

Chinese Bulletin of Botany ›› 2018, Vol. 53 ›› Issue (6): 741-744.DOI: 10.11983/CBB18187 cstr: 32102.14.CBB18187
• COMMENTARY • Previous Articles Next Articles
Shen Jinbo1,*( ), Jiang Liwen2,3,*(
), Jiang Liwen2,3,*( )
)
Received:2018-09-02
Online:2018-11-01
Published:2018-12-05
Contact:
Shen Jinbo,Jiang Liwen
Shen Jinbo, Jiang Liwen. Chinese Scientists Make Groundbreaking Discoveries in Plant Cytoskeleton[J]. Chinese Bulletin of Botany, 2018, 53(6): 741-744.
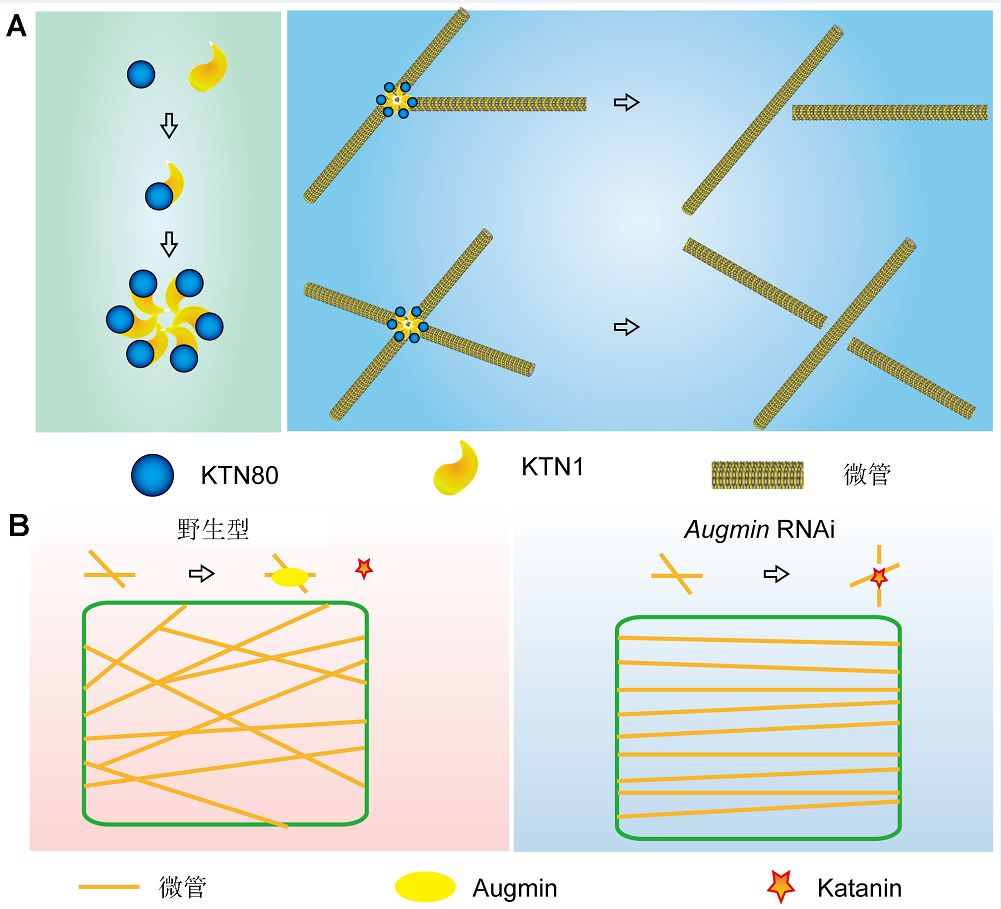
Figure 1 Schematic models of Katanin and Augmin functions on microtubules (MTs) organization in plants (modified from Wang et al., 2017, 2018) (A) The formation of Katanin complexes and MTs severing in Arabidopsis cells. Katanin is composed of a p60 subunit KTN1 and a p80 subunit KTN80, which exists as a KTN1-KTN80 heterodimer in the cytosol (left panel). KTN80 determines the precise targeting of KTN1-KTN80 heterodimers to MTs crossover and branching nucleation sites. KTN1 further triggers the oligomerization of the mixed types of KTN1-KTN80 heterodimers that sense MT geometry to confer precise MT severing (right panel); (B) Augmin functions in regulating microtubule arrays by preventing katanin-mediated microtubule severing and maintaining the stability of crossovers (left panel). In the Augmin knockdown mutant (amiR-AUG6), a significantly higher microtubule severing frequency occurs and a greater proportion of aligned microtubule arrays (right panel) compared to the fine-network microtubule architectures observed in wild-type (left panel).
| [1] |
Ehrhardt DW (2008). Straighten up and fly right: microtubule dynamics and organization of non-centrosomal arrays in higher plants.Curr Opin Cell Biol 20, 107-116.
DOI URL |
| [2] |
Li SD, Bashline L, Zheng YZ, Xin XR, Huang SX, Kong ZS, Kim SH, Cosgrove DJ, Gu Y (2016). Cellulose synthase complexes act in a concerted fashion to synthesize highly aggregated cellulose in secondary cell walls of plants.Proc Natl Acad Sci USA 113, 11348-11353.
DOI URL PMID |
| [3] |
Liu T, Tian J, Wang GD, Yu YJ, Wang CF, Ma YP, Zhang XX, Xia GX, Liu B, Kong ZS (2014). Augmin triggers mic- rotubule-dependent microtubule nucleation in interphase plant cells.Curr Biol 24, 2708-2713.
DOI URL PMID |
| [4] |
McFarlane HE, D$\ddot{o}$ring A, Persson S (2014). The cell biology of cellulose synthesis.Annu Rev Plant Biol 65, 69-94.
DOI URL PMID |
| [5] |
McNally FJ, Vale RD (1993). Identification of katanin, an ATPase that severs and disassembles stable microtu- bules.Cell 75, 419-429.
DOI URL PMID |
| [6] |
Nick P (2013). Microtubules, signaling and abiotic stress.Plant J 75, 309-323.
DOI URL PMID |
| [7] |
Paredez AR, Somerville CR, Ehrhardt DW (2006). Vis- ualization of cellulose synthase demonstrates functional association with microtubules.Science 312, 1491-1495.
DOI URL PMID |
| [8] |
Roll-Mecak A, McNally FJ (2010). Microtubule-severing enzymes.Curr Opin Cell Biol 22, 96-103.
DOI URL |
| [9] |
Sharp DJ, Ross JL (2012). Microtubule-severing enzymes at the cutting edge.J Cell Sci 125, 2561-2569.
DOI URL PMID |
| [10] |
Shaw SL (2013). Reorganization of the plant cortical microtubule array.Curr Opin Plant Biol 16, 693-697.
DOI URL PMID |
| [11] |
Wang CF, Liu WW, Wang GD, Li J, Dong L, Han LB, Wang Q, Tian J, Yu YJ, Gao CX, Kong ZS (2017). KTN80 confers precision to microtubule severing by specific targeting of katanin complexes in plant cells.EMBO J 36, 3435-3447.
DOI URL PMID |
| [12] |
Wang GD, Wang CF, Liu WW, Ma YP, Dong L, Tian J, Yu YJ, Kong ZS (2018). Augmin antagonizes Katanin at microtubule crossovers to control the dynamic organi- zation of plant cortical arrays.Curr Biol 28, 1311-1317.
DOI URL PMID |
| [13] |
Wasteneys GO, Ambrose JC (2009). Spatial organization of plant cortical microtubules: close encounters of the 2D kind.Trends Cell Biol 19, 62-71.
DOI URL PMID |
| [14] |
Watanabe Y, Meents MJ, McDonnell LM, Barkwill S, Sampathkumar A, Cartwright HN, Demura T, Ehrhardt DW, Samuels AL, Mansfield SD (2015). Visualization of cellulose synthases in Arabidopsis secondary cell walls.Science 350, 198-203.
DOI URL PMID |
| [1] | Yunhui Wang, Yifan Wang, Jiayu Lin, Jinhong Li, Shien Yao, Xiangchi Feng, Zhenlin Cao, Jun Wang, Meina Li. Plant Kinesin: from Microtubule Arrays to Physiological Regulation [J]. Chinese Bulletin of Botany, 2022, 57(3): 358-374. |
| [2] | Jianru Yue, Yunjian He, Tianqi Qiu, Nannan Guo, Xueping Han, Xianling Wang. Research Advances in the Molecular Mechanisms of Plant Microtubules in Regulating Hypocotyl Elongation [J]. Chinese Bulletin of Botany, 2021, 56(3): 363-371. |
| [3] | Yajing Wang,Xinying Zhang,Guirong Huang,Xiaoying Liu,Rui Guo,Fengxue Gu,Xiuli Zhong,Xurong Mei. Characteristics of Phosphatidic Acid and the Underlying Mechanisms of ABA-induced Stomatal Movement in Plants [J]. Chinese Bulletin of Botany, 2019, 54(2): 245-154. |
| [4] | Lingfei Li, Jianzong Peng, Xiaojing Wang. Preliminary Functional Analysis of Microtubule-associated Protein GMAP65-1 from Gerbera hybrida [J]. Chinese Bulletin of Botany, 2015, 50(1): 12-21. |
| [5] | Zhigang Li;Xincheng Zhang;Li Lin;Suli Li;Litao Yang;Yangrui Li . Microtubule Cytoskeleton Change During Mitosis in the Shoot Apex of Sugarcane [J]. Chinese Bulletin of Botany, 2008, 25(03): 276-283. |
| [6] | Xiaohua Wang;Huaiqing Hao;Qinli Wang;Maozhong Zheng;Jinxing Lin. Structure of the Pollen Tube and the Mechanism of Tip Growth [J]. Chinese Bulletin of Botany, 2007, 24(03): 340-354. |
| [7] | HE Qun;YOU Rui-Lin. Steedman's Wax Sectioning for Observation of the Microtubule Cytoskeleton in Plant Cells [J]. Chinese Bulletin of Botany, 2004, 21(05): 547-555. |
| [8] | LIU Ning LIU Jian-Wu. The Studies of the Locomotory Apparatus and Cytoskeleton in the Spermatozoid of Ferns [J]. Chinese Bulletin of Botany, 2004, 21(02): 164-171. |
| [9] | HE Xin-Qiang CUI Ke-Ming. Progress in Study of Secondary Wall Formation in Plants [J]. Chinese Bulletin of Botany, 2002, 19(05): 513-522. |
| [10] | HOU Lei-PingLI Mei-Lan. Progress of Studies on the Plant Growth Promoting Mechanism of Brassinolide (BR) [J]. Chinese Bulletin of Botany, 2001, 18(05): 560-566. |
| [11] | CAl Xue. The Dynamics and Significance of Cytoskeletons in Pollen Formation of Angiosperms [J]. Chinese Bulletin of Botany, 1999, 16(04): 339-344. |
| [12] | YU Rong;YUAN Ming and WANG Xue-Chen. Dynamics and Regulation of Microtubule System [J]. Chinese Bulletin of Botany, 1998, 15(06): 19-29. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||