

Chinese Bulletin of Botany ›› 2017, Vol. 52 ›› Issue (2): 128-147.DOI: 10.11983/CBB16001
Previous Articles Next Articles
Lili Qiu1,2, Qi Zhao1,2, Yuhong Zhang1*, Shaojun Dai2*
Received:2016-01-05
Accepted:2016-06-14
Online:2017-03-01
Published:2017-04-05
Contact:
Zhang Yuhong,Dai Shaojun
About author:# Co-first authors
Lili Qiu, Qi Zhao, Yuhong Zhang, Shaojun Dai. Research Advances in Stress-responsive Plant Plasma Membrane Proteomic[J]. Chinese Bulletin of Botany, 2017, 52(2): 128-147.
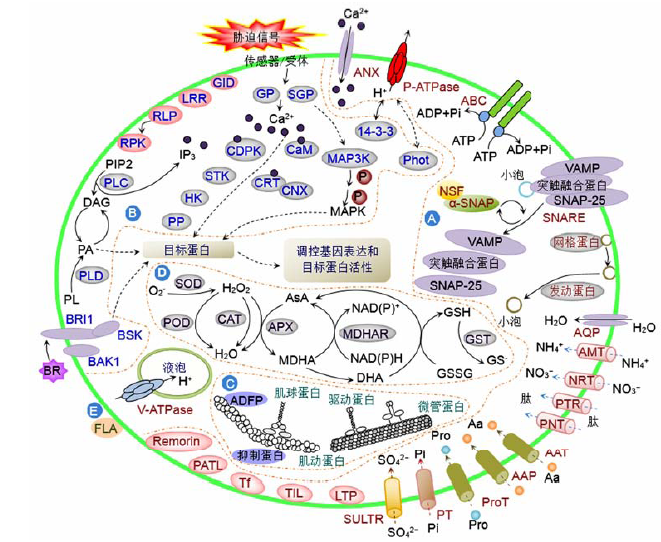
Figure 1 The stress-responsive metabolic pathway in plant plasma membranes by proteomics researches(A) Membrane and transporting proteins; (B) Signaling; (C) Cytoskeleton; (D) ROS scavenging; (E) Cell wall related proteins. Aa: Amino acid; AAP: Amino acid permease; AAT: Amino acid transporter; ABC: ATP-binding cassette transporter; ADFP: Actin depolymerizing factor-like protein; ADP: Adenosine diphosphate; AMT: Ammonium transporter; ANX: Annexin; APX: Ascorbate peroxidase; AQP: Aquaporin; AsA: Ascorbate; ATP: Adenosine triphosphate; BAK1: Brassinosteroid insensitive1-associated receptor kinase 1; BR: Brassinosteroids; BRI1: Brassinosteroid insensitive1; BSK: Brassinosteroid-signaling kinase; CaM: Calmodulin; CAT: Catalase; CDPK: Calcium-dependent protein kinase; CNX: Calnexin; CRT: Calreticulin; DAG: Diacylglycerol; DHA: Dehydroascorbate; FLA: Fasciclin-like arabinogalactan protein; GID: Gibberellin receptor GID1L2; GP: GTP binding protein; GSH: Reduced glutathione; GSSG: Oxidized glutathione; GST: Glutathione S-transferase; HK: Histidine kinase; IP3: Inositol-1,4,5-trisphosphate; LRR: Leucine-rich repeat protein; LTP: Lipid transfer protein; MAP3K: Mitogen activated protein kinase kinase kinase; MAPK: Mitogen-activated protein kinase; MDHA: Monodehydroascorbate; MDHAR: Monodehydroascorbate reductase; NADP+/NADPH: Nicotinamide adenine dinucleotide phosphate; NRT: Nitrate transporter; NSF: N-ethylmaleimide sensitive factor; PA: Phosphatidic acid; PATL: Patellin; P-ATPase: Plasma membrane H+-ATPase; Phot: Phototropin; PIP2: Phosphatidylinositol-4,5-bisphosphate; PL: Phospholipid; PLC: Phospholipase C; PLD: Phospholipase D; PNT: Peptide/nitrate transporter; POD: Peroxidase; PP: Protein phosphatase; Pro: Proline; ProT: Proline transporter; PT: Phosphate transporter; PTR: Peptide transporter; RLP: Receptor like protein; RPK: Receptor protein kinase; SGP: Small GTP binding protein; SNAP-25: Synaptosomal-associated protein of 25 kDa; SNARE: N-ethylmaleimide sensitive factor attachment protein receptor; SOD: Superoxide dismutase; STK: Serine/threonine kinase; SULTR: Sulphate transporter; Tf: Transferrin; TIL: Temperature-induced lipocalin; VAMP: Vesicle-associated membrane protein; V-ATPase: Vacuolar H+-ATPase; α-SNAP: Alpha-soluble N-ethyl- maleimide sensitive factor attachment protein
| [1] | 范玲玲, 陈刚, 陈义芳, 周卫东, 戴绍军, 孙国荣 (2010). NaHCO3胁迫下星星草根中Ca2+与Ca2+-ATPase的超微细胞化学定位. 植物学报 45, 337-344. |
| [2] | 林植芳, 刘楠 (2012). 活性氧调控植物生长发育的研究进展. 植物学报 47, 74-86. |
| [3] | 孙颖, 孙大业 (2006). 植物细胞质外体多肽与蛋白研究. 植物学通报 23, 578-583. |
| [4] | Alexandersson E, Saalbach G, Larsson C, Kjellbom P (2004). Arabidopsis plasma membrane proteomics identifies components of transport, signal transduction and membrane trafficking.Plant Cell Physiol 45, 1543-1556. |
| [5] | Baisakh N, RamanaRao MV, Rajasekaran K, Subudhi P, Janda J, Galbraith D, Vanier C, Pereira A (2012). Enhanced salt stress tolerance of rice plants expressing a vacuolar H+-ATPase subunit c1 (SaVHAc1) gene from the halophyte grass Spartina alterniflora Löisel.Plant Biotechnol J 10, 453-464. |
| [6] | Bariola PA, Retelska D, Stasiak A, Kammerer RA, Fleming A, Hijri M, Frank S, Farmer EE (2004). Remorins form a novel family of coiled coil-forming oligomeric and filamentous proteins associated with apical, vascular and embryonic tissues in plants.Plant Mol Biol 55, 579-594. |
| [7] | Bloom AJ, Sukrapanna SS, Warner RL (1992). Root respiration associated with ammonium and nitrate absorption and assimilation by barley.Plant Physiol 99, 1294-1301. |
| [8] | Boman AL, Kahn RA (1995). Arf proteins: the membrane traffic police?Trends Biochem Sci 20, 147-150. |
| [9] | Bunney TD, van den Wijngaard PW, de Boer AH (2002). 14-3-3 protein regulation of proton pumps and ion channels.Plant Mol Biol 50, 1041-1051. |
| [10] | Cameron KD, Teece MA, Smart LB (2006). Increased accumulation of cuticular wax and expression of lipid transfer protein in response to periodic drying events in leaves of tree tobacco.Plant Physiol 140, 176-183. |
| [11] | Charron JBF, Breton G, Badawi M, Sarhan F (2002). Molecular and structural analyses of a novel temperature stress-induced lipocalin from wheat and Arabidopsis.FEBS Lett 517, 129-132. |
| [12] | Charron JBF, Ouellet F, Pelletier M, Danyluk J, Chauve C, Sarhan F (2005). Identification, expression, and evolutionary analyses of plant lipocalins.Plant Physiol 139, 2017-2028. |
| [13] | Checker VG, Khurana P (2013). Molecular and functional characterization of mulberry EST encoding remorin (Mi- REM) involved in abiotic stress.Plant Cell Rep 32, 1729-1741. |
| [14] | Chen F, Li Q, He Z (2007a). Proteomic analysis of rice plasma membrane-associated proteins in response to chitooligosaccharide elicitors.J Integr Plant Biol 49, 863-870. |
| [15] | Chen F, Yuan Y, Li Q, He Z (2007b). Proteomic analysis of rice plasma membrane reveals proteins involved in early defense response to bacterial blight.Proteomics 7, 1529-1539. |
| [16] | Cheng Y, Qi Y, Zhu Q, Chen X, Wang N, Zhao X, Chen H, Cui X, Xu L, Zhang W (2009). New changes in the plasma-membrane-associated proteome of rice roots un- der salt stress.Proteomics 9, 3100-3114. |
| [17] | Chu B, Snustad DP, Carter JV (1993). Alteration of β-tubulin gene expression during low-temperature exposure in leaves of Arabidopsis thaliana.Plant Physiol 103, 371-377. |
| [18] | Clouse SD (2011). Brassinosteroid signal transduction: from receptor kinase activation to transcriptional networks regu- lating plant development.Plant Cell 23, 1219-1230. |
| [19] | De Lorenzo L, Merchan F, Laporte P, Thompson R, Clarke J, Sousa C, Crespi M (2009). A novel plant leucine-rich repeat receptor kinase regulates the response of Medicago truncatula roots to salt stress.Plant Cell 21, 668-680. |
| [20] | DeFalco TA, Bender KW, Snedden WA (2010). Breaking the code: Ca2+ sensors in plant signaling.Biochem J 425, 27-40. |
| [21] | Drøbak BK, Franklin-Tong VE, Staiger CJ (2004). The role of the actin cytoskeleton in plant cell signaling.New Phytol 163, 13-30. |
| [22] | Ellard-Ivey M, Hopkins RB, White TJ, Lomax TL (1999). Cloning, expression and N-terminal myristoylation of Cp- CPK1, a calcium-dependent protein kinase from zucchini (Cucurbita pepo L.).Plant Mol Biol 39, 199-208. |
| [23] | Ellis C, Karafyllidis I, Wasternack C, Turner JG (2002). The Arabidopsis mutant cev1 links cell wall signaling to jasmonate and ethylene responses.Plant Cell 14, 1557-1566. |
| [24] | Fisher M, Gokhman I, Pick U, Zamir A (1997). A structu- rally novel transferrin-like protein accumulates in the plasma membrane of the unicellular green alga Dunaliella salina grown in high salinities.J Biol Chem 272, 1565-1570. |
| [25] | Fukuda A, Chiba K, Maeda M, Nakamura A, Maeshima M, Tanaka Y (2004). Effect of salt and osmotic stresses on the expression of genes for the vacuolar H+-pyropho- sphatase, H+-ATPase subunit A, and Na+/H+ antiporter from barley.J Exp Bot 55, 585-594. |
| [26] | Fulgosi H, Soll J, de Faria Maraschin S, Korthout HA, Wang M, Testerink C (2002). 14-3-3 proteins and plant development.Plant Mol Biol 50, 1019-1029. |
| [27] | Galen C, Rabenold JJ, Liscum E (2007). Functional eco- logy of a blue light photoreceptor: effects of phototropin-1 on root growth enhance drought tolerance in Arabidopsis thaliana.New Phytol 173, 91-99. |
| [28] | Gaxiola RA, Palmgren MG, Schumacher K (2007). Plant proton pumps.FEBS Lett 581, 2204-2214. |
| [29] | Gazzarrini S, Lejay L, Gojon A, Ninnemann O, Frommer WB, von Wirén N (1999). Three functional transporters for constitutive, diurnally regulated, and starvation-induc- ed uptake of ammonium into Arabidopsis roots.Plant Cell 11, 937-947. |
| [30] | Gens JS, Fujiki M, Pickard BG (2000). Arabinogalactan- protein and wall-associated kinase in a plasmalemmal reticulum with specialized vertices.Protoplasma 212, 115-134. |
| [31] | Gerke V, Moss SE (2002). Annexins: from structure to function.Physiol Rev 82, 331-371. |
| [32] | Gómez JM, Jiménez A, Olmos E, Sevilla F (2004). Loca- tion and effects of long-term NaCl stress on superoxide dismutase and ascorbate peroxidase isoenzymes of pea (Pisum sativum cv. ‘Puget’) chloroplasts.J Exp Bot 55, 119-130. |
| [33] | Han B, Chen S, Dai S, Yang N, Wang T (2010). Isobaric tags for relative and absolute quantification-based comparative proteomics reveals the features of plasma membrane-associated proteomes of pollen grains and pollen tubes from Lilium davidii.J Integr Plant Biol 52, 1043-1058. |
| [34] | Harris TJ, Siu CH (2002). Reciprocal raft-receptor inter- actions and the assembly of adhesion complexes.Bioessays 24, 996-1003. |
| [35] | Hashimoto M, Toorchi M, Matsushita K, Iwasaki Y, Komatsu S (2009). Proteome analysis of rice root plasma membrane and detection of cold stress responsive proteins.Protein Pept Lett 16, 685-697. |
| [36] | Hinshaw JE (2000). Dynamin and its role in membrane fission.Annu Rev Cell Dev Biol 16, 483-519. |
| [37] | Hoeflich KP, Ikura M (2002). Calmodulin in action: diversity in target recognition and activation mechanisms.Cell 108, 739-742. |
| [38] | Holweg C, Nick P (2004). Arabidopsis myosin XI mutant is defective in organelle movement and polar auxin transport.Proc Natl Acad Sci USA 101, 10488-10493. |
| [39] | Hong W (2005). SNAREs and traffic.BBA-Mol Cell Res 1744, 120-144. |
| [40] | Hong Z, Geisler-Lee CJ, Zhang Z, Verma DPS (2003). Phragmoplastin dynamics: multiple forms, microtubule association and their roles in cell plate formation in plants.Plant Mol Biol 53, 297-312. |
| [41] | Hopff D, Wienkoop S, Lüthje S (2013). The plasma membrane proteome of maize roots grown under low and high iron conditions.J Proteomics 91, 605-618. |
| [42] | Howitt SM, Udvardi MK (2000). Structure, function and regulation of ammonium transporters in plants.BBA-Bio- membranes 1465, 152-170. |
| [43] | Huang F, Fulda S, Hagemann M, Norling B (2006). Proteomic screening of salt-stress-induced changes in plas- ma membranes of Synechocystis sp. strain PCC 6803.Proteomics 6, 910-920. |
| [44] | Hunt L, Otterhag L, Lee JC, Lasheen T, Hunt J, Seki M, Shinozaki K, Sommarin M, Gilmour DJ, Pical C, Gray JE (2004). Gene-specific expression and calcium activation of Arabidopsis thaliana phospholipase C isoforms.New Phytol 162, 643-654. |
| [45] | Hwang I, Sze H, Harper JF (2000). A calcium-dependent protein kinase can inhibit a calmodulin-stimulated Ca2+ pump (ACA2) located in the endoplasmic reticulum of Arabidopsis.Proc Natl Acad Sci USA 97, 6224-6229. |
| [46] | Jamet E, Canut H, Boudart G, Pont-Lezica RF (2006). Cell wall proteins: a new insight through proteomics.Trends Plant Sci 11, 33-39. |
| [47] | Jang JY, Kim DG, Kim YO, Kim JS, Kang H (2004). An expression analysis of a gene family encoding plasma membrane aquaporins in response to abiotic stresses in Arabidopsis thaliana.Plant Mol Biol 54, 713-725. |
| [48] | Johnson KL, Jones BJ, Bacic A, Schultz CJ (2003). The fasciclin-like arabinogalactan proteins of Arabidopsis. A multigene family of putative cell adhesion molecules.Pl- ant Physiol 133, 1911-1925. |
| [49] | Jones AM, Assmann SM (2004). Plants: the latest model system for G-protein research.EMBO Rep 5, 572-578. |
| [50] | Kader JC (1996). Lipid-transfer proteins in plants.Annu Rev Plant Biol 47, 627-654. |
| [51] | Kalampanayil BD, Wimmers LE (2001). Identification and characterization of a salt-stress-induced plasma membrane H+-ATPase in tomato.Plant Cell Environ 24, 999-1005. |
| [52] | Karpinska B, Karlsson M, Schinkel H, Streller S, Süss KH, Melzer M, Wingsle G (2001). A novel superoxide dismutase with a high isoelectric point in higher plants. Expression, regulation, and protein localization.Plant Phy- siol 126, 1668-1677. |
| [53] | Katz A, Waridel P, Shevchenko A, Pick U (2007). Salt- induced changes in the plasma membrane proteome of the halotolerant alga Dunaliella salina as revealed by blue native gel electrophoresis and nano-LC-MS/MS analysis.Mol Cell Proteomics 6, 1459-1472. |
| [54] | Kawamura Y, Uemura M (2003). Mass spectrometric approach for identifying putative plasma membrane proteins of Arabidopsis leaves associated with cold acclimation.Plant J 36, 141-154. |
| [55] | Komatsu S, Wada T, Abaléa Y, Nouri MZ, Nanjo Y, Nakayama N, Shimamura S, Yamamoto R, Nakamura T, Furukawa K (2009). Analysis of plasma membrane proteome in soybean and application to flooding stress response.J Proteome Res 8, 4487-4499. |
| [56] | Leborgne-Castel N, Lherminier J, Der C, Fromentin J, Houot V, Simon-Plas F (2008). The plant defense elicitor cryptogein stimulates clathrin-mediated endocytosis correlated with reactive oxygen species production in bright yellow-2 tobacco cells.Plant Physiol 146, 1255-1266. |
| [57] | Lee J, Rudd JJ (2002). Calcium-dependent protein kinases: versatile plant signaling components necessary for patho- gen defence.Trends Plant Sci 7, 97-98. |
| [58] | Leustek T, Martin MN, Bick JA, Davies JP (2000). Pathways and regulation of sulfur metabolism revealed throu- gh molecular and genetic studies.Annu Rev Plant Physiol 51, 141-165. |
| [59] | Leustek T, Saito K (1999). Sulfate transport and assimila- tion in plants.Plant Physiol 120, 637-644. |
| [60] | Li B, Takahashi D, Kawamura Y, Uemura M (2012a). Comparison of plasma membrane proteomic changes of Arabidopsis suspension-cultured cells (T87 Line) after cold and ABA treatment in association with freezing tole- rance development.Plant Cell Physiol 53, 543-554. |
| [61] | Li J, Wen J, Lease KA, Doke JT, Tax FE, Walker JC (2002). BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling.Cell 110, 213-222. |
| [62] | Li W, Li M, Zhang W, Welti R, Wang X (2004). The plasma membrane-bound phospholipase Dδ enhances freezing tolerance in Arabidopsis thaliana.Nat Biotechnol 22, 427-433. |
| [63] | Li ZY, Xu ZS, He GY, Yang GX, Chen M, Li LC, Ma YZ (2012b). A mutation in Arabidopsis BSK5 encoding a bras- sinosteroid-signaling kinase protein affects responses to salinity and abscisic acid.Biochem Biophs Res Commun 426, 522-527. |
| [64] | Lin W (1981). Inhibition of anion transport in corn root protoplasts.Plant Physiol 68, 435-438. |
| [65] | Liu X, Bush DR (2006). Expression and transcriptional regulation of amino acid transporters in plants.Amino Acids 30, 113-120. |
| [66] | Ma Y, Dai X, Xu Y, Luo W, Zheng X, Zeng D, Pan Y, Lin X, Liu H, Zhang D, Xiao J, Guo X, Xu S, Niu Y, Jin J, Zhang H, Xu X, Li L, Wang W, Qian Q, Ge S, Chong K (2015). COLD1 confers chilling tolerance in rice.Cell 160, 1209-1221. |
| [67] | Magnotta SM, Gogarten JP (2002). Multi site polyadenyla- tion and transcriptional response to stress of a vacuolar type H+-ATPase subunit A gene in Arabidopsis thaliana.BMC Plant Biol 2, 1-13. |
| [68] | Malakshah SN, Rezaei MH, Heidari M, Salekdeh GH (2007). Proteomics reveals new salt responsive proteins associated with rice plasma membrane.Biosci Biotechnol Biochem 71, 2144-2154. |
| [69] | Martín ML, Busconi L (2000). Membrane localization of a rice calcium-dependent protein kinase (CDPK) is mediated by myristoylation and palmitoylation.Plant J 24, 429-435. |
| [70] | Martín ML, Busconi L (2001). A rice membrane-bound calcium-dependent protein kinase is activated in response to low temperature.Plant Physiol 125, 1442-1449. |
| [71] | Maruyama-Nakashita A, Inoue E, Watanabe-Takahashi A, Yamaya T, Takahashi H (2003). Transcriptome profiling of sulfur-responsive genes in Arabidopsis reveals global effects of sulfur nutrition on multiple metabolic pa- thways.Plant Physiol 132, 597-605. |
| [72] | Meisrimler CN, Planchon S, Renaut J, Sergeant K, Lüthje S (2011). Alteration of plasma membrane-bound redox systems of iron deficient pea roots by chitosan.J Proteomics 74, 1437-1449. |
| [73] | Michelet B, Boutry M (1995). The plasma membrane H+-ATPase (a highly regulated enzyme with multiple phy- siological functions).Plant Physiol 108, 1-6. |
| [74] | Mika A, Lüthje S (2003). Properties of guaiacol peroxidase activities isolated from corn root plasma membranes.Pl- ant Physiol 132, 1489-1498. |
| [75] | Mills LN, Hunt L, Leckie CP, Aitken FL, Wentworth M, McAinsh MR, Gray JE, Hetherington AM (2004). The effects of manipulating phospholipase C on guard cell ABA-signaling.J Exp Bot 55, 199-204. |
| [76] | Minami A, Fujiwara M, Furuto A, Fukao Y, Yamashita T, Kamo M, Kawamura Y, Uemura M (2009). Alterations in detergent-resistant plasma membrane microdomains in Arabidopsis thaliana during cold acclimation.Plant Cell Physiol 50, 341-359. |
| [77] | Mishra NS, Tuteja R, Tuteja N (2006). Signaling through MAP kinase networks in plants.Arch Biochem Biophys 452, 55-68. |
| [78] | Mitsukawa N, Okumura S, Shirano Y, Sato S, Kato T, Harashima S, Shibata D (1997). Overexpression of an Arabidopsis thaliana high-affinity phosphate transporter gene in tobacco cultured cells enhances cell growth under phosphate-limited conditions.Proc Natl Acad Sci USA 94, 7098-7102. |
| [79] | Mongrand S, Stanislas T, Bayer EM, Lherminier J, Simon-Plas F (2010). Membrane rafts in plant cells.Tr- ends Plant Sci 15, 656-663. |
| [80] | Morgan A, Dimaline R, Burgoyne RD (1994). The ATPase activity of N-ethylmaleimide-sensitive fusion protein (NSF) is regulated by soluble NSF attachment proteins. J Biol Chem 269, 29347-29350. |
| [81] | Mousley CJ, Tyeryar KR, Vincent-Pope P, Bankaitis VA (2007). The Sec14-superfamily and the regulatory inter- face between phospholipid metabolism and membrane trafficking.Biochim Biophys Acta 1771, 727-736. |
| [82] | Moustafa K, AbuQamar S, Jarrar M, Al-Rajab AJ, Trémouillaux-Guiller J (2014). MAPK cascades and major abiotic stresses.Plant Cell Rep 33, 1217-1225. |
| [83] | Munnik T (2001). Phosphatidic acid: an emerging plant lipid second messenger.Trends Plant Sci 6, 227-233. |
| [84] | Nam KH, Li J (2002). BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling.Cell 110, 203-212. |
| [85] | Nie L, Feng J, Fan P, Chen X, Guo J, Lv S, Bao H, Jia W, Tai F, Jiang P, Wang J, Li Y (2015). Comparative pro- teomics of root plasma membrane proteins reveals the involvement of calcium signaling in NaCl-facilitated nitrate uptake in Salicornia europaea.J Exp Bot 66, 4497-4510. |
| [86] | No EG, Loopstra CA (2000). Hormonal and developmental regulation of two arabinogalactan-proteins in xylem of loblolly pine (Pinus taeda).Physiol Plantarum 110, 524-529. |
| [87] | Nouri MZ, Komatsu S (2010). Comparative analysis of soybean plasma membrane proteins under osmotic stress using gel-based and LC MS/MS-based proteomics approaches.Proteomics 10, 1930-1945. |
| [88] | Örvar BL, Sangwan V, Omann F, Dhindsa RS (2000). Early steps in cold sensing by plant cells: the role of actin cytoskeleton and membrane fluidity.Plant J 23, 785-794. |
| [89] | Palmgren MG (2001). Plant plasma membrane H+-ATP- ases: powerhouses for nutrient uptake.Annu Rev Plant Physiol 52, 817-845. |
| [90] | Perfus-Barbeoch L, Jones AM, Assmann SM (2004). Plant heterotrimeric G protein function: insights from Arabidopsis and rice mutants.Curr Opin Plant Biol 7, 719-731. |
| [91] | Peskan T, Westermann M, Oelmüller R (2000). Identifi- cation of low-density Triton X-100-insoluble plasma mem- brane microdomains in higher plants.Eur J Biochem 267, 6989-6995. |
| [92] | Pokorná J, Schwarzerová K, Zelenková S, Petrášek J, Janotová I, Čapková V, Opatrný Z (2004). Sites of actin filament initiation and reorganization in cold-treated tobacco cells.Plant Cell Environ 27, 641-653. |
| [93] | Popova OV, Dietz KJ, Golldack D (2003). Salt-dependent expression of a nitrate transporter and two amino acid transporter genes in Mesembryanthemum crystallinum.Plant Mol Biol 52, 569-578. |
| [94] | Qiu QS, Zhang N (2000). Water stress inhibits p-nitrophenyl phosphate hydrolysis activity of the plasma membrane H+-ATPase from soybean hypocotyls.Aust J Plant Phy- siol 27, 717-721. |
| [95] | Raghothama KG (2000). Phosphate transport and signaling.Curr Opin Plant Biol 3, 182-187. |
| [96] | Randall SK, Sze H (1986). Properties of the partially purified tonoplast H+-pumping ATPase from oat roots.J Biol Chem 261, 1364-1371. |
| [97] | Rentsch D, Hirner B, Schmelzer E, Frommer WB (1996). Salt stress-induced proline transporters and salt stress- repressed broad specificity amino acid permeases identified by suppression of a yeast amino acid permease- targeting mutant.Plant Cell 8, 1437-1446. |
| [98] | Roberts MR (2003). 14-3-3 proteins find new partners in plant cell signaling.Trends Plant Sci 8, 218-223. |
| [99] | Roxas VP, Lodhi SA, Garrett DK, Mahan JR, Allen RD (2000). Stress tolerance in transgenic tobacco seedlings that overexpress glutathione S-transferase/glutathione per- oxidase.Plant Cell Physiol 41, 1229-1234. |
| [100] | Saijo Y, Hata S, Kyozuka J, Shimamoto K, Izui K (2000). Over-expression of a single Ca2+-dependent protein kin- ase confers both cold and salt/drought tolerance on rice plants.Plant J 23, 319-327. |
| [101] | Sasayama D, Matsuoka D, Oka M, Shitamichi N, Furuya T, Azuma T, Itoh K, Nanmori T (2011). MAP3Kδ4, an Arabidopsis Raf-like MAP3K, regulates plant growth and shoot branching.Plant Biotechnol 28, 463-470. |
| [102] | Schmidt SM, Panstruga R (2007). Cytoskeleton functions in plant-microbe interactions.Physiol Mol Plant Pathol 71, 135-148. |
| [103] | Schwarz M, Zamir A, Pick U (2003). Iron-binding properties of TTf, a salt-induced transferrin from the alga Dunaliella salina.J Plant Nutr 26, 2081-2091. |
| [104] | Seo HM, Jung Y, Song S, Kim Y, Kwon T, Kim DH, Jeung SJ, Yi YB, Yi G, Nam MH, Nam J (2008). Increased expression of OsPT1, a high-affinity phosphate transporter, enhances phosphate acquisition in rice.Biotechnol Lett 30, 1833-1838. |
| [105] | Shibagaki N, Rose A, McDermott JP, Fujiwara T, Hayashi H, Yoneyama T, Davies JP (2002). Selenate-resistant mutants of Arabidopsis thaliana identify Sultr1; 2, a sulfate transporter required for efficient transport of sulfate into roots.Plant J 29, 475-486. |
| [106] | Shibaoka H (1994). Plant hormone-induced changes in the orientation of cortical microtubules: alterations in the cross- linking between microtubules and the plasma membrane.Annu Rev Plant Physiol 45, 527-544. |
| [107] | Shitamichi N, Matsuoka D, Sasayama D, Furuya T, Nanmori T (2013). Over-expression of MAP3Kδ4, an ABA-inducible Raf-like MAP3K that confers salt tolerance in Arabidopsis.Plant Biotechnol 30, 111-118. |
| [108] | Söllner T, Whiteheart SW, Brunner M, Erdjument-Bro- mage H, Geromanos S, Tempst P, Rothman JE (1993). SNAP receptors implicated in vesicle targeting and fusion.Nature 362, 318-324. |
| [109] | Song W, Steiner HY, Zhang L, Naider F, Stacey G, Becker JM (1996). Cloning of a second Arabidopsis peptide tran- sport gene.Plant Physiol 110, 171-178. |
| [110] | Stacey G, Koh S, Granger C, Becker JM (2002). Peptide transport in plants.Trends Plant Sci 7, 257-263. |
| [111] | Surowy TK, Boyer JS (1991). Low water potentials affect expression of genes encoding vegetative storage proteins and plasma membrane proton ATPase in soybean.Plant Mol Biol 16, 251-262. |
| [112] | Sutton RB, Fasshauer D, Jahn R, Brunger AT (1998). Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 Å resolution.Nature 395, 347-353. |
| [113] | Takahashi D, Kawamura Y, Uemura M (2013). Changes of detergent-resistant plasma membrane proteins in oat and rye during cold acclimation: association with differential freezing tolerance.J Proteome Res 12, 4998-5011. |
| [114] | Tang D, Ade J, Frye CA, Innes RW (2006). A mutation in the GTP hydrolysis site of Arabidopsis dynamin-related protein 1E confers enhanced cell death in response to powdery mildew infection.Plant J 47, 75-84. |
| [115] | Testerink C, Munnik T (2005). Phosphatidic acid: a multifunctional stress signaling lipid in plants.Trends Plant Sci 10, 368-375. |
| [116] | Tuteja N, Sopory SK (2008). Chemical signaling under abiotic stress environment in plants.Plant Signal Behav 3, 525-536. |
| [117] | Ueda A, Shi W, Sanmiya K, Shono M, Takabe T (2001). Functional analysis of salt-inducible proline transporter of barley roots.Plant Cell Physiol 42, 1282-1289. |
| [118] | Ueno K, Kinoshita T, Inoue SI, Emi T, Shimazaki KI (2005). Biochemical characterization of plasma memb- rane H+-ATPase activation in guard cell protoplasts of Ara- bidopsis thaliana in response to blue light.Plant Cell Phy- siol 46, 955-963. |
| [119] | Vergnolle C, Vaultier MN, Taconnat L, Renou JP, Kader JC, Zachowski A, Ruelland E (2005). The cold-induced early activation of phospholipase C and D pathways determines the response of two distinct clusters of genes in Arabidopsis cell suspensions.Plant Physiol 139, 1217-1233. |
| [120] | Vogel JP, Raab TK, Somerville CR, Somerville SC (2004). Mutations in PMR5 result in powdery mildew resistance and altered cell wall composition.Plant J 40, 968-978. |
| [121] | Von Wittgenstein NJ, Le CH, Hawkins BJ, Ehlting J (2014). Evolutionary classification of ammonium, nitrate, and peptide transporters in land plants.BMC Evol Biol 14, 11. |
| [122] | Wang X, Li X, Meisenhelder J, Hunter T, Yoshida S, Asami T, Chory J (2005). Autoregulation and homodi- merization are involved in the activation of the plant ste- roid receptor BRI1.Dev Cell 8, 855-865. |
| [123] | Wasteneys GO, Yang Z (2004). New views on the plant cytoskeleton.Plant Physiol 136, 3884-3891. |
| [124] | White PJ, Bowen HC, Demidchik V, Nichols C, Davies JM (2002). Genes for calcium-permeable channels in the plasma membrane of plant root cells.BBA-Biomembranes 1564, 299-309. |
| [125] | Xiong L, Schumaker KS, Zhu JK (2002). Cell signaling during cold, drought, and salt stress.Plant Cell 14, S165-S183. |
| [126] | Xiong L, Zhu JK (2001). Abiotic stress signal transduction in plants: molecular and genetic perspectives.Physiol Plan- tarum 112, 152-166. |
| [127] | Yadeta KA, Elmore JM, Coaker G (2013). Advancements in the analysis of the Arabidopsis plasma membrane pro- teome.Front Plant Sci 4, 1-10. |
| [128] | Yang YL, Guo JK, Zhang F, Zhao LQ, Zhang LX (2004). NaCl induced changes of the H+-ATPase in root plasma membrane of two wheat cultivars.Plant Sci 166, 913-918. |
| [129] | Yang Z (2002). Small GTPases: versatile signaling switches in plants. Plant Cell 14, S375-S388. |
| [130] | Yazaki K (2006). ABC transporters involved in the transport of plant secondary metabolites.FEBS Lett 580, 1183-1191. |
| [131] | Yoshimoto N, Takahashi H, Smith FW, Yamaya T, Saito K (2002). Two distinct high-affinity sulfate transporters with different inducibilities mediate uptake of sulfate in Arabidopsis roots.Plant J 29, 465-473. |
| [132] | Zhang H, Han B, Wang T, Chen S, Li H, Zhang Y, Dai S (2012). Mechanisms of plant salt response: insights from proteomics.J Proteome Res 11, 49-67. |
| [133] | Zhang J, Ma H, Feng J, Zeng L, Wang Z, Chen S (2008). Grape berry plasma membrane proteome analysis and its differential expression during ripening.J Exp Bot 59, 2979-2990. |
| [134] | Zhang LF, Yang HM, Cui SX, Hu J, Wang J, Kuang TY, Norling B, Huang F (2009). Proteomic analysis of plasma membranes of cyanobacterium Synechocystis sp. strain PCC 6803 in response to high pH stress.J Proteome Res 8, 2892-2902. |
| [135] | Zwiewka M, Nodzyński T, Robert S, Vanneste S, Friml J (2015). Osmotic stress modulates the balance between exocytosis and clathrin-mediated endocytosis in Arabidopsis thaliana.Mol Plant 8, 1175-1187. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||