

Chinese Bulletin of Botany ›› 2019, Vol. 54 ›› Issue (3): 396-404.DOI: 10.11983/CBB18099 cstr: 32102.14.CBB18099
• SPECIAL TOPICS • Previous Articles
Tingting Shan1,Xiaomei Chen1,*( ),Shunxing Guo1,*(
),Shunxing Guo1,*( ),Lixia Tian1,Lin Yan2,Xin Wang1
),Lixia Tian1,Lin Yan2,Xin Wang1
Received:2018-04-16
Accepted:2018-10-06
Online:2019-05-01
Published:2019-11-24
Contact:
Xiaomei Chen,Shunxing Guo
Tingting Shan,Xiaomei Chen,Shunxing Guo,Lixia Tian,Lin Yan,Xin Wang. Advances in Molecular Regulation of Sphingolipids in Plant-fungus Interactions[J]. Chinese Bulletin of Botany, 2019, 54(3): 396-404.
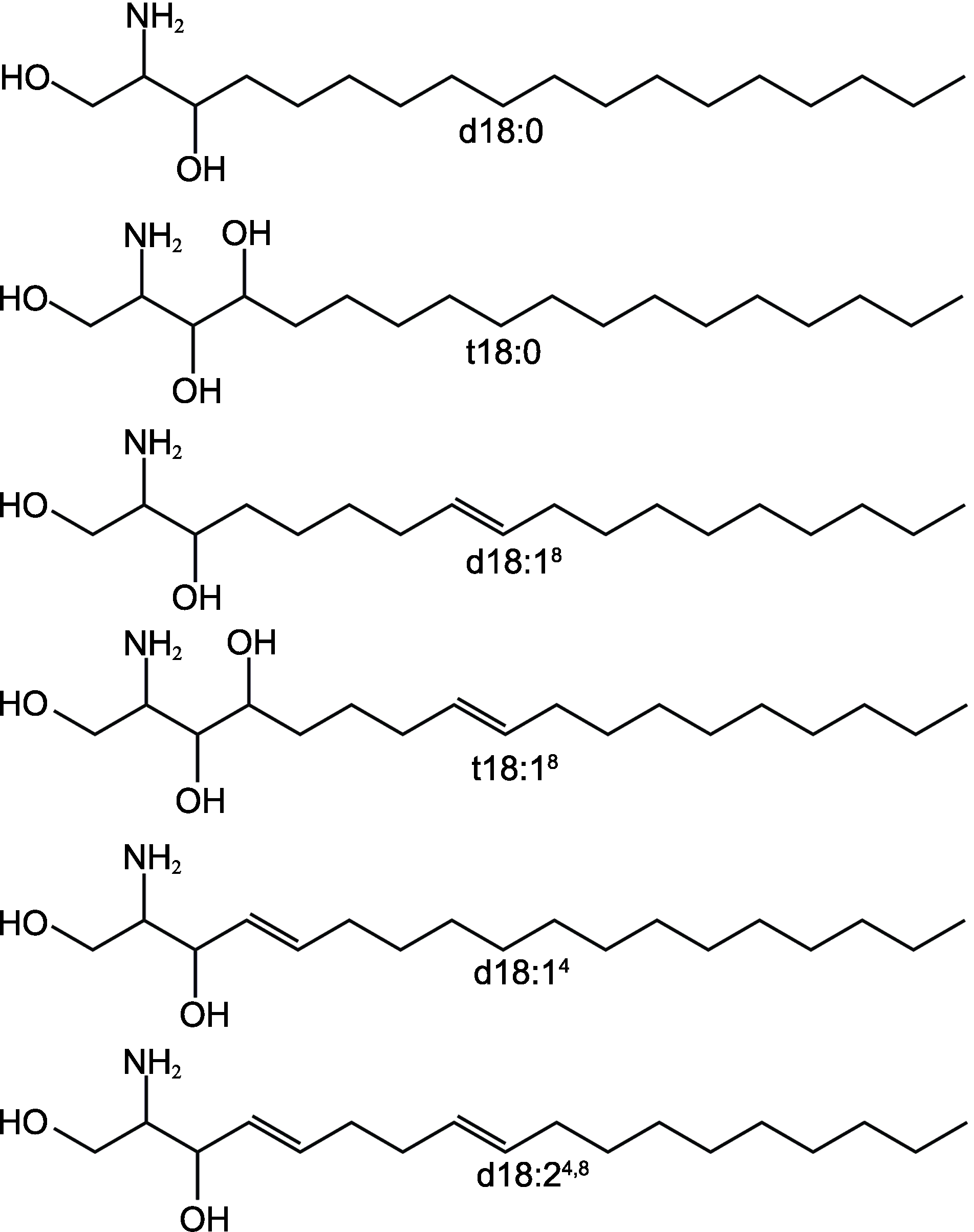
Figure 2 Sphingosines in the structure of plant sphingolipids (modified from Warnecke and Heinz, 2003)d18:0: Dihydrosphingosine; t18:0: Phytosphingosine; d18:18: Sphing-8-enine; t18:18: 4-hydroxysphing-8-enine; d18:14: Sphingosine; d18:24,8: Sphinga-4,8-dienine
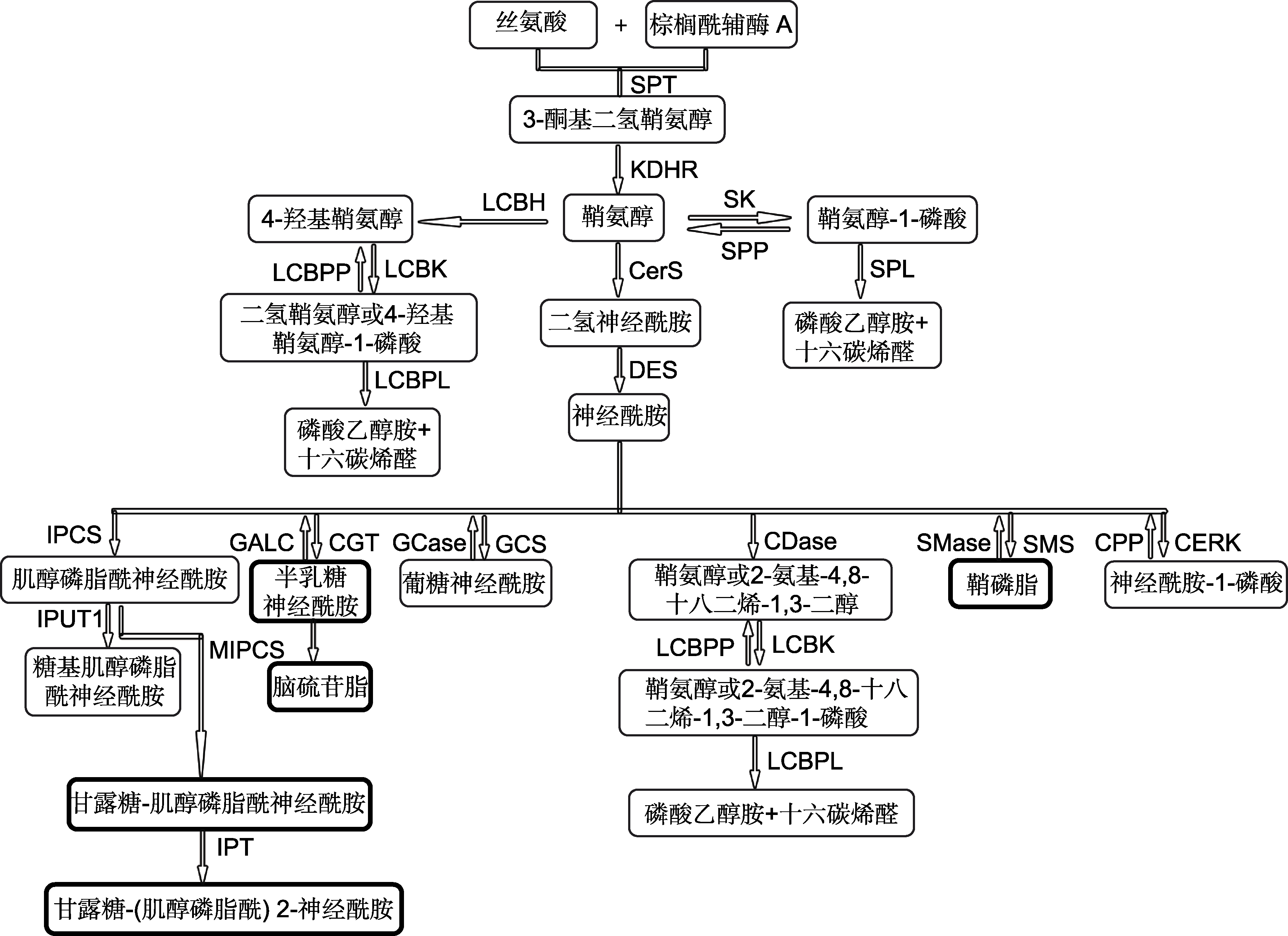
Figure 3 Sphingolipid biosynthesis in plant and fungi (modified from Michaelson et al., 2016; Rollin-pinheiro et al., 2016) The bold border boxes are unique products of fungi. SPT: Serine palmitoyltransferase; KDHR: 3-ketodihydrosphingosine reductase; CerS: (dihydro) Ceramide synthase; DES: Dihydroceramide desaturase; SK: Sphingosine kinase; SPP: higoine- 1-phosphate phosphatases; SPL: Sphingosine-1-phosphate lyase; LCBH: LCB hydroxylase; CERK: Ceramide kinase; CPP: Ceramide-1-phosphate phosphatases; SMS: Sphingomyelin synthase; SMase: Sphingomyelinases; LCBK: LCB kinase; LCBPP: LCB-1-phosphate phosphatases; LCBPL: LCB-1-phosphate lyase; IPCS: Inositol phosphoryl ceramide synthase; IPUT1: Inositol phosphorylceramide glucuronosyl transferases 1; MIPCS: Mannosylinositol phosphorylceramide synthase; IPT: Inositol phosphoryl transferase; CGT: Galactosyltransferase; GALC: Galactosylceramidase; GCS: Glucosylceramide synthase; GCase: Glucosylceramidase; CDase: Ceramidase
| [1] | 郭顺星 ( 2016). 药用植物内生真菌生物学. 北京: 科学出版社. pp. 190-191. |
| [2] | 林久生, 王根轩 ( 2001). 活性氧与植物细胞编程性死亡. 植物生理学通讯 37, 551-555. |
| [3] |
刘润华, 江文波, 余迪求 ( 2009). 植物鞘脂的结构、代谢途径及其功能. 植物学报 44, 619-628.
DOI URL |
| [4] |
田磊, 李元敬, 何兴元, 田春杰 ( 2016). 丛枝菌根真菌脂类代谢对共生信号调控的响应和反馈. 微生物学报 56, 26-34.
DOI URL |
| [5] | 张海涵 ( 2011). 黄土高原枸杞根际微生态特征及其共生真菌调控宿主生长与耐旱响应机制. 博士论文. 杨凌: 西北农林科技大学. pp. 38-40. |
| [6] |
Aerts AM, François IEJA, Bammens L, Cammue BPA, Smets B, Winderickx J, Accardo S, De Vos DE, Thevissen K ( 2006). Level of M(IP)2C sphingolipid affects plant defensin sensitivity, oxidative stress resistance and chronological life-span in yeast. FEBS Lett 580, 1903-1907.
DOI PMID |
| [7] |
Arora P, Porcelli SA ( 2008). A glycan shield for bacterial sphingolipids. Chem Biol 15, 642-644.
DOI URL PMID |
| [8] |
Barreto-Bergter E, Sassaki GL, De Souza LM ( 2011). Structural analysis of fungal cerebrosides. Front Microbiol 2, 239.
DOI URL PMID |
| [9] |
Bi FC, Liu Z, Wu JX, Liang H, Xi XL, Fang C, Sun TJ, Yin J, Dai GY, Rong C, Greenberg JT, Su WW, Yao N ( 2014). Loss of ceramide kinase in Arabidopsis impairs defenses and promotes ceramide accumulation and mito- chondrial H2O2 bursts. Plant Cell 26, 3449-3467.
DOI URL PMID |
| [10] |
Brandwagt BF, Kneppers TJA, Nijkamp HJJ, Hille J ( 2002). Overexpression of the tomato ASC-1 gene mediates high insensitivity to AAL toxins and fumonisin B1 in tomato hairy roots and confers resistance to Alternaria alternata f. sp. lycopersici in Nicotiana umbratica plants. Mol Plant Microbe Interact 15, 35-42.
DOI URL PMID |
| [11] |
Chen M, Markham JE, Cahoon EB ( 2012). Sphingolipid Δ8 unsaturation is important for glucosylceramide biosynthesis and low-temperature performance in Arabidopsis. Plant J 69, 769-781.
DOI URL PMID |
| [12] |
De Zélicourt A, Montiel G, Pouvreau JB, Thoiron S, Delgrange S, Simier P, Delavault P ( 2009). Susceptibility of Phelipanche and Orobanche species to AAL-toxin. Planta 230, 1047-1055.
DOI URL PMID |
| [13] |
Deepak SA, Raj SN, Umemura K, Kono T, Shetty HS ( 2003). Cerebroside as an elicitor for induced resistance against the downy mildew pathogen in pearl millet. Ann Appl Biol 143, 169-173.
DOI URL |
| [14] |
Healey KR, Challa KK, Edlind TD, Katiyar SK ( 2015). Sphingolipids mediate differential echinocandin susceptibility in Candida albicans and Aspergillus nidulans. Anti microb Agents Chemother 59, 3377-3384.
DOI URL PMID |
| [15] |
Hechtberger P, Zinser E, Saf R, Hummel K, Paltauf F, Daum G ( 1994). Characterization, quantification and subcellular localization of inositol-containing sphingolipids of the yeast, Saccharomyces cerevisiae. Eur J Biochem 225, 641-649.
DOI URL PMID |
| [16] |
Lachaud C, Silva DD, Cotelle V, Thuleau P, Xiong TC, Jauneau A, Brière C, Graziana A, Bellec Y, Faure JD, Ranjeva R, Mazars C ( 2010). Nuclear calcium controls the apoptotic-like cell death induced by D-erythro-sphinganine in tobacco cells. Cell Calcium 47, 92-100.
DOI URL PMID |
| [17] |
Lindahl L, Santos AXS, Olsson H, Olsson L, Bettiga M ( 2017). Membrane engineering of S. cerevisiae targeting sphingolipid metabolism. Sci Rep 7, 41868.
DOI URL PMID |
| [18] |
Magnin-Robert M, Le Bourse D, Markham J, Dorey S, Clément C, Baillieul F, Dhondt-Cordelier S ( 2015). Modifications of sphingolipid content affect tolerance to hemibiotrophic and necrotrophic pathogens by modulating plant defense responses in Arabidopsis. Plant Physiol 169, 2255-2274.
DOI URL PMID |
| [19] |
Maillet F, Poinsot V, André O, Puech-Pagès V, Haouy A, Gueunier M, Cromer L, Giraudet D, Formey D, Niebel A, Martinez EA, Driguez H, Bécard G, Dénarié J ( 2011). Fungal lipochitooligosaccharide symbiotic signals in arbu- scular mycorrhiza. Nature 469, 58-63.
DOI URL PMID |
| [20] |
Markham JE, Lynch DV, Napier JA, Dunn TM, Cahoon EB ( 2013). Plant sphingolipids: function follows form. Curr Opin Plant Biol 16, 350-357.
DOI URL PMID |
| [21] |
Michaelson LV, Napier JA, Molino D, Faure JD ( 2016). Plant sphingolipids: their importance in cellular organization and adaption. Biochim Biophys Acta 1861, 1329-1335.
DOI URL PMID |
| [22] |
Mortimer JC, Yu X, Albrecht S, Sicilia F, Huichalaf M, Ampuero D, Michaelson LV, Murphy AM, Matsunaga T, Kurz S, Stephens E, Baldwin TC, Ishii T, Napier JA, Weber APM, Handford MG, Dupree P ( 2013). Abnormal glycosphingolipid mannosylation triggers salicylic acid mediated responses in Arabidopsis. Plant Cell 25, 1881-1894.
DOI URL PMID |
| [23] |
Oguro Y, Yamazaki H, Takagi M, Takaku H ( 2014). Antifungal activity of plant defensin AFP1 in Brassica juncea involves the recognition of the methyl residue in glucosylceramide of target pathogen Candida albicans. Curr Genet 60, 89-97.
DOI URL PMID |
| [24] |
Olsen I, Jantzen E ( 2001). Sphingolipids in bacteria and fungi. Anaerobe 7, 103-112.
DOI URL |
| [25] |
Peer M, Stegmann M, Mueller MJ, Waller F ( 2010). Pseudomonas syringae infection triggers de novo synthesis of phytosphingosine from sphinganine in Arabidopsis thaliana. FEBS Lett 584, 4053-4056.
DOI URL PMID |
| [26] |
Ramamoorthy V, Cahoon EB, Li J, Thokala M, Minto RE, Shah DM ( 2007). Glucosylceramide synthase is essential for alfalfa defensin-mediated growth inhibition but not for pathogenicity of Fusarium graminearum. Mol Microbiol 66, 771-786.
DOI URL PMID |
| [27] |
Ramamoorthy V, Cahoon EB, Thokala M, Kaur J, Li J, Shah DM ( 2009). Sphingolipid C-9 methyltransferases are important for growth and virulence but not for sensitivity to antifungal plant defensins in Fusarium graminearum. Eukaryot Cell 8, 217-229.
DOI URL PMID |
| [28] |
Rittershaus PC, Kechichian TB, Allegood JC, Merrill AH, Hennig M, Luberto C, Del Poeta M ( 2006). Glucosylceramide synthase is an essential regulator of pathogenicity of Cryptococcus neoformans. J Clin Invest 116, 1651-1659.
DOI URL PMID |
| [29] |
Rollin-Pinheiro R, Singh A, Barreto-Bergter E, Poeta MD ( 2016). Sphingolipids as targets for treatment of fungal infections. Future Med Chem 8, 1469-1484.
DOI URL PMID |
| [30] |
Saucedo-García M, Guevara-García A, González-Solís A, Cruz-García F, Vázquez-Santana S, Markham JE, Guadalupe Lozano-Rosas M, Dietrich CR, Ramos-Vega M, Cahoon EB, Gavilanes-Ruíz M ( 2011). MPK6, sphinganine and the LCB2a gene from serine palmitoyltransferase are required in the signaling pathway that mediates cell death induced by long chain bases in Arabidopsis. New Phytol 191, 943-957.
DOI URL PMID |
| [31] |
Sharma L, Prakash H ( 2017). Sphingolipids are dual specific drug targets for the management of pulmonary infections: perspective. Front Immunol 8, 378.
DOI URL PMID |
| [32] |
Shi LH, Bielawski J, Mu JY, Dong HL, Teng C, Zhang J, Yang XH, Tomishige N, Hanada K, Hannun YA, Zuo J ( 2007). Involvement of sphingoid bases in mediating reactive oxygen intermediate production and programmed cell death in Arabidopsis. Cell Res 17, 1030-1040.
DOI URL PMID |
| [33] |
Siebers M, Brands M, Wewer V, Duan YJ, Hölzl G, Dörmann P ( 2016). Lipids in plant-microbe interactions. Biochim Biophys Acta 1861, 1379-1395.
DOI URL PMID |
| [34] |
Spassieva SD, Markham JE, Hille J ( 2002). The plant disease resistance gene Asc-1 prevents disruption of sphingolipid metabolism during AAL-toxin-induced programmed cell death. Plant J 32, 561-572.
DOI URL PMID |
| [35] |
Tani Y, Amaishi Y, Funatsu T, Ito M, Itonor S, Hata Y, Ashida H, Yamamoto K ( 2014). Structural analysis of cerebrosides from Aspergillus fungi: the existence of galactosylceramide in A. oryzae. Biotechnol Lett 36, 2507-2513.
DOI URL PMID |
| [36] |
Tartaglio V, Rennie EA, Cahoon R, Wang G, Baidoo E, Mortimer JC, Cahoon EB, Scheller HV ( 2017). Glycosylation of inositol phosphorylceramide sphingolipids is required for normal growth and reproduction in Arabidosis. Plant J 89, 278-290.
DOI URL PMID |
| [37] |
Thevissen K, François IEJA, Takemoto JY, Ferket KKA, Meert EMK, Cammue BPA ( 2003). DmAMP1, an antifungal plant defensin from dahlia (Dahlia merckii), interacts with sphingolipids from Saccharomyces cerevisiae. FEMS Microbiol Lett 226, 169-173.
DOI URL PMID |
| [38] |
Thevissen K, Warnecke DC, François IEJA, Leipelt M, Heinz E, Ott C, Zähringer U, Thomma BPHJ, Ferket KKA, Cammue BPA ( 2004). Defensins from insects and plants interact with fungal glucosylceramides. J Biol Chem 279, 3900-3905.
DOI URL PMID |
| [39] |
Townley HE, McDonald K, Jenkins GI, Knight MR, Leaver CJ ( 2005). Ceramides induce programmed cell death in Arabidopsis cells in a calcium-dependent manner. Biol Chem 386, 161-166.
DOI URL PMID |
| [40] |
Ueda N ( 2017). Sphingolipids in genetic and acquired forms of chronic kidney diseases. Curr Med Chem 24, 1238-1275.
DOI URL PMID |
| [41] |
Umemura K, Ogawa N, Koga J, Iwata M, Usami H ( 2002). Elicitor activity of cerebroside, a sphingolipid elicitor, in cell suspension cultures of rice. Plant Cell Physiol 43, 778-784.
DOI URL PMID |
| [42] |
Umemura K, Ogawa N, Yamauchi T, Iwata M, Shimura M, Koga J ( 2000). Cerebroside elicitors found in diverse phytopathogens activate defense responses in rice plants. Plant Cell Physiol 41, 676-683.
DOI URL PMID |
| [43] | Virolainen E, Blokhina O, Fagerstedt K ( 2002). Ca 2+-in- duced high amplitude swelling and cytochrome c release from wheat (Triticum aestivum L.) mitochondria under anoxic stress . Ann Bot 90, 509-516. |
| [44] |
Wang WM, Yang XH, Tangchaiburana S, Ndeh R, Markham JE, Tsegaye Y, Dun TM, Wang GL, Bellizzi M, Parsons JF, Morrissey D, Bravo JE, Lynch DV, Xiao SY ( 2008). An inositolphosphorylceramide synthase is involved in regulation of plant programmed cell death associated with defense in Arabidopsis. Plant Cell 20, 3163-3179.
DOI URL |
| [45] |
Warnecke D, Heinz E ( 2003). Recently discovered functions of glucosylceramides in plants and fungi. Cell Mol Life Sci 60, 919-941.
DOI URL PMID |
| [46] |
Yanagawa D, Ishikawa T, Imai H ( 2017). Synthesis and degradation of long-chain base phosphates affect fumonisin B1-induced cell death in Arabidopsis thaliana. J Plant Res 130, 571-585.
DOI URL PMID |
| [47] |
Zhu CY, Wang MS, Wang WL, Ruan RJ, Ma HJ, Mao CG, Li HY ( 2014). Glucosylceramides are required for mycelial growth and full virulence in Penicillium digitatum. Biochem Biophys Res Commun 455, 165-171.
DOI URL PMID |
| [1] | Zihong Chen, Yifei Zhang, Kai Chen, Jianying Chen, Ling Xu. Species diversity of entomopathogenic fungi and the influencing factors in the Southern Gaoligong Mountains [J]. Biodiv Sci, 2025, 33(1): 24228-. |
| [2] | Haidi Qi, Dinghai Zhang, Lishan Shan, Guopeng Chen, Bo Zhang. Advances in the mechanisms of entomopathogenic fungi infecting insect hosts and the defense strategies of insects [J]. Biodiv Sci, 2023, 31(11): 23273-. |
| [3] | Tiantian Zhi, Zhou Zhou, Chengyun Han, Chunmei Ren. PAD4 Mutation Accelerating Programmed Cell Death in Arabidopsis thaliana Tyrosine Degradation Deficient Mutant sscd1 [J]. Chinese Bulletin of Botany, 2022, 57(3): 288-298. |
| [4] | Hai-Sheng Yuan, Yulian Wei, Liwei Zhou, Wenmin Qin, Baokai Cui, Shuanghui He. Potential distribution and ecological niches of four butt-rot pathogenic fungi in Northeast China [J]. Biodiv Sci, 2019, 27(8): 873-879. |
| [5] | Zhao Xijuan, Qian Lichao, Liu Yule. Chinese Scientists Made Breakthrough Progresses in Plant Programmed Cell Death [J]. Chinese Bulletin of Botany, 2018, 53(4): 447-450. |
| [6] | Zhang Xian-sheng. Chinese Scientists Have Made a Great Breakthrough in the Mechanism of Programmed Cell Death [J]. Chinese Bulletin of Botany, 2018, 53(4): 445-446. |
| [7] | He Guangming, Deng Xingwang. Death Signal Transduction: Chloroplast-to-Mitochondrion Communication Regulates Programmed Cell Death in Plants [J]. Chinese Bulletin of Botany, 2018, 53(4): 441-444. |
| [8] | Xiao Huang, Faqiang Li. Roles of Autophagy in Plant Programmed Cell Death [J]. Chinese Bulletin of Botany, 2016, 51(6): 859-862. |
| [9] | Lin Li, Kang Tan, Xiuguang Tang, Xiaoting Chao, Chenxi Wen, Zhuangdong Bai Hualing Feng, Wenzhe Liu, Hui Su. Characterization of Programmed Cell Death During the Senescence of Root Hairs in Arabidopsis [J]. Chinese Bulletin of Botany, 2016, 51(2): 194-201. |
| [10] | Xinqiang He, Hong Wu. Mechanisms of Developmental Programmed Cell Death in Plants [J]. Chinese Bulletin of Botany, 2013, 48(4): 357-370. |
| [11] | Hongjuan Jing, Guangzhou Zhou, Xiaorong Tan, Kangkang Ping, Xuejian Ren. Research Progress in Regulation of Reactive Oxygen Species in Plant Autophagy [J]. Chinese Bulletin of Botany, 2012, 47(5): 534-542. |
| [12] | Cong Ma, Weiwen Kong. Research Progress in Plant Metacaspase [J]. Chinese Bulletin of Botany, 2012, 47(5): 543-549. |
| [13] | MA Huai-Yu, LÜ De-Guo, YANG Hong-Qiang. Characteristics of mitochondria and cell death in roots of Malus hupehensis var. pingyiensis under NaCl stress [J]. Chin J Plant Ecol, 2010, 34(12): 1448-1453. |
| [14] | LI Rong-Feng, CAI Miao-Zhen, LIU Peng, XU Gen-Di, CHEN Min-Yan, LIANG He. PHYTOECOLOGICAL EFFECT OF Al3+ ON THE INDUCTIVITY OF PROGRAMMED CELL DEATH OF BORDER CELLS IN SOYBEAN ROOT [J]. Chin J Plant Ecol, 2008, 32(3): 690-697. |
| [15] | Yuliang Chen;Feixiong Zhang;Guiyou Zhang*. Key Caspase-like Enzymes in Programmed Cell Death in Plants [J]. Chinese Bulletin of Botany, 2008, 25(05): 616-623. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||