

植物学报 ›› 2023, Vol. 58 ›› Issue (5): 712-719.DOI: 10.11983/CBB22117 cstr: 32102.14.CBB22117
史君星1, 闫一嘉2, 董汝2, 陶轩3, 孙晓龙4, 黄聪聪5,*( )
)
收稿日期:2022-06-07
接受日期:2022-11-15
出版日期:2023-09-01
发布日期:2023-09-21
通讯作者:
*E-mail: huangcong163@126.com
基金资助:
Shi Junxing1, Yan Yijia2, Dong Ru2, Tao Xuan3, Sun Xiaolong4, Huang Congcong5,*( )
)
Received:2022-06-07
Accepted:2022-11-15
Online:2023-09-01
Published:2023-09-21
Contact:
*E-mail: huangcong163@126.com
摘要: 几丁质是真菌细胞壁的主要成分, 同时也是微生物相关分子模式(MAMP), 通过定位于细胞膜上的模式识别受体CERK1和LYK5识别, 激发植物的先天防御反应。该研究克隆了CERK1的胞内激酶域, 在酵母文库中筛选到1个分子伴侣HSP1与CERK1胞内域互作。通过CRISPR-Cas介导的基因编辑技术, 我们在野生型拟南芥(Arabidopsis thaliana) Col-0中敲除HSP1基因, 获得hsp1 v63删除突变体。使用几丁质处理Col-0和hsp1 v63删除突变体后发现, 下游防御相关基因的表达及丝裂原活化蛋白激酶磷酸化通路受到抑制。此外, 我们还发现CERK1蛋白水平在hsp1 v63删除突变体中低于野生型, 并且该突变体中CERK1含量降低与内质网降解系统有关。以上结果表明, HSP1是几丁质激发防御反应通路中的1个关键基因。该研究揭示了分子伴侣调控膜受体蛋白含量对植物防御反应的重要意义, 为农作物广谱抗性遗传改良奠定了理论基础。
史君星, 闫一嘉, 董汝, 陶轩, 孙晓龙, 黄聪聪. 拟南芥HSP1调节CERK1蛋白水平影响几丁质激发的防御反应. 植物学报, 2023, 58(5): 712-719.
Shi Junxing, Yan Yijia, Dong Ru, Tao Xuan, Sun Xiaolong, Huang Congcong. The Arabidopsis HSP1 Mediates Chitin-induced Defense Response by Regulating CERK1 Protein Level. Chinese Bulletin of Botany, 2023, 58(5): 712-719.
| Primer name | Primer sequence (5′-3′) |
|---|---|
| ACTIN-F | TCCCTCAGCACATTCCAGCAGAT |
| ACTIN-R | AACGATTCCTGGACCTGCCTCATC |
| WRKY33-F | GGTCACAACAATCCGGAAGA |
| WRKY33-R | GGAGAGACAAGAGAAGGAGAGA |
| WRKY53-F | CGACGGCTGTTGCTGAGAC |
| WRKY53-R | TCCGTTTATCGATGCCGGAG |
表1 qRT-PCR引物序列
Table 1 The primers used for qRT-PCR
| Primer name | Primer sequence (5′-3′) |
|---|---|
| ACTIN-F | TCCCTCAGCACATTCCAGCAGAT |
| ACTIN-R | AACGATTCCTGGACCTGCCTCATC |
| WRKY33-F | GGTCACAACAATCCGGAAGA |
| WRKY33-R | GGAGAGACAAGAGAAGGAGAGA |
| WRKY53-F | CGACGGCTGTTGCTGAGAC |
| WRKY53-R | TCCGTTTATCGATGCCGGAG |
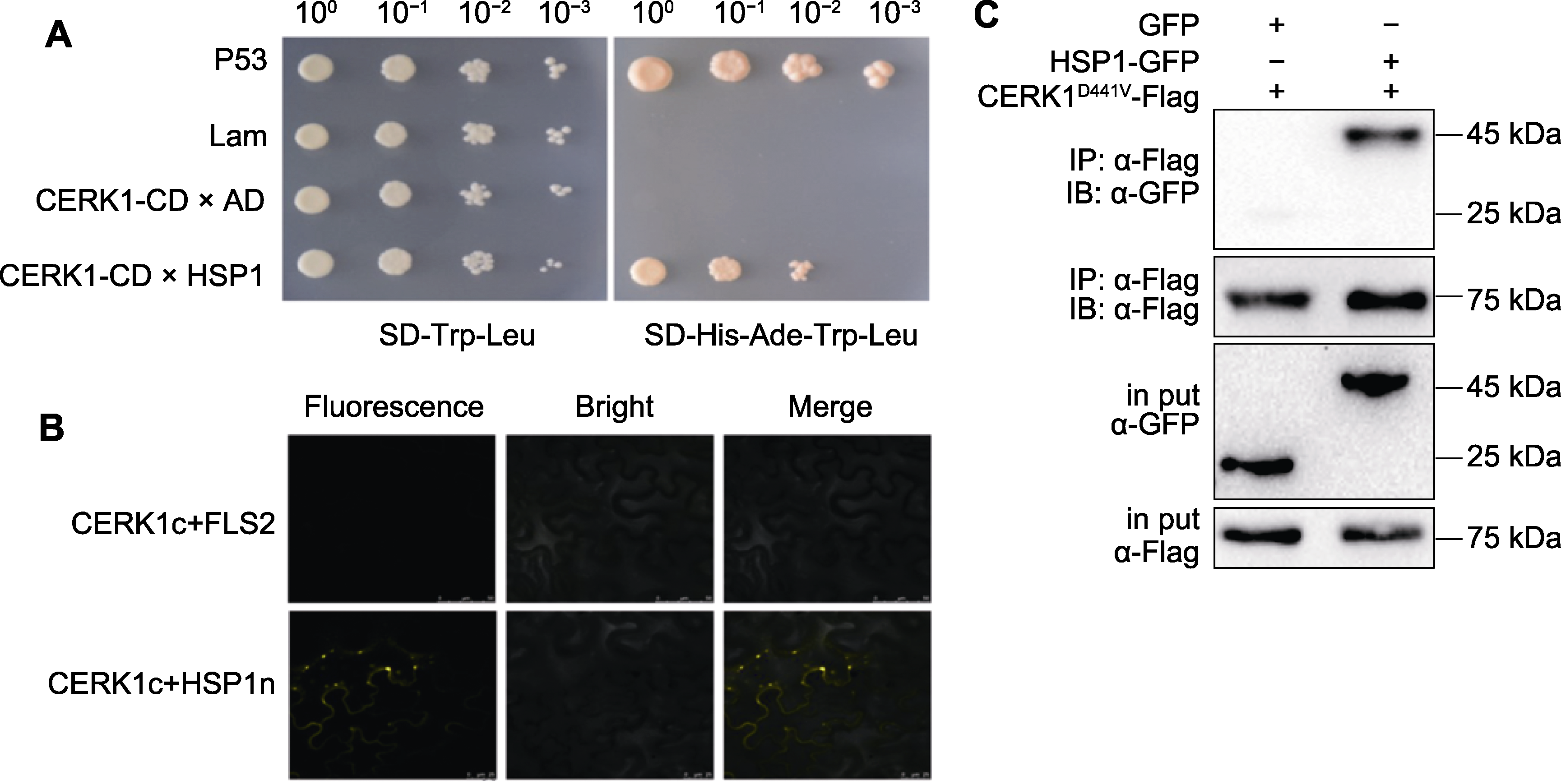
图1 CERK1-BD与HSP1-AD在酵母细胞中互作 (A) HSP1蛋白的N端融合AD, CERK1-CD的C端融合BD, 分别转化到Y187和AH109酵母菌株中。杂交后涂布在SD-Trp-Leu/ SD-Ade-Trp-Leu/SD-His-Ade-Trp-Leu平板上, 在温度28°C下培养, P53和Lam分别代表正对照和负对照; (B) 将CERK1的C端连接cYFP, HSP1和CPK5连接nYFP, 与CERK1-cYFP共注射到烟草叶肉细胞中分别作为实验组和阴性对照组, 共注射72小时后在荧光显微镜下检测YFP的荧光强度; (C) 采用免疫共沉淀实验检测HSP1与CERK1在烟草中的互作。将CERK1D441V的C端融合Flag标签, HSP1的C端融合GFP标签, 共注射到烟草叶肉细胞中, 使用Flag抗体沉淀CERK1蛋白, 通过CoIP检测HSP1-GFP。3次重复的结果相似。
Figure 1 Interaction between CERK1-BD and HSP1-AD in yeast cells (A) The HSP1 protein was fused with AD at the N-terminal, and CERK1-CD was fused with BD at the C-terminal, then these two recombinants were transformed into Y187 and AH109 yeast strains, respectively. After hybridization, it was plated on SD-Trp-Leu/SD-Ade-Trp-Leu/SD-His-Ade-Trp-Leu plates, and cultivated at 28°C, P53 and Lam represent positive and negative controls, respectively. (B) C-terminal of CERK1 was fused with cYFP, HSP1 and CPK5 were fused with nYFP, and then co-injected into Nicotiana benthamiana mesophyll cells, CPK5 and HSP1 served as negative control. After 72 hours of injection, the fluorescence intensity of YFP fluorescent protein was detected under fluorescence microscope; (C) Interaction between HSP1 and CERK1 was detected in N. benthamiana by Western blot (WB). C-terminal of CERK1D441V was fused with Flag tag, and C-terminal of HSP1 was fused with GFP tag, then were co-injected into N. benthamiana mesophyll cell. The CERK1 protein was precipitated using a Flag antibody, and the HSP1-GFP was detected by CoIP. The experiment was repeated for three times with similar results.
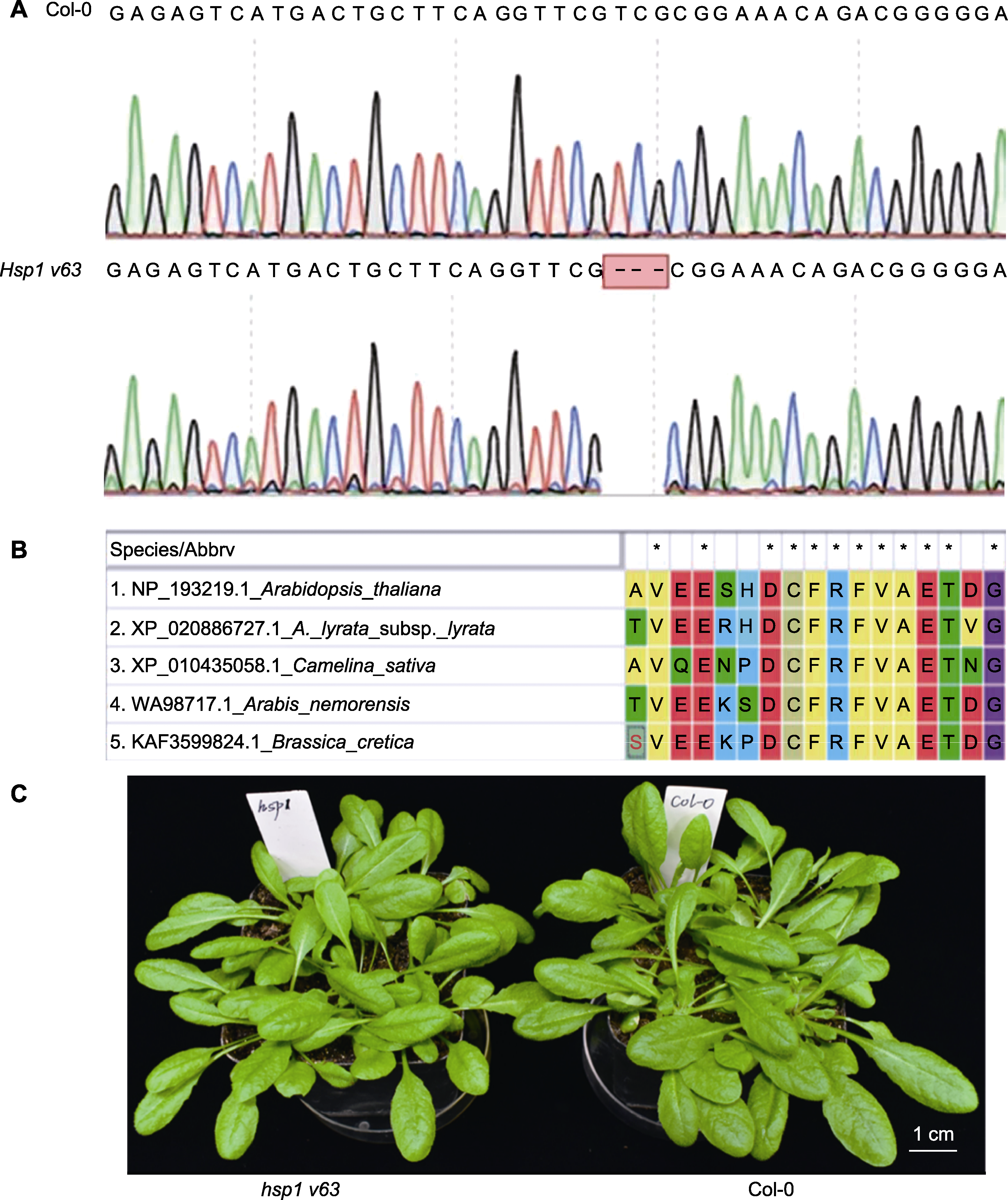
图2 拟南芥hsp1 v63删除突变体的鉴定 (A) 提取hsp1基因组DNA, 对HSP1基因进行扩增, PCR产物用于测序分析(通过测序结果比对, 发现HSP1第63位缬氨酸被敲除, 蓝色序列为gRNA的靶标位点, 红色序列表示NGG序列); (B) 使用Clustal W比对拟南芥(Arabidopsis thaliana)、深山南芥(A. lyrata)、亚麻芥(Camelina sativa)、筷子芥(Arabis)以及芸苔属(Brassica)中HSP1第63位氨基酸序列的保守性(*代表保守的氨基酸位点); (C) 生长4周的hsp1 v63删除突变体(左)和Col-0 (右), 二者生长状况类似。
Figure 2 Identification of hsp1 v63 deletion mutants in Arabidopsis thaliana (A) The hsp1 genomic DNA was extracted, then the HSP1 gene was amplified by PCR, and the PCR product was used for sequencing analysis (by comparing the sequencing results, it was found that the 63 rd valine of HSP1 was knocked out, the blue sequence represents the target site of gRNA, and the red sequence represents the NGG sequence; (B) Analysis of the conservation of the amino acid sequence of HSP1 in Arabidopsis thaliana, A. lyrata, Camelina sativa, Arabis and Brassica using Clustal W alignment (* represent the conserved amino acid sites); (C) The hsp1 v63 deletion mutant (left) and Col-0 (right) grown for 4 weeks, and their growth conditions were similar.
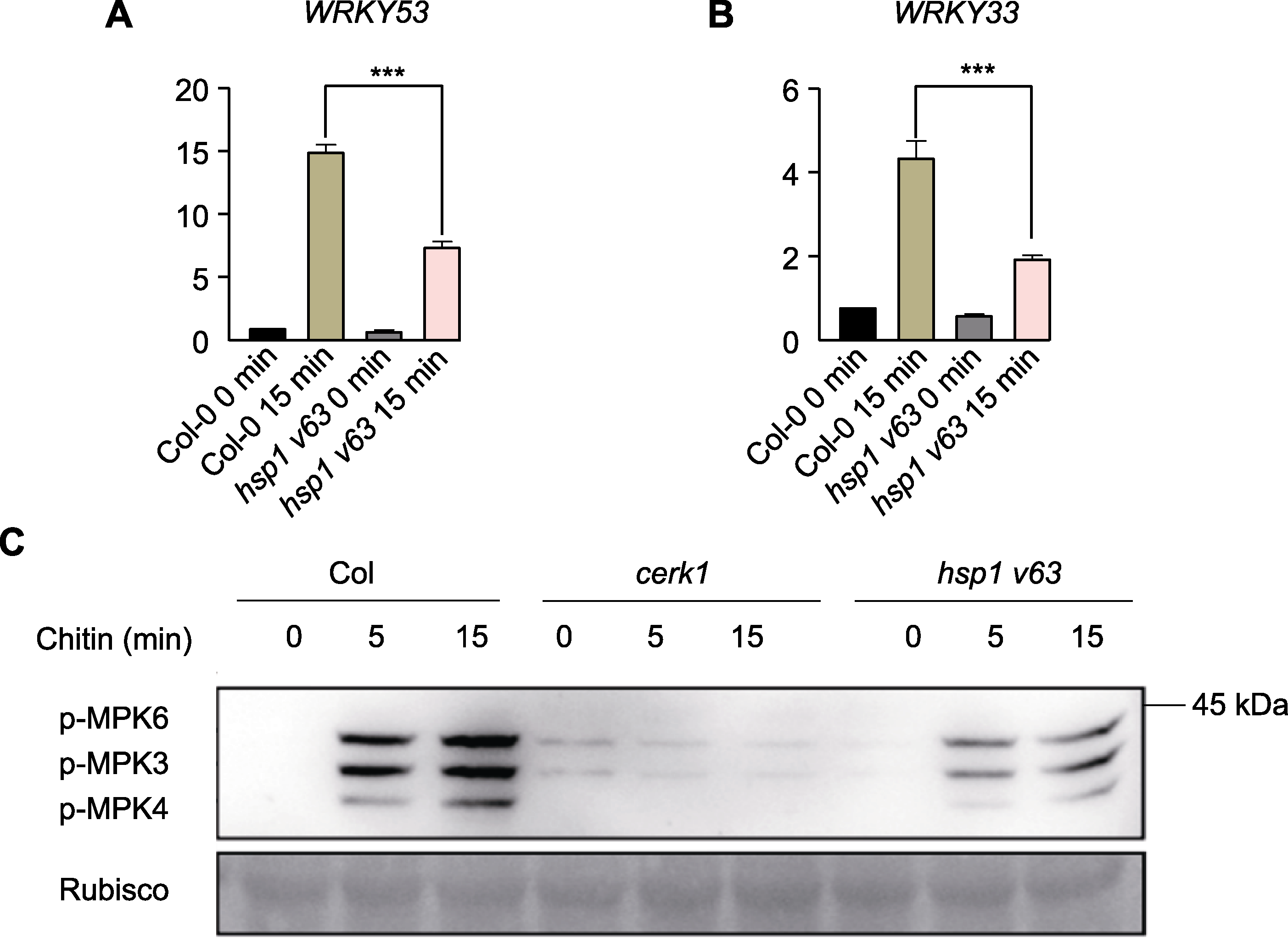
图3 拟南芥hsp1 v63删除突变体防御反应的测定 (A), (B) 几丁质诱导的拟南芥hsp1 v63删除突变体中WRKY53和WRKY33基因表达分析(通过qRT-PCR实验检测几丁质诱导前后hsp1 v63删除突变体和Col-0植株中WRKY53和WRKY33基因的表达水平。使用内参基因ACTIN2标准化所有表达水平, 误差线表示平均值±标准差, 单因素方差分析, *** P<0.001, n=3); (C) 在拟南芥cerk1、hsp1 v63删除突变体及野生型Col-0中, 几丁质诱导的MAPK激活实验(检测cerk1、hsp1 v63删除突变体和野生型(Col-0)植株中几丁质诱导的MAPK磷酸化水平。用几丁质按图中所示时间处理拟南芥幼苗, 收样后采用蛋白免疫印迹实验检测MAPK3/6的磷酸化水平。该实验所用抗体为Anti-P44/P42。3次重复, 结果相似)。
Figure 3 Determination of defense responses in the hsp1 v63 deletion mutant of Arabidopsis thaliana (A), (B) Chitin-induced gene expression analysis of WRKY53 and WRKY33 in Arabidopsis hsp1 v63 deletion mutant (the tran-scriptional level of WRKY53 and WRKY33 genes in hsp1 v63 deletion mutant and Col-0 seedlings was measured by qRT-PCR before and after chitin induction. All expression levels were normalized using the internal reference gene ACTIN2, data are pre-sented as means±SD, one-way ANOVA, Tukey post-test, *** P<0.001, n=3); (C) Chitin-induced MAPK activation experiments in Arabidopsis cerk1, hsp1 v63 deletion mutant, wild-type Col-0 (chitin-induced MAPK phosphorylation was examined in cerk1, hsp1 v63 deletion mutant and wild-type Col-0. The A. thaliana seedlings were treated with chitin for the time indicated in the figure. After the samples were collected, the phosphorylation of MAPK3/6 was detected by immunoblot. The antibody used in this experiment was Anti-P44/P42. The experiment was repeated three times with similar results).
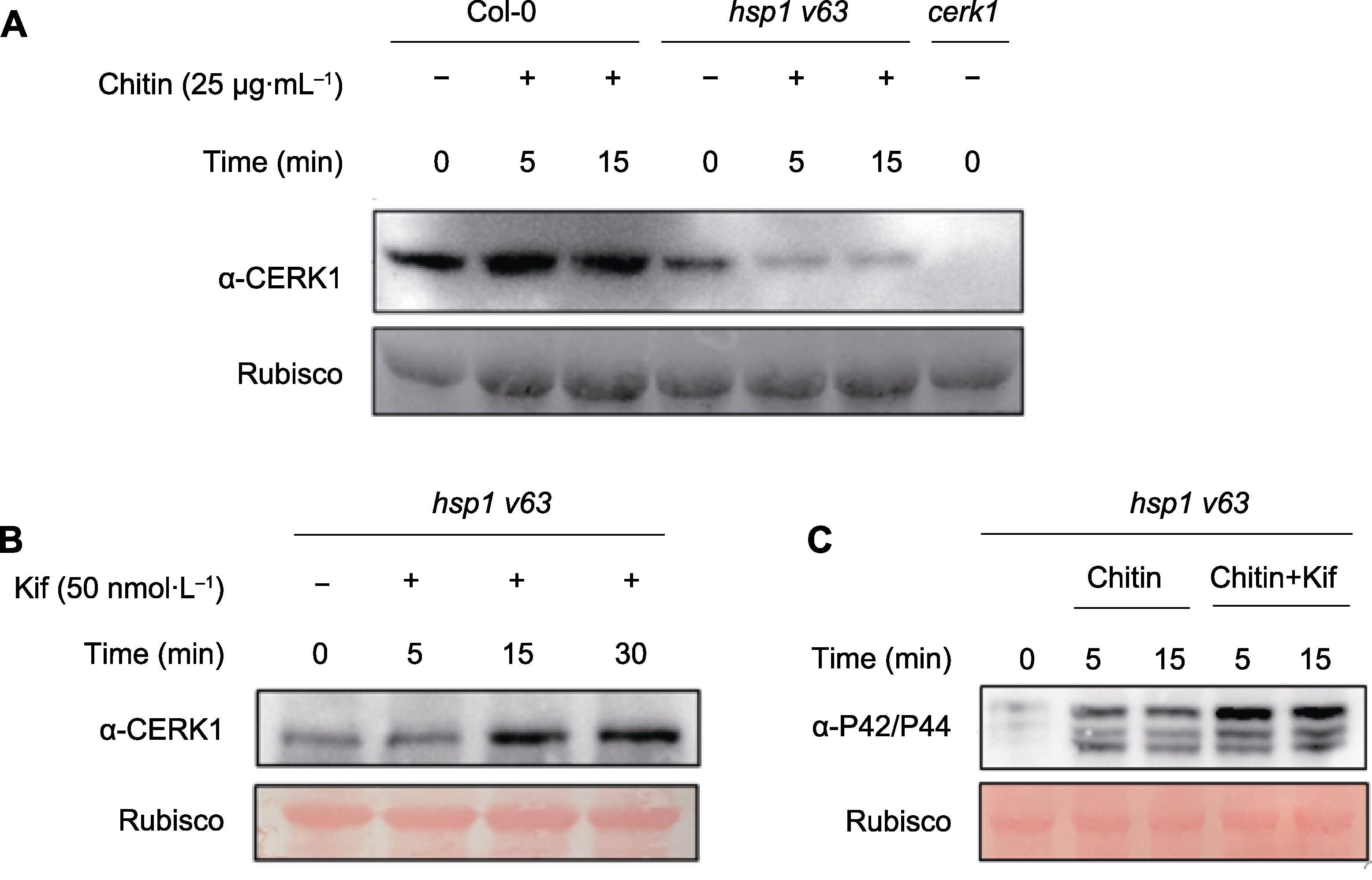
图4 cerk1、hsp1 v63删除突变体和野生型(Col-0)拟南芥中几丁质诱导的CERK1蛋白水平检测 (A) 将拟南芥幼苗在MS平板中浸泡7天, 用几丁质按图所示时间处理幼苗。收样后用Western blot (WB)检测CERK1蛋白水平; (B) 用50 nmol·L-1 Kif处理hsp1 v63删除突变体0、5、15和30分钟。收样后, 使用WB检测CERK1蛋白水平; (C) 用Chitin (25 μg·mL-1)及Chitin (25 μg·mL-1)和Kif (50 nmol·L-1)混合物按图中所示时间处理hsp1 v63删除突变体。收样后, 使用WB检测MAPK3/MAPK6的磷酸化强度。3次重复, 结果相似。
Figure 4 Detection of chitin-induced CERK1 protein levels in cerk1, hsp1 v63 deletion mutants and wild-type Col-0 of Arabidop-sis (A) Arabidopsis thaliana seedlings were soaked in MS plate for 7 days, and the seedlings were treated with 25 μg·mL-1 chitin for the indicated times as shown in the figure. After samples were collected, CERK1 protein level was detected by Western blot (WB) by CERK1 antibody; (B) CERK1 protein level were measured after treated with 50 nmol·L-1 Kif for 0, 5, 15, and 30 minutes under hsp1 v63 deletion mutant; (C) Comparison the phosphorylation of MAPK3/6 after treated by 25 μg·mL-1 Chitin or 25 μg·mL-1 Chitin and 50 nmol·L-1 Kif mixture for indicated time. The experiment was repeated three times with similar results.
| [1] | 欧阳石文, 赵开军, 冯兰香 (2001). 植物几丁质酶的结构与功能、分类及进化. 植物学通报 18, 418-426. |
| [2] | 张静, 杨洪强, 魏钦平 (2003). 几丁质及其衍生物的生物活性与在农业中的应用. 植物学通报 20, 178-183. |
| [3] |
Antolín-Llovera M, Ried MK, Binder A, Parniske M (2012). Receptor kinase signaling path-ways in plant-microbe in-teractions. Annu Rev Phytopathol 50, 451-473.
DOI PMID |
| [4] |
Bi GZ, Zhou ZY, Wang WB, Li L, Rao SF, Wu Y, Zhang XJ, Menke FLH, Chen S, Zhou JM (2018). Receptor-like cytoplasmic kinases directly link diverse pattern recogni-tion receptors to the activation of mitogen-activated pro-tein kinase cascades in Arabidopsis. Plant Cell 30, 1543-1561.
DOI URL |
| [5] |
Bigeard J, Colcombet J, Hirt H (2015). Signaling mecha-nisms in pattern-triggered immunity (PTI). Mol Plant 8, 521-539.
DOI PMID |
| [6] |
Chen LT, Hamada S, Fujiwara M, Zhu TH, Thao NP, Wong HL, Krishna P, Ueda T, Kaku H, Shibuya N, Kawasaki T, Shimamoto K (2010). The Hop/Sti1-Hsp90 chaperone complex facilitates the maturation and transport of a PAMP receptor in rice innate immunity. Cell Host Microbe 7, 185-196.
DOI PMID |
| [7] |
Gong BQ, Guo JH, Zhang NN, Yao XR, Wang HB, Li JF (2019). Cross-microbial protection via priming a conserved immune co-receptor through juxtamembrane phos-phorylation in plants. Cell Host Microbe 26, 810-822.
DOI URL |
| [8] |
Hong Z, Jin H, Tzfira T, Li JM (2008). Multiple mechanism-mediated retention of adefective brassinosteroid receptor in the endoplasmic reticulum of Arabidopsis. Plant Cell 20, 3418-3429.
DOI PMID |
| [9] |
Huang CC, Yan YJ, Zhao HL, Ye Y, Cao YR (2020). Ara-bidopsis CPK5 phosphorylates the chitinreceptor LYK5 to regulate plant innate immunity. Front Plant Sci 11, 702.
DOI URL |
| [10] |
Jones JDG, Dangl JL (2006). The plant immune system. Nature 444, 323-329.
DOI |
| [11] |
Li J, Zhao-Hui C, Batoux M, Nekrasov V, Roux M, Chin-chilla D, Zipfel C, Jones JDG (2009). Specific ER quality control components required for biogenesis of the plant innate immune receptor EFR. Proc Natl Acad Sci USA 106, 15973-15978.
DOI PMID |
| [12] |
Li W, Deng YW, Ning YS, He ZH, Wang GL (2020). Ex-ploiting broad-spectrum disease resistance in crops: from molecular dissection to breeding. Annu Rev Plant Biol 71, 575-603.
DOI URL |
| [13] |
Liu J, Liu B, Chen SF, Gong BQ, Chen LJ, Zhou Q, Xiong F, Wang ML, Feng DR, Li JF, Wang HB, Wang JF (2018). A tyrosine phosphorylation cycle regulates fungal activation of a plant receptor Ser/Thr kinase. Cell Host Microbe 23, 241-253.
DOI PMID |
| [14] |
Lu D, Lin W, Gao X, Wu S, Cheng C, Avila J, Heese A, Devarenne TP, He P, Shan L (2011). Direct ubiquitination of pattern recognition receptor FLS2 attenuates plant in-nate immunity. Science 332, 1439-1442.
DOI URL |
| [15] |
Nagashima Y, von Schaewen A, Koiwa H (2018). Function of N-glycosylation in plants. Plant Sci 274, 70-79.
DOI PMID |
| [16] |
Savary S, Willocquet L, Pethybridge SJ, Esker P, McRoberts N, Nelson A (2019). The global burden of pathogens and pests on major food crops. Nat Ecol Evol 3, 430-439.
DOI PMID |
| [17] |
Strasser R (2018). Protein quality control in the endoplasmic reticulum of plants. Annu Rev Plant Biol 69, 147-172.
DOI PMID |
| [18] |
Tang DZ, Wang GX, Zhou JM (2017). Receptor kinases in plant-pathogen interactions: more than pattern recognition. Plant Cell 29, 618-637.
DOI URL |
| [19] | Zhou JG, Liu DR, Wang P, Ma XY, Lin WW, Chen SX, Mishev K, Lu DP, Kumar R, Vanhoutte I, Meng XZ, He P, Russinova E, Shan LB (2018). Regulation of Arabidopsis brassinosteroid receptor BRI1 endocytosis and degradation by plant U-box PUB12/PUB-13-mediated ubiquitination. Proc Natl Acad Sci USA 115, E1906-E1915. |
| [20] |
Zou JJ, Wei FJ, Wang C, Wu JJ, Ratnasekera D, Liu WX, Wu WH (2010). Arabidopsis calcium-dependent protein kinase CPK10 functions in abscisic acid- and Ca2+-mediated stomatal regulation in response to drought stress. Plant Physiol 154, 1232-1243.
DOI URL |
| [1] | 郭尚敬 陈娜 孟庆伟. 叶绿体小分子量热激蛋白介绍[J]. 植物学报, 2005, 22(02): 223-230. |
| [2] | 张静 杨洪强 魏钦平. 几丁质及其衍生物的生物活性与在农业中的应用[J]. 植物学报, 2003, 20(02): 178-183. |
| [3] | 欧阳石文 赵开军 冯兰香. 植物几丁质酶的结构与功能、分类及进化[J]. 植物学报, 2001, 18(04): 418-426. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||