

Chinese Bulletin of Botany ›› 2018, Vol. 53 ›› Issue (2): 227-237.DOI: 10.11983/CBB17136 cstr: 32102.14.CBB17136
• EXPERIMENTAL COMMUNICATIONS • Previous Articles Next Articles
Zhao Xuehui1, Xiao Wei1,2, Guo Jianmin1, Gao Dongsheng1,2, Fu Xiling1,2,*( ), Li Dongmei1,2,3,*(
), Li Dongmei1,2,3,*( )
)
Received:2017-06-14
Accepted:2017-10-19
Online:2018-03-01
Published:2018-08-10
Contact:
Fu Xiling,Li Dongmei
Zhao Xuehui, Xiao Wei, Guo Jianmin, Gao Dongsheng, Fu Xiling, Li Dongmei. Effect of Blue Light on Photosynthetic Performance and Accumulation of Sugar and Organic Acids in Greenhouse Nectarine[J]. Chinese Bulletin of Botany, 2018, 53(2): 227-237.
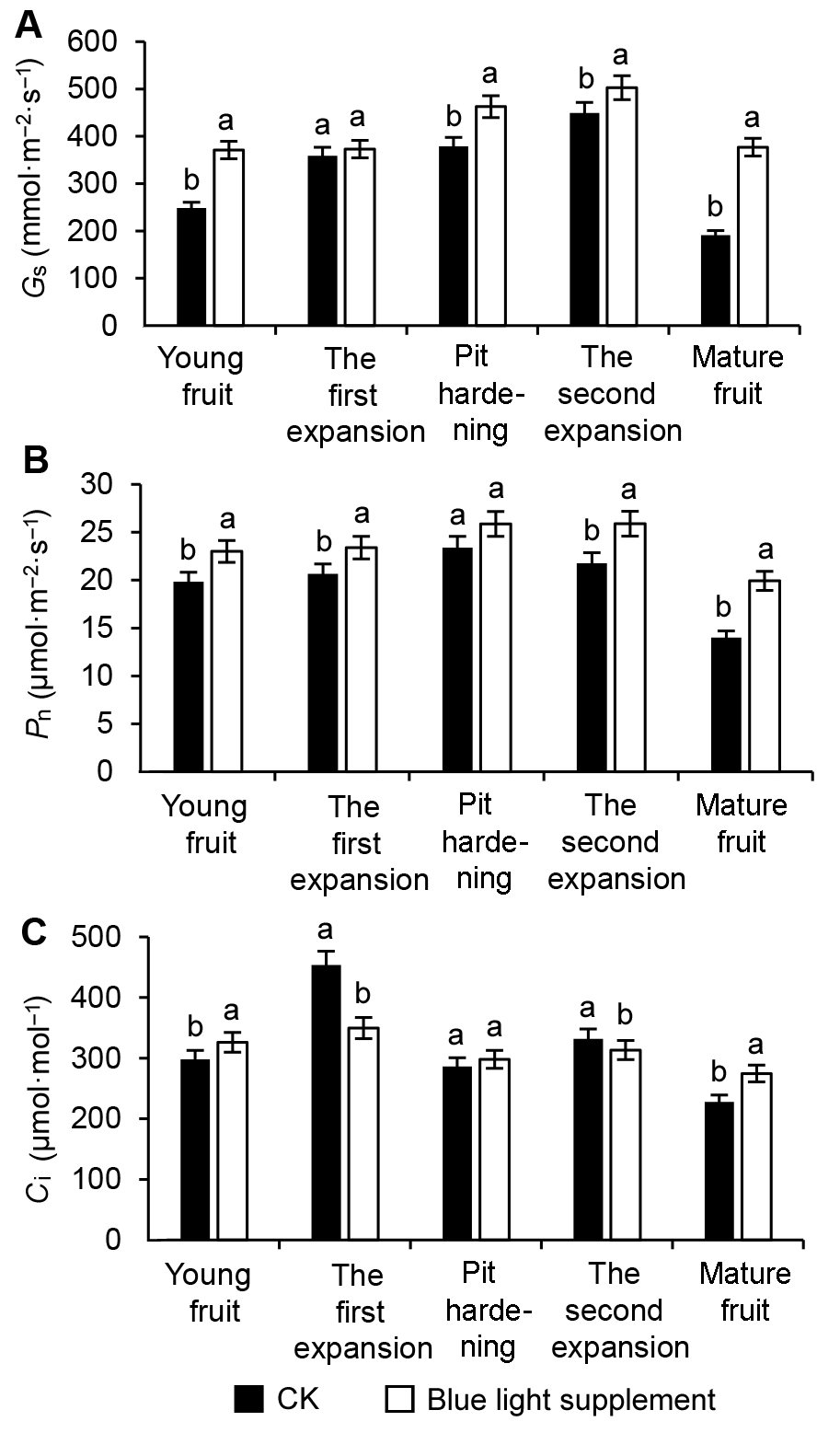
Figure 1 Effects of supplementary blue light on photosynthetic parameters in leaves during peach fruit development(A) Stomatal conductance (Gs); (B) Net photosynthetic rate (Pn); (C) Intercellular CO2 concentration (Ci). Different lowercase letters indicate significant differences at 0.05 level among treatments.
| Treatment | Chlorophyll a (mg·g-1 FW) | Chlorophyll b (mg·g-1 FW) | Chlorophyll (a+b) (mg·g-1 FW) | Chlorophyll a/b | Carotenoids (mg·g-1 FW) | Leaf area (cm2) |
|---|---|---|---|---|---|---|
| Blue light | 2.630* | 0.868* | 3.498* | 3.030 | 0.491 | 75.100* |
| CK | 2.305 | 0.706 | 3.011 | 3.265* | 0.522 | 46.458 |
Table 1 Effects of supplementary blue light on pigment content and leaf area
| Treatment | Chlorophyll a (mg·g-1 FW) | Chlorophyll b (mg·g-1 FW) | Chlorophyll (a+b) (mg·g-1 FW) | Chlorophyll a/b | Carotenoids (mg·g-1 FW) | Leaf area (cm2) |
|---|---|---|---|---|---|---|
| Blue light | 2.630* | 0.868* | 3.498* | 3.030 | 0.491 | 75.100* |
| CK | 2.305 | 0.706 | 3.011 | 3.265* | 0.522 | 46.458 |
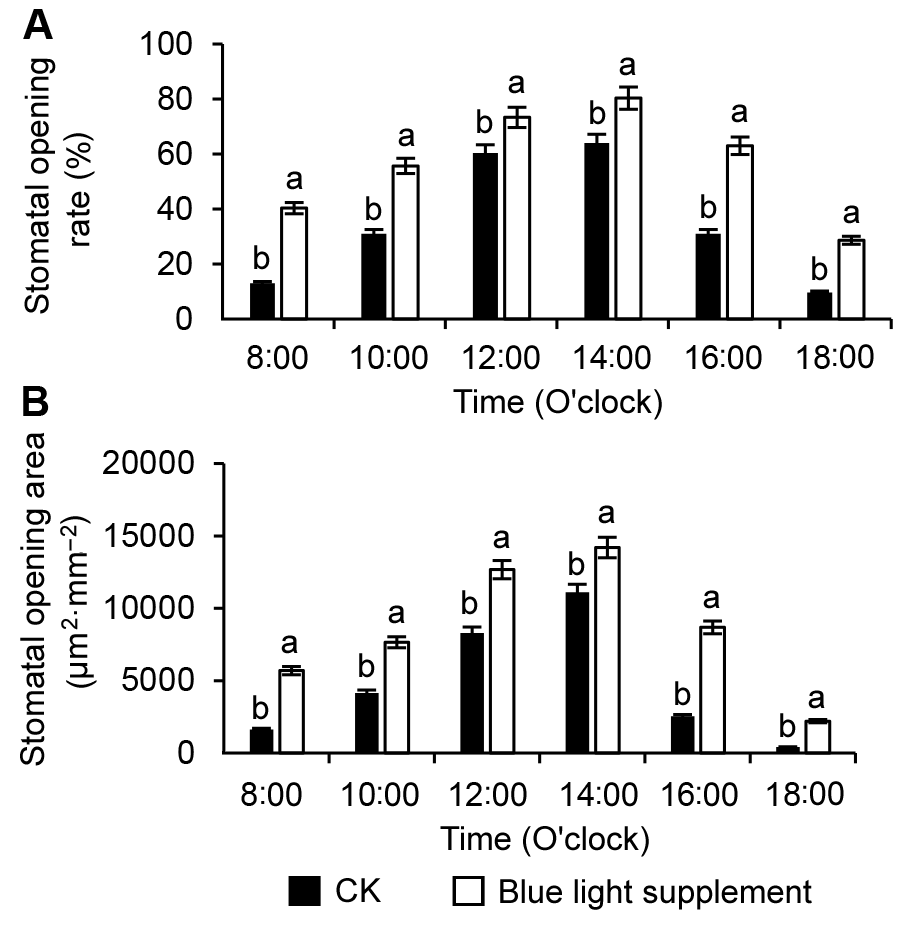
Figure 2 The statistical analysis of diurnal variation of stomatal opening rate (A) and stomatal opening area (B) of peach leavesDifferent lowercase letters indicate significant differences at 0.05 level among treatments.
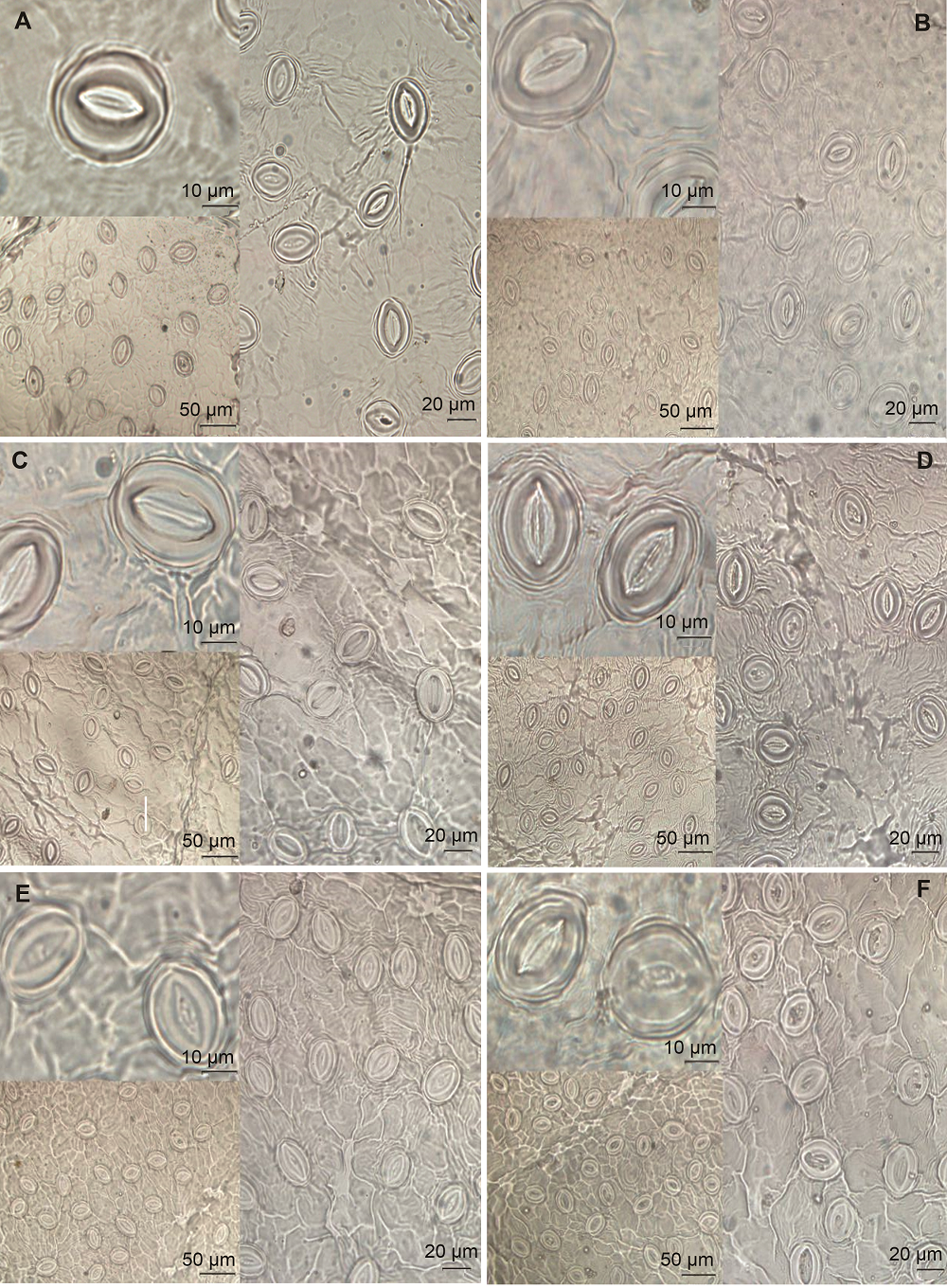
Figure 3 Observation of stomatal morphology of peach leaves at some critical moments in a day(A), (C) and (E) The stomatal morphology in the control of peach leaves at 8:00, 14:00 and 18:00, respectively; (B), (D) and (F) The stomatal morphology in the supplemental blue light of peach leaves at 8:00, 14:00 and 18:00, respectively.
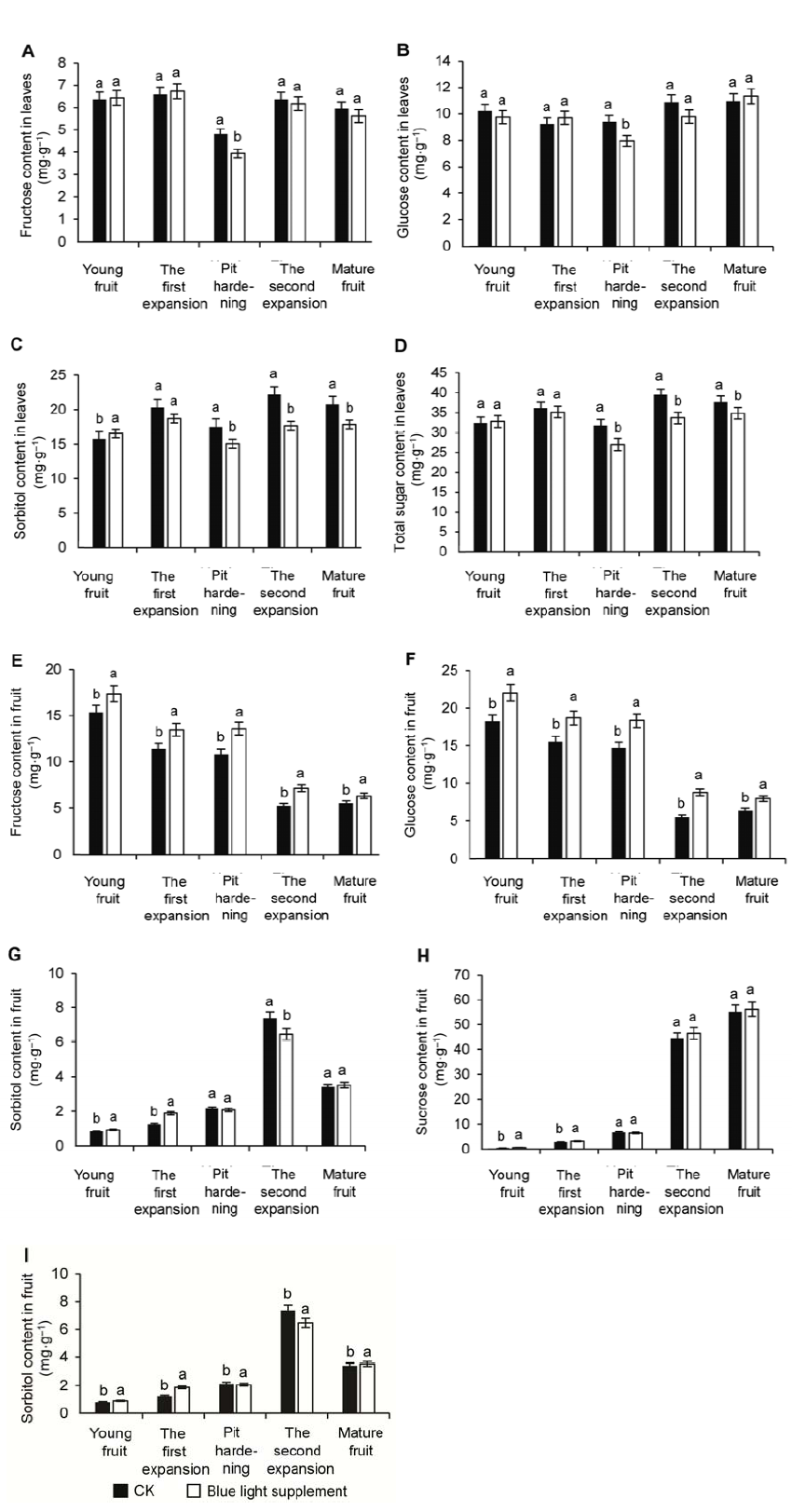
Figure 4 Effects of supplementary blue light on photo-assi- milate in leaves and fruit during peach fruit development(A) The contents of fructose in leaves; (B) The contents of glucose in leaves; (C) The contents of sorbitol in leaves; (D) The contents of total sugar in leaves; (E) The contents of fructose in fruit; (F) The contents of glucose in fruit; (G) The contents of sorbitol in fruit; (H) The contents of sucrose in fruit; (I) The contents of total sugar in fruit. Different lowercase letters indicate significant differences at 0.05 level among treatments.
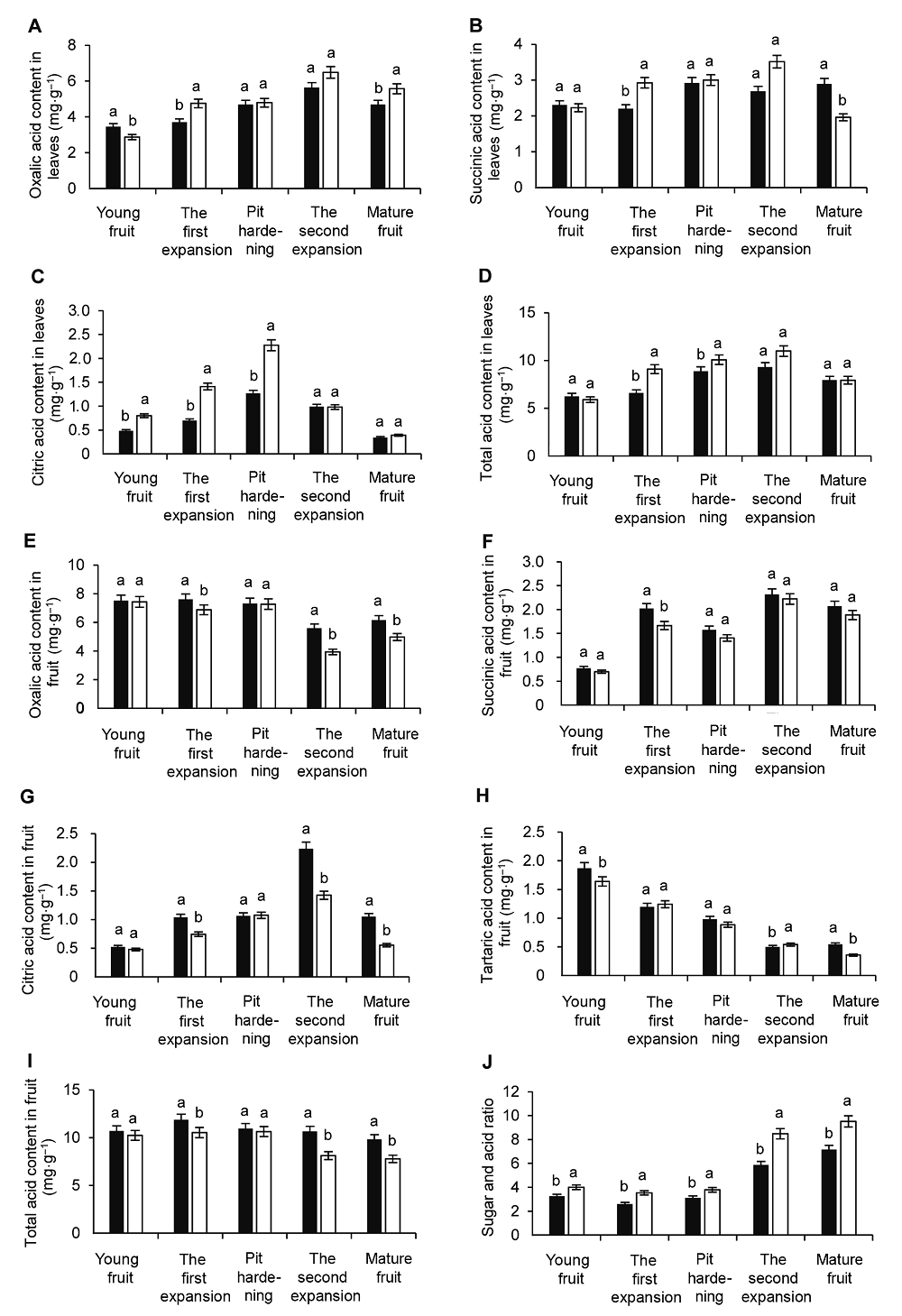
Figure 5 Effects of supplementary blue light on organic acid in leaves and fruit and the sugar and acid ratio of fruit during peach fruit development(A) The contents of oxalic acid in leaves; (B) The contents of succinic acid in leaves; (C) The contents of citric acid in leaves; (D) The contents of total acid in leaves; (E) The contents of oxalic acid in fruit; (F) The contents of succinic acid in fruit; (G) The contents of citric acid in fruit; (H) The contents of tartaric acid in fruit; (I) The contents of total acid in fruit; (J) Sugar and acid ratio of fruit. Different lowercase letters indicate significant differences at 0.05 level among treatments.
| [1] | 白宝璋, 张宪政 (1992). 植物生理学. 北京: 中国科学技术出版社. pp. 219. |
| [2] | 包维楷, 冷俐 (2005). 苔藓植物光合色素含量测定方法——以暖地大叶藓为例. 应用与环境生物学报 11, 235-237. |
| [3] | 车根, 刘晓, 窦同考 (2011). 太平洋嘎拉苹果果实风味评价. 山东农业科学 (1), 44-46. |
| [4] | 陈根云, 陈娟, 许大全 (2010). 关于净光合速率和胞间CO2浓度关系的思考. 植物生理学通讯 46, 64-66. |
| [5] | 崔瑾, 徐志刚, 邸秀茹 (2008). LED在植物设施栽培中的应用和前景. 农业工程学报 24(8), 249-253. |
| [6] | 巩彪, 靳志勇, 刘娜, 刘世琦, 王秀峰, 艾希珍, 魏珉, 史庆华 (2016). 光质对紫背天葵生长、次生代谢和抗氧化胁迫的影响. 应用生态学报 27, 3577-3584. |
| [7] | 郭从俭, 张新胜, 张万钦 (1996). 气孔性状对楸树生长及早期选择的影响. 河南农业大学学报 30, 65-71. |
| [8] | 国志信 (2016). 红光对番茄光合作用启动过程的系统调控机制. 博士论文. 杭州: 浙江大学. pp. 20-24. |
| [9] | 景艳军, 林荣呈 (2017). 我国植物光信号转导研究进展概述. 植物学报 52, 257-270. |
| [10] | 李冬梅, 谭秋平, 高东升, 陈修德, 李玲 (2014). 光周期对休眠诱导期桃树光合及PSII光系统性能的影响. 应用生态学报 25, 1933-1939. |
| [11] | 李俊芝, 王功帅, 胡艳丽, 陈学森, 毛志泉 (2014). 几种低分子量有机酸对连作平邑甜茶幼苗光合与根系生长的影响. 园艺学报 41, 2489-2496. |
| [12] | 李勇, 朱更瑞, 方伟超, 闫顺杰, 赵佩, 赵娟 (2014). 桃设施栽培研究进展. 江苏农业科学 42, 162-166. |
| [13] | 刘金豹 (2004). 加工苹果果实中糖酸和酚类物质研究. 硕士论文. 泰安: 山东农业大学. pp. 25-27. |
| [14] | 刘庆, 连海峰, 刘世琦, 孙亚丽, 于新会, 郭会平 (2015). 不同光质LED光源对草莓光合特性、产量及品质的影响. 应用生态学报 26, 1743-1750. |
| [15] | 刘士华, 吕国胜 (2011). 草莓果实成熟过程中糖、酸含量的动态变化. 安徽林业科技 37(5), 24-27. |
| [16] | 刘晓英, 常涛涛, 郭世荣, 徐志刚, 陈文昊 (2010). 红蓝LED光全生育期照射对樱桃番茄果实品质的影响. 中国蔬菜 (22), 21-27. |
| [17] | 刘张垒 (2014). 光质对黄瓜生长、生理特性、产量及品质的影响. 硕士论文. 泰安: 山东农业大学. pp.18-19. |
| [18] | 彭小芮 (2016). 草莓果实发育过程糖积累差异及光质对糖代谢途径的影响. 硕士论文. 雅安: 四川农业大学. pp. 25-34. |
| [19] | 蒲高斌, 刘世琦, 刘磊, 任丽华 (2005). 不同光质对番茄幼苗生长和生理特性的影响. 园艺学报 32, 420-425. |
| [20] | 秦改花, 黄文江, 赵建荣, 邓盾, 周玉丽 (2011). 石榴果实的糖酸组成及风味特点. 热带作物学报 32, 2148-2151. |
| [21] | 苏娜娜, 邬奇, 崔瑾 (2012). LED光质补光对黄瓜幼苗生长和光合特性的影响. 中国蔬菜 (24), 48-54. |
| [22] | 孙娜 (2015). 光质对番茄生长、生理代谢及果实产量品质的影响. 硕士论文. 泰安: 山东农业大学. pp. 22. |
| [23] | 王海波, 李林光, 陈学森, 李慧峰, 杨建明, 刘嘉芬, 王超 (2010). 中早熟苹果品种果实的风味物质和风味品质. 中国农业科学 43, 2300-2306. |
| [24] | 王帅, 王海波, 王孝娣, 史祥宾, 王宝亮, 郑晓翠, 刘凤之 (2015). 红光和蓝光对葡萄叶片衰老与活性氧代谢的影响. 园艺学报 42, 1066-1076. |
| [25] | 许大全, 高伟, 阮军 (2015). 光质对植物生长发育的影响. 植物生理学报 51, 1217-1234. |
| [26] | 张欢, 徐志刚, 崔瑾, 谷艾素, 郭银生 (2010). 光质对番茄和莴苣幼苗生长及叶绿体超微结构的影响. 应用生态学报 21, 959-965. |
| [27] | 郑洁, 胡美君, 郭延平 (2008). 光质对植物光合作用的调控及其机理. 应用生态学报 19, 1619-1624. |
| [28] | 周成波, 张旭, 刘彬彬, 艾希珍, 毕焕改, 李清明 (2015). 补光光质对叶用莴苣光合特性的影响. 植物生理学报 51, 2255-2262. |
| [29] | Kara AN, Kotov AA, Bukhov NG (1997). Specific distribution of gibberellins, cytokinins, indole-3-acetic acid, and abscisic acid in radish plants closely correlates with photomorphogenetic responses to blue or red light.J Plant Physiol 151, 51-59. |
| [30] | McLachlan DH, Lan J, Geilfus CM, Dodd AN, Larson T, Baker A, Hõrak H, Kollist H, He ZS, Graham I, Mickelbart MV, Hetherington AM (2016). The breakdown of stored triacylglycerols is required during light-induced stomatal opening.Curr Biol 26, 707-712. |
| [31] | Muneer S, Kim EJ, Park JS, Lee JH (2014). Influence of green, red and blue light emitting diodes on multiprotein complex proteins and photosynthetic activity under different light intensities in lettuce leaves (Lactuca sativa L.). Int J Mol Sci 15, 4657-4670. |
| [32] | Zhang DY, Wang XH, Chen Y, Xu DQ (2005). Determinant of photosynthetic capacity in rice leaves under ambient air conditions.Photosynthetica 43, 273-276. |
| [1] | SHANGGUAN Yao-Yao, SU Shi-Ping, GU Xue-Dan, ZHANG Zheng-Zhong, ZHAO Hu, LI Yi, WEI Xing-Yu. Response of Reaumuria songorica seedlings to photoperiod and light quality ratio [J]. Chin J Plant Ecol, 2025, 49(5): 788-800. |
| [2] | Xuanwen Mao, Zhichao Wang, Xinyi Ruan, Jingfei Sun, Yating Zhang, Jinhao Lu, Tiantian Shao, Xian Wang, Jiamin Xiao, Li Xiao, Mengyao Ye, Yuhuan Wu, Peng Liu. Regulatory Effects of Exogenous Organic Acids on the Physiological Responses of Helianthus tuberosus Under Aluminium Stress [J]. Chinese Bulletin of Botany, 2023, 58(4): 573-589. |
| [3] | Li Chen, Liu Jianting, Fan Yongxin, Zhao Xuehui, Xiao Wei, Chen Xiude, Fu Xiling, Li Ling, Li Dongmei. Effects of UV-B on Photosynthetic Function and Chloroplast Ultrastructure of Peach Leaves Grown in Greenhouse [J]. Chinese Bulletin of Botany, 2022, 57(4): 434-443. |
| [4] | Qingping Zhao,Shifan Ma,Ruixi Li,Tianyu Wang,Xiang Zhao. Advances of NPH3/RPT2-Like (NRL) Family Proteins in Phototropin-mediated Signaling in Arabidopsis thaliana [J]. Chinese Bulletin of Botany, 2020, 55(2): 240-253. |
| [5] | LIU Na-Na,TIAN Qiu-Ying,ZHANG Wen-Hao. Comparison of adaptive strategies to phosphorus-deficient soil between dominant species Artemisia frigida and Stipa krylovii in typical steppe of Nei Mongol [J]. Chin J Plant Ecol, 2014, 38(9): 905-915. |
| [6] | Xiang Zhao, Lindan Wang, Yuanyuan Li, Qingping Zhao, Xiao Zhang. Isolation and Characterization of Regulators Involved in PHOT2-mediated Phototropism of Hypocotyls in Arabidopsis [J]. Chinese Bulletin of Botany, 2014, 49(3): 254-261. |
| [7] | PANG Li, ZHANG Yi, ZHOU Zhi-Chun, FENG Zhong-Ping, CHU De-Yu. Effects of simulated nitrogen deposition on root exudates and phosphorus efficiency in Pinus massoniana families under low phosphorus stress [J]. Chin J Plant Ecol, 2014, 38(1): 27-35. |
| [8] | FU Xiao-Ping, DOU Chang-Ming, HU Shao-Ping, CHEN Xin-Cai, SHI Ji-Yan, CHEN Ying-Xu. A review of progress in roles of organic acids on heavy metal resistance and detoxification in plants [J]. Chin J Plant Ecol, 2010, 34(11): 1354-1358. |
| [9] | Sujuan Gao;Xiuzhi Xie;Zhaoping Chen;Zhigang Huang;Qi Zhao;Xiaojing Wang. Blue Light-mediated De-etiolation in Sorghum Mutant har1 [J]. Chinese Bulletin of Botany, 2009, 44(01): 69-78. |
| [10] | SUN Qin NI Wu-Zhong YANG Xiao-E. Role of Organic Acid in Detoxification of Aluminum in Higher Plants and Its Mechanisms [J]. Chinese Bulletin of Botany, 2002, 19(04): 496-503. |
| [11] | WANG Man WANG Xiao_Jing. Photoreceptors of Ultraviolet Light and Blue Light and Induction of CHS Expression [J]. Chinese Bulletin of Botany, 2002, 19(03): 265-271. |
| [12] | DENG Jiang-Ming CAI Qun-Ying PAN Rui-Chi. Effect of Light Quality on the Contents of Protein and Free Amino Acids in Rice Seedlings [J]. Chinese Bulletin of Botany, 2000, 17(05): 419-423. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||