [13] | Huang DB, Wang SG, Zhang BC, Shang-Guan KK, Shi YY, Zhang DM, Liu XL, Wu K, Xu ZP, Fu XD, Zhou YH (2015). A gibberellin-mediated DELLA-NAC signaling cascade regulates cellulosesynthesis in rice. Plant Cell 27, 1681-1696. |
| [14] | Hubbard L, McSteen P, Doebley J, Hake S (2002). Expression patterns and mutant phenotype of teosinte branched1 correlate with growth suppression in maize and teosinte. Genetics 162, 1927-1935. |
| [15] | Ji SH, Gururani MA, Lee JW, Ahn BO, Chun SC (2014). Isolation and characterisation of a dwarf rice mutant exhibiting defective gibberellins biosynthesis. Plant Biol 16, 428-439. |
| [16] | Jia DF, Gong XQ, Li MJ, Li C, Sun TT, Ma FW (2018). Overexpression of a novel apple NAC transcription factor gene, MdNAC1, confers the dwarf phenotype in transgenic apple (Malus domestica). Genes 9, 229. |
| [17] | Kn?ller AS, Blakeslee JJ, Richards EL, Peer WA, Murphy AS (2010). Brachytic2/ZmABCB1 functions in IAA export from intercalary meristems. J Exp Bot 61, 3689-3696. |
| [18] | Langer RHM, Prasad PC, Laude HM (1973). Effects of kinetin on tiller bud elongation in wheat (Triticum aestivum L.). Ann Bot 37, 565-571. |
| [19] | Li ZX, Zhang XR, Zhao YJ, Li YJ, Zhang GF, Peng ZH, Zhang JR (2018). Enhancing auxin accumulation in maize root tips improves root growth and dwarfs plant height. Plant Biotechnol J 16, 86-99. |
| [20] | Liu T, Wang TH, Chun Y, Li XY, Zhao JF (2022). Research progresses on epigenetic regulation of plant branching/ tillering. Chin Bull Bot 57, 532-548. (in Chinese). |
| 刘婷, 王天浩, 淳雁, 李学勇, 赵金凤 (2022). 表观遗传调控植物分枝/分蘖研究进展. 植物学报 57, 532-548. | |
| [21] | Liu Y, Ding YF, Wang QS, Li GH, Xu JX, Liu ZH, Wang SH (2011). Effect of plant growth regulators on the growth of rice tiller bud and the changes of endogenous hormones. Acta Agronomica Sinica 37, 670-676. (in Chinese). |
| 刘杨, 丁艳锋, 王强盛, 李刚华, 许俊旭, 刘正辉, 王绍华 (2011). 植物生长调节剂对水稻分蘖芽生长和内源激素变化的调控效应. 作物学报 37, 670-676. | |
| [22] | Nakano M, Omae N, Tsuda K (2022). Inter-organismal phytohormone networks in plant-microbe interactions. Curr Opin Plant Biol 68, 102258. |
| [23] | Nakata M, Mitsuda N, Herde M, Koo AJK, Moreno JE, Suzuki K, Howe GA, Ohme-Takagi M (2013). A bHLH- type transcription factor, ABA-INDUCIBLE BHLH-TYPE TRANSCRIPTION FACTOR/JA-ASSOCIATED MYC2-LIKE1, acts as a repressor to negatively regulate jasmonate signaling in Arabidopsis. Plant Cell 25, 1641-1656. |
| [24] | Rongen MV, Bennett T, Ticchiarelli F, Leyser O (2019). Connective auxin transport contributes to strigolactone- mediated shoot branching control independent of the transcription factor BRC1. PLoS Genet 15, 1008023. |
| [25] | Shen JJ, Zhang YQ, Ge DF, Wang ZY, Song WY, Gu R, Che G, Cheng ZH, Liu RY, Zhang XL (2019). CsBRC1 inhibits axillary bud outgrowth by directly repressing the auxin efflux carrier CsPiN3 in cucumber. Proc Natl Acad Sci USA 116, 17105-17114. |
| [26] | Srinivasan C, Liu ZR, Scorza R (2011). Ectopic expression of class 1 KNOX genes induce adventitious shoot regeneration and alter growth and development of tobacco (Nicotiana tabacum L.) and European plum (Prunus domestica L.) Plant Cell Rep 30, 655-664. |
| [27] | Tong HN, Xiao YH, Liu DP, Gao SP, Liu LC, Yin YH, Jin Y, Qian Q, Chu CC (2014). Brassinosteroid regulates cell elongation by modulating gibberellin metabolism in rice. Plant Cell 26, 4376-4393. |
| [28] | Waldie T, Leyser O (2018). Cytokinin targets auxin transport to promote shoot branching. Plant Physiol 177, 803-818. |
| [29] | Wang CZ, Pan XJ, Zhang CM, Hu XF, Yang YX (2006). Effects of exogenous ABA on hormone content in different varieties of fall dormancy Medicago sativa varieties. Acta Prataculturae Sinica 15(2), 30-36. (in Chinese). |
| 王成章, 潘晓建, 张春梅, 胡喜峰, 杨雨鑫 (2006). 外源ABA对不同秋眠型苜蓿品种植物激素含量的影响. 草业学报 15(2), 30-36. | |
| [30] | Wang L, Mu C, Du MW, Chen Y, Tian XL, Zhang MC, Li ZH (2014). The effect of mepiquat chloride on elongation of cotton (Gossypium hirsutum L.) internode is associated with low concentration of gibberellic acid. Plant Sci 225, 15-23. |
| [31] | Xia XJ, Dong H, Yin YL, Song XW, Gu XH, Sang KQ, Zhou J, Shi K, Zhou YH, Foyer CH, Yu JQ (2021). Brassinosteroid signaling integrates multiple pathways to release apical dominance in tomato. Proc Natl Acad Sci USA 118, 2004384118. |
| [32] | Yamaguchi S (2008). Gibberellin metabolism and its regulation. Annu Rev Plant Biol 59, 225-251. |
| [33] | Yang TZ, Yang XL, Yin QY, Guo ZM (2006). Changes in endogenous hormone contents in shoot-tip of tobacco (Nicotiana tabacum L.) genotypes with different plant height and response to the exogenous hormones. Plant Physiology Journal 42, 643-647. (in Chinese). |
| 杨铁钊, 杨欣玲, 殷全玉, 郭志民 (2006). 烟草株高变异体的茎尖中内源激素含量变化及其对外源激素的响应. 植物生理学通讯 42, 643-647. | |
| [34] | Yin WN, Kong GC, Wang XY, Gao JT, He X (2011). Dynamic changes of plant hormone contents in hexapoid triticale (× Triticosecale Wittmack) with different plant height. Journal of Triticeae Crops 31, 953-958. (in Chinese). |
| 殷稳娜, 孔广超, 王雪玉, 高静涛, 何萱 (2011). 六倍体小黑麦株高形成中内源激素含量的变化. 麦类作物学报 31, 953-958. | |
| [35] | Zeevaart JAD (1983). Metabolism of abscisic acid and its regulation in Xanthium leaves during and after water stress. Plant Physiol 71, 477-481. |
| [36] | Zhang YL, Wang LY, Liang YT, Chen N, Li SF, Gao WJ (2024). GA3 treatment affects sex expression and plant architecture in spinach. Journal of Henan Normal University (Nat Sci Ed) 52(2), 130-138. (in Chinese). |
| 张玉兰, 王丽颖, 梁艺涛, 陈宁, 李书粉, 高武军 (2024). GA3处理影响菠菜性别及其株型发育. 河南师范大学学报(自然科学版) 52(2), 130-138. | |
| [37] | Zhou SG, Hu ZL, Li FF, Yu XH, Naeem M, Zhang YJ, Chen GP (2018). Manipulation of plant architecture and flowering time by down-regulation of the GRAS transcription factor SlGRAS26 in Solanum lycopersicum. Plant Sci 271, 81-93. |
/
| 〈 |
|
〉 |