

不同钾水平下烟草根内皮层木栓化的适应性发育差异
收稿日期: 2024-12-09
录用日期: 2024-03-18
网络出版日期: 2025-03-18
基金资助
中国烟草总公司山东省公司科技重大专项(202417);山东省自然科学基金(ZR2023MC150);中国农业科学院科技创新工程(ASTIP-TRIC03)
Differences in the Adaptive Development of Suberin in the Tobacco Root Endothelial Layer under Different Potassium Levels
Received date: 2024-12-09
Accepted date: 2024-03-18
Online published: 2025-03-18
根内皮层分化的质外体屏障在植物抗逆和养分吸收过程中均发挥重要作用, 其中木栓层发育为近年的研究热点。以烟草(Nicotiana tabacum)栽培品种中烟100为材料, 通过0.1-4.0 mmol∙L-1钾浓度梯度水培实验, 探讨供钾水平对根内皮层木栓化发育的影响及其生理和分子机制。结果表明, 低钾胁迫(0.1 mmol∙L-1)显著增强内皮层木栓化: 完全木栓化区域绝对长度由对照的0-2 cm延长至4-6 cm, 相对占比从0-15.0%提升至33.2%-44.3%, 表明木栓化是烟草响应低钾胁迫的关键形态适应机制之一。表型分析显示, 低钾胁迫下植株根系伸长但生物量下降, 地上部与根系钾离子含量及积累量均减少, 木质部伤流液流量及钾离子运输效率降低。内源激素含量检测发现, 低钾胁迫提高根系内源脱落酸含量, 并降低乙烯和茉莉酸甲酯含量, 形成特异性激素调控网络。转录组数据进一步佐证木栓化发育的分子基础, 木栓质合成与转运相关基因(如CYP86、GPAT和ABCG)及其上游正调控基因MYB36/41/92/93显著上调表达。综上, 该研究阐明了低钾胁迫下烟草通过脱落酸介导的激素信号调控木栓化发育程序, 为解析作物钾胁迫适应机制提供了全新视角。
许耘祥 , 张莉汶 , 王朋 , 顾迎晨 , 张标 , 朱莹莹 , 刘海伟 . 不同钾水平下烟草根内皮层木栓化的适应性发育差异[J]. 植物学报, 2025 , 60(6) : 914 -930 . DOI: 10.11983/CBB24191
INTRODUCTION: Apoplastic barriers differentiated from the root endoderm play important roles in plant stress resistance and nutrient uptake, and the development of suberin lamellae has become a popular research topic in recent years.
RATIONALE: Hydroponic experiments with potassium concentrations ranging from 0.1 to 4.0 mmol∙L-1 were conducted, using the cultivated tobacco variety Zhongyan 100 as the experimental material, to explore the effects of different potassium supplies on the endodermal suberization and its physiological and molecular mechanisms.
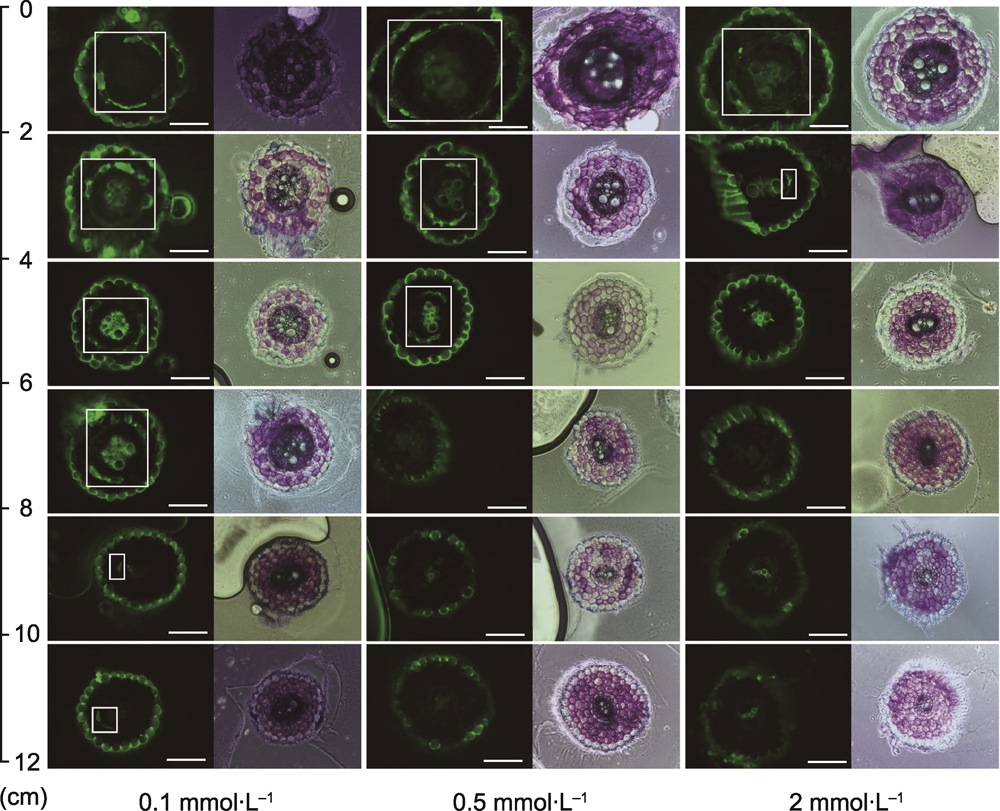
RESULTS: Low potassium (0.1 mmol∙L-1) stress significantly enhanced the endodermal suberization: the absolute length of the fully suberized region ranged from 0-2 cm in the control to 4-6 cm, and the relative proportion increased from 0-15.0% to 33.2%-44.3%. These findings indicate that suberization is one of the key morphological adaptation mechanisms in tobacco under low potassium stress. Phenotypic analysis revealed that under low potassium stress, root elongation increased while plant biomass decreased, and the potassium ion content and accumulation in both the aboveground and root parts decreased. Additionally, the flow rate and potassium ion concentration in the xylem sap decreased, indicating reduced transport efficiency. Endogenous hormone analysis revealed that low potassium stress increased the abscisic acid (ABA) content in roots while suppressing ethylene and methyl jasmonate levels, forming a specific hormonal regulatory network. The transcriptome data further supported the molecular basis of suberization development, showing significant upregulation of genes related to suberin synthesis and transport (e.g., CYP86, GPAT, and ABCG) and their upstream positive regulatory factors (MYB36, MYB41, MYB92, and MYB93).
CONCLUSION: This study is the first to reveal that low potassium stress regulates the suberization developmental program of tobacco roots through ABA-mediated hormonal signaling reprogramming, providing a novel perspective for understanding the adaptation mechanisms of plants to potassium stress.
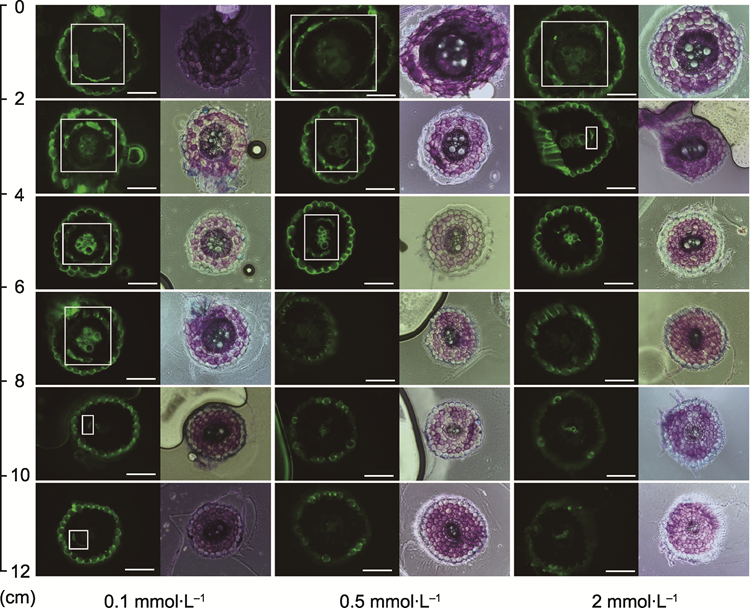
Fluorescence imaging of transverse sections of the endodermis development in tobacco roots under three potassium concentrations (bars=130 μm)

Key words: suberization; potassium; root endodermis; hormone
| [1] | Andersen TG, Barberon M, Geldner N (2015). Suberization—the second life of an endodermal cell. Curr Opin Plant Biol 28, 9-15. |
| [2] | Barberon M (2017). The endodermis as a checkpoint for nutrients. New Phytol 213, 1604-1610. |
| [3] | Barberon M, Vermeer JEM, De Bellis D, Wang P, Naseer S, Andersen TG, Humbel BM, Nawrath C, Takano J, Salt DE, Geldner N (2016). Adaptation of root function by nutrient-induced plasticity of endodermal differentiation. Cell 164, 447-459. |
| [4] | Baxter I, Hosmani PS, Rus A, Lahner B, Borevitz JO, Muthukumar B, Mickelbart MV, Schreiber L, Franke RB, Salt DE (2009). Root suberin forms an extracellular barrier that affects water relations and mineral nutrition in Arabidopsis. PLoS Genet 5, e1000492. |
| [5] | Chen AL, Liu T, Deng Y, Xiao R, Zhang T, Wang Y, Yang YH, Lakshmanan P, Shi XJ, Zhang FS, Chen XP (2023). Nitrate-dependent suberization regulates cadmium uptake and accumulation in maize. Sci Total Environ 878, 162-848. |
| [6] | Doblas VG, Geldner N, Barberon M (2017). The endodermis, a tightly controlled barrier for nutrients. Curr Opin Plant Biol 39, 136-143. |
| [7] | Guo Z, Li ZS, Dai XY, Wang YF (2019). Effects of auxin on tobacco root growth and potassium uptake under low potassium stress. Plant Nutr Fert Sci 25, 1173-1184. (in Chinese) |
| 郭泽, 李子绅, 代晓燕, 王英锋 (2019). 低钾胁迫下外源生长素对烟草根系生长及钾吸收的影响. 植物营养与肥料学报 25, 1173-1184. | |
| [8] | Leide J, Hildebrandt U, Hartung W, Riederer M, Vogg G (2012). Abscisic acid mediates the formation of a suberized stem scar tissue in tomato fruits. New Phytol 194, 402-415. |
| [9] | Liu HW, Zhang Y, Wang HY, Zhang B, He Y, Wang HH, Zhu YY, Holm PE, Shi Y (2023). Comparing cadmium uptake kinetics, xylem translocation, chemical forms, and subcellular distribution of two tobacco (Nicotiana tabacum L.) cultivars. Ecotoxicol Environ Saf 254, 114738. |
| [10] | Liu YK, Lu M, Persson DP, Luo JP, Liang YC, Li TQ (2022). The involvement of nitric oxide and ethylene on the formation of endodermal barriers in response to Cd in hyperaccumulator Sedum alfredii. Environ Pollut 307, 11-9530. |
| [11] | Liu YK, Tao Q, Li JX, Guo XY, Luo JP, Jupa R, Liang YC, Li TQ (2021). Ethylene-mediated apoplastic barriers development involved in cadmium accumulation in root of hyperaccumulator Sedum alfredii. J Hazard Mater 403, 123729. |
| [12] | Lulai EC, Suttle JC, Pederson SM (2008). Regulatory involvement of abscisic acid in potato tuber wound-healing. J Exp Bot 59, 1175-1186. |
| [13] | Luo HB, Huang CM, Cao HQ, Jiang SL, Wu XJ, Ye LP, Wei YW (2022). Effects of different potassium levels on root development and endogenous hormone content for passion fruit tissue cultured seedling. China Fruits (4), 53-58. (in Chinese) |
| 罗海斌, 黄诚梅, 曹辉庆, 蒋胜理, 吴兴剑, 叶丽萍, 魏源文 (2022). 不同浓度钾元素对西番莲组培苗根系生长和内源激素含量的影响. 中国果树 (4), 53-58. | |
| [14] | Lux A, Morita S, Abe J, Ito K (2005). An improved method for clearing and staining free-hand sections and whole- mount samples. Ann Bot 96, 989-996. |
| [15] | Melino VJ, Plett DC, Bendre P, Thomsen HC, Zeisler- Diehl VV, Schreiber L, Kronzucker HJ (2021). Nitrogen depletion enhances endodermal suberization without restricting transporter-mediated root NO3- influx. J Plant Physiol 257, 153334. |
| [16] | Naseer S, Lee Y, Lapierre C, Franke R, Nawrath C, Geldner N (2012). Casparian strip diffusion barrier in Arabidopsis is made of a lignin polymer without suberin. Proc Natl Acad Sci USA 109, 10101-10106. |
| [17] | Pfister A, Barberon M, Alassimone J, Kalmbach L, Lee Y, Vermeer JE, Yamazaki M, Li GW, Maurel C, Takano J, Kamiya T, Salt DE, Roppolo D, Geldner N (2014). A receptor-like kinase mutant with absent endodermal diffusion barrier displays selective nutrient homeostasis defects. eLife 3, e03115. |
| [18] | Tao Q, Jupa R, Liu YK, Luo JP, Li JX, Ková? J, Li B, Li QQ, Wu KR, Liang YC, Lux A, Wang CQ, Li TQ (2019). Abscisic acid-mediated modifications of radial apoplastic transport pathway play a key role in cadmium uptake in hyperaccumulator Sedum alfredii. Plant Cell Environ 42, 1425-1440. |
| [19] | Tao Q, Li M, Xu Q, Ková? J, Yuan S, Li B, Li QQ, Huang R, Gao XS, Wang CQ (2022). Radial transport difference mediated by root endodermal barriers contributes to differential cadmium accumulation between japonica and indica subspecies of rice (Oryza sativa L.). J Hazard Mater 425, 128008. |
| [20] | Vestenaa MW, Husted S, Minutello F, Persson DP (2024). Endodermal suberin restricts root leakage of cesium: a suitable tracer for potassium. Physiol Plant 176, e14393. |
| [21] | Wang JB, Zhang QL, Tung J, Zhang X, Liu D, Deng YT, Tian ZD, Chen HL, Wang TT, Yin WX, Li B, Lai ZB, Dinesh-Kumar SP, Baker B, Li F (2024). High-quality assembled and annotated genomes of Nicotiana tabacum and Nicotiana benthamiana reveal chromosome evolution and changes in defense arsenals. Mol Plant 17, 423-437. |
| [22] | Wang LM, Liu YQ, Ruan YJ (2015). The research progress of potassium to plant. Chin Hortic Abstr 31(5), 71, 148. (in Chinese) |
| 王立梅, 刘奕清, 阮玉娟 (2015). 植物钾素研究进展. 中国园艺文摘 31(5), 71, 148. | |
| [23] | Wang P, Wang CM, Gao L, Cui YN, Yang HL, de Silva NDG, Ma Q, Bao AK, Flowers TJ, Rowland O, Wang SM (2020). Aliphatic suberin confers salt tolerance to Arabidopsis by limiting Na+ influx, K+ efflux and water backflow. Plant Soil 448, 603-620. |
| [24] | Wei XP, Liu LY, Jin XY, Xue J, Geng P, Xu ZH, Zhang LH, Wang XY, Zong W, Zhang L, Mao LC (2024a). Exogenous methyl jasmonate promotes wound healing of Chinese yam tubers (Dioscorea opposita) through the deposition of suberin polyaliphatics at the wound sites. Postharvest Biol Technol 207, 112586. |
| [25] | Wei XP, Liu LY, Liu G, Geng P, Wei XB, Yao X, Chen JY, Gong WJ, Ge ZZ, Liu MP, Mao LC (2024b). Methyl jasmonate promotes suberin biosynthesis by stimulating transcriptional activation of AchMYC2 on AchFHT in wound healing of kiwifruit. Postharvest Biol Technol 210, 112741. |
| [26] | Wei XP, Liu LY, Xu ZH, Xue J, Geng P, Ge ZZ, Wang XY, Zhang L, Zong W, Mao LC (2023). Methyl jasmonate facilitates wound healing of Chinese yam tubers via positively regulating the biosynthesis and polymerization of suberin polyphenolics. Sci Hortic 312, 111840. |
| [27] | Xu HM, Liu P, Wang CH, Wu SS, Dong CQ, Lin QY, Sun WR, Huang BB, Xu MZ, Tauqeer A, Wu S (2019). Transcriptional networks regulating suberin and lignin in endodermis link development and ABA response. Plant Physiol 190, 1165-1181. |
| [28] | Yan HF, Shi Y, Li NH, Zhang YC (2013). Progress in tobacco potassium nutrition. J Agric Sci Technol 15, 123-129. (in Chinese) |
| 闫慧峰, 石屹, 李乃会, 张永春 (2013). 烟草钾素营养研究进展. 中国农业科技导报 15, 123-129. | |
| [29] | Yang TZ, Shu HY, Zhao XZ (2002). Recent advances in tobacco potassium nutrition in China. Tob Sci Technol (7), 39-43. (in Chinese) |
| 杨铁钊, 舒海燕, 赵献章 (2002). 我国烟草钾素营养研究现状与进展. 烟草科技 (7), 39-43. | |
| [30] | Zhang B, Wu J, Zhang Y, Dong XW, Han S, Gao X, Du CW, Li HY, Chong XF, Zhu YY, Liu HW (2023). Research progress on physiological functions of suberin lamellae in water and solutes transport. Chin Bull Bot 58, 1008-1018. (in Chinese) |
| 张标, 吴健, 张杨, 董小卫, 韩硕, 高昕, 杜从伍, 李慧英, 种学法, 朱莹莹, 刘海伟 (2023). 木栓层在水和溶质运输中的生理功能研究进展. 植物学报 58, 1008-1018. | |
| [31] | Zhang B, Xu YX, Zhang LW, Yu SY, Zhu YY, Liu CJ, Wang P, Shi Y, Li LZ, Liu HW (2024). Root endodermal suberization induced by nitrate stress regulate apoplastic pathway rather than nitrate uptake in tobacco (Nicotiana tabacum L.). Plant Physiol Biochem 216, 109166. |
| [32] | Zhang B, Xu YX, Zhang LW, Zhu YY, Liu HW (2024). Mechanism of low NO3- stress inhibiting apoplastic transport in tobacco roots. Chin Tob Sci 45(2), 25-34. (in Chinese) |
| 张标, 许耘祥, 张莉汶, 朱莹莹, 刘海伟 (2024). 低NO3-胁迫抑制烟草根系质外体运输的机制研究. 中国烟草科学 45(2), 25-34. | |
| [33] | Zhou Y, An YP, Ma R, Wang P (2024). Transcriptional regulation of suberin and its response to the environment. Acta Bot Boreali-Occident Sin 44, 1993-2006. (in Chinese) |
| 周月, 安永平, 马蓉, 王沛(2024). 木栓质的转录调控及其对环境的响应. 西北植物学报 44, 1993-2006. |
/
| 〈 |
|
〉 |