

Cloning and Functional Analysis of the BnaA02.CPSF6 Gene from Brassica napus
Received date: 2024-05-05
Accepted date: 2024-07-23
Online published: 2024-07-29
INTRODUCTION: The CPSF family (cleavage and polyadenylation specificity factor) is a crucial protein family that is responsible for polyadenylation signal recognition in mRNA precursors, cleavage and the addition of poly(A) tails to mRNAs in plants. This family plays crucial roles in the regulation of flowering time, the environmental response, and seed development. Currently, the function of the CPSF family genes in Brassica napus is unclear.
RATIONALE: To explore the function and expression patterns of the CPSF gene family, this study cloned BnaA02.CPSF6 from B. napus variety Zhongshuang No.11 and conducted bioinformatics analysis, subcellular localization, expression pattern, and functional characterization of the gene.
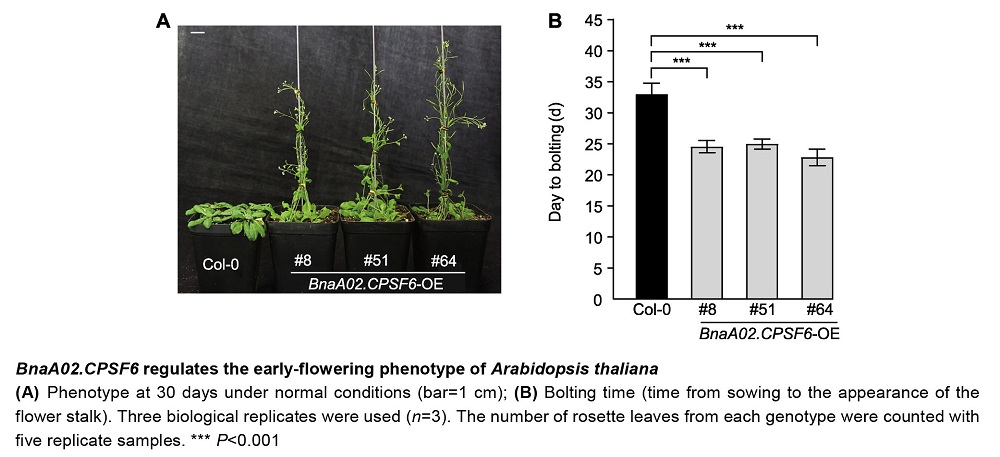
RESULTS: These results indicate that the coding region of the BnaA02.CPSF6 gene is 1 938 bp in length and encodes 646 amino acids without intron structures. Its promoter region contains multiple cis-acting elements involved in light responses and MYB binding sites. Additionally, there are six genes homologous to BnaA02.CPSF6 in B. napus. The BnaA02.CPSF6 gene expressed in the roots, stems, leaves, flowers and different developmental seeds of B. napus, especially significantly higher in 15-35 d developmental seeds, and its encoded protein was localized in the nucleus. The BnaA02.CPSF6 gene expression is upregulated under salt and drought stress. Under treatment with hormones such as ABA, IAA, GA3, SA, and MeJA, the expression of BnaA02.CPSF6 gene is initially inhibited and then gradually recovers to normal levels. Under normal conditions, the overexpression of the BnaA02.CPSF6 gene in Arabidopsis thaliana results in an early bolting phenotype, along with a reduced number of rosette leaves.
CONCLUSION: In summary, the above results indicate that the BnaA02.CPSF6 is involved in abiotic stress responses, is regulated by phytohormones, and may also play a promoting role in flowering regulation.

Qingyang Li , Cui Liu , Li He , Shan Peng , Jiayin Ma , Ziyi Hu , Hongbo Liu . Cloning and Functional Analysis of the BnaA02.CPSF6 Gene from Brassica napus[J]. Chinese Bulletin of Botany, 2025 , 60(1) : 62 -73 . DOI: 10.11983/CBB24068
| [1] | Bao SJ, Hua CM, Shen LS, Yu H (2020). New insights into gibberellin signaling in regulating flowering in Arabidopsis. J Integr Plant Biol 62,118-131. |
| [2] | Boreikait? V, Passmore LA (2023). 3′-end processing of eukaryotic mRNA: machinery, regulation, and impact on gene expression. Annu Rev Biochem 92, 199-225. |
| [3] | Brown KM, Gilmartin GM (2003). A mechanism for the regulation of pre-mRNA 3′ processing by human cleavage factor Im. Mol Cell 12, 1467-1476. |
| [4] | Cai FF, Shao CS, Sun YQ (2022). The role of alternative splicing in floral transition. Chin Bull Bot 57, 69-79. (in Chinese) |
| 蔡芳芳, 邵长生, 孙玉强 (2022). 可变剪切在植物成花转换中的作用. 植物学报 57, 69-79. | |
| [5] | Cui GX, Hou J, Tong L, Xu ZR (2010). Light responsive elements and binding proteins of plant genes. Plant Physiol Commun 46, 991-1000. (in Chinese) |
| 崔国新, 侯杰, 佟玲, 许志茹 (2010). 植物基因光反应元件及其结合蛋白. 植物生理学通讯 46, 991-1000. | |
| [6] | Dai YQ, Luo LJ, Zhao Z (2023). Genetic robustness control of auxin output in priming organ initiation. Proc Natl Acad Sci USA 120, e2221606120. |
| [7] | Eckardt NA (2002). Alternative splicing and the control of flowering time. Plant Cell 14, 743-747. |
| [8] | Edwalds-Gilbert G, Milcarek C (1995). The binding of a subunit of the general polyadenylation factor cleavage- polyadenylation specificity factor (CPSF) to polyadenylation sites changes during B cell development. Nucleic Acids Symp Ser (33), 229-233. |
| [9] | Feng W, Jacob Y, Veley KM, Ding L, Yu XH, Choe G, Michaels SD (2011). Hypomorphic alleles reveal FCA- independent roles for FY in the regulation of FLOWERING LOCUS C. Plant Physiol 155, 1425-1434. |
| [10] | Hao SQ, Zhang LD, Zhao DH, Zhou JW, Ye CT, Qu HD, Li QQ (2023). Inhibitor AN3661 reveals biological functions of Arabidopsis CLEAVAGE and POLYADENYLATION SPECIFICITY FACTOR 73. Plant Physiol 193, 537-554. |
| [11] | Hardy JG, Norbury CJ (2016). Cleavage factor Im (CFIm) as a regulator of alternative polyadenylation. Biochem Soc Trans 44, 1051-1057. |
| [12] | Henderson IR, Liu FQ, Drea S, Simpson GG, Dean C (2005). An allelic series reveals essential roles for FY in plant development in addition to flowering-time control. Development 132, 3597-3607. |
| [13] | Herr AJ, Molnàr A, Jones A, Baulcombe DC (2006). Defective RNA processing enhances RNA silencing and influences flowering of Arabidopsis. Proc Natl Acad Sci USA 103, 14994-15001. |
| [14] | Hornyik C, Terzi LC, Simpson GG (2010). The spen family protein FPA controls alternative cleavage and polyadenylation of RNA. Dev Cell 18(2), 203-213. |
| [15] | Hou YF, Sun J, Wu BX, Gao YY, Nie HB, Nie ZT, Quan SX, Wang Y, Cao XF, Li SS (2021). CPSF30-L-mediated recognition of mRNA m6A modification controls alternative polyadenylation of nitrate signaling-related gene transcripts in Arabidopsis. Mol Plant 14, 688-699. |
| [16] | Huang Y, Zhao PS, Xie LL, Xu JS, Cheng Y, Zhang XK, Xu BB (2024). Analysis on yield composition and breeding strategy of winter rape varieties in the Yangtze River Basin. Chin J Oil Crop Sci 46, 13-18. (in Chinese) |
| 黄郢, 赵培森, 谢伶俐, 徐劲松, 程勇, 张学昆, 许本波 (2024). 长江流域冬油菜品种产量构成及育种策略分析. 中国油料作物学报 46, 13-18. | |
| [17] | Kumar A, Clerici M, Muckenfuss LM, Passmore LA, Jinek M (2019). Mechanistic insights into mRNA 3′-end processing. Curr Opin Struct Biol 59, 143-150. |
| [18] | Li GC, Niu QC, Leng BF, Ding YF, Tong T, Fan LX (2024). The decade of rapeseed industry in the new era: development and its path choice. Chin J Oil Crop Sci 46, 228-235. (in Chinese) |
| 李谷成, 牛秋纯, 冷博峰, 丁逸飞, 童婷, 范丽霞 (2024). 新时代十年: 我国油菜产业发展与路径选择. 中国油料作物学报 46, 228-235. | |
| [19] | Li QX, Zhang L, Wang Y, Huang XX (2019). The research progress of gibberellin on the regulation of flowering and floral organ development in plant. Chin J Cell Biol 41, 746-758. (in Chinese) |
| 李巧峡, 张丽, 王玉, 黄小霞 (2019). 赤霉素调控植物开花及花器官发育的研究进展. 中国细胞生物学学报 41, 746-758. | |
| [20] | Lin JC, Xu RQ, Wu XH, Shen YJ, Li QQ (2017). Role of cleavage and polyadenylation specificity factor 100: anchoring poly (A) sites and modulating transcription termination. Plant J 91, 829-839. |
| [21] | Liu YT, Wu GX, Zhao YP, Wang HHL, Dai ZY, Xue WC, Yang J, Wei HB, Shen RX, Wang HY (2021). DWARF53 interacts with transcription factors UB2/UB3/TSH4 to regulate maize tillering and tassel branching. Plant Physiol 187, 947-962. |
| [22] | Ma LY, Guo C, Li QQ (2014). Role of alternative polyadenylation in epigenetic silencing and antisilencing. Proc Natl Acad Sci USA 111, 9-10. |
| [23] | Macknight R, Duroux M, Laurie R, Dijkwel P, Simpson G, Dean C (2002). Functional significance of the alternative transcript processing of the Arabidopsis floral promoter FCA. Plant Cell 14, 877-888. |
| [24] | Mandel CR, Bai Y, Tong L (2008). Protein factors in pre- mRNA 3′-end processing. Cell Mol Life Sci 65, 1099-1122. |
| [25] | Mouradov A, Cremer F, Coupland G (2002). Control of flowering time: interacting pathways as a basis for diversity. Plant Cell 14, S111-S130. |
| [26] | Proudfoot N (2004). New perspectives on connecting messenger RNA 3′ end formation to transcription. Curr Opin Cell Biol 16, 272-278. |
| [27] | Quesada V, Macknight R, Dean C, Simpson GG (2003). Autoregulation of FCA pre-mRNA processing controls Arabidopsis flowering time. EMBO J 22, 3142-3152. |
| [28] | Schul W, Groenhout B, Koberna K, Takagaki Y, Jenny A, Manders EM, Raska I, van Driel R, de Jong L (1996). The RNA 3′ cleavage factors CstF 64 kDa and CPSF 100 kDa are concentrated in nuclear domains closely associated with coiled bodies and newly synthesized RNA. EMBO J 15, 2883-2892. |
| [29] | Song PZ, Yang JB, Wang CL, Lu Q, Shi LQ, Tayier S, Jia GF (2021). Arabidopsis N6-methyladenosine reader CPSF30-L recognizes FUE signals to control polyadenylation site choice in liquid-like nuclear bodies. Mol Plant 14, 571-587. |
| [30] | Thomas PE, Wu XH, Liu M, Gaffney B, Ji GL, Li QQ, Hunt AG (2012). Genome-wide control of polyadenylation site choice by CPSF30 in Arabidopsis. Plant Cell 24, 4376-4388. |
| [31] | Tzafrir I, Pena-Muralla R, Dickerman A, Berg M, Rogers R, Hutchens S, Sweeney TC, McElver J, Aux G, Patton D, Meinke D (2004). Identification of genes required for embryo development in Arabidopsis. Plant J 135, 1206-1220. |
| [32] | Venkataraman K, Brown KM, Gilmartin GM (2005). Analysis of a noncanonical poly (A) site reveals a tripartite mechanism for vertebrate poly (A) site recognition. Genes Dev 19, 1315-1327. |
| [33] | Wang XP, Niu YL, Zheng Y (2021). Multiple functions of MYB transcription factors in abiotic stress responses. Int J Mol Sci 22, 6125. |
| [34] | Yang Q, Coseno M, Gilmartin GM, Doublié S (2011). Crystal structure of a human cleavage factor CFIm25/CFIm68/RNA complex provides an insight into poly(A) site recognition and RNA looping. Structure 19, 368-377. |
| [35] | Yu ZB, Lin JC, Li QQ (2019). Transcriptome analyses of FY mutants reveal its role in mRNA alternative polyadenylation. Plant Cell 31, 2332-2352. |
| [36] | Zhang CS, Wei T, Zhou YP, Fan T, Lü TX, Tian CE (2021). Progress in flowering regulation mechanisms of FLC. Chin Bull Bot 56,651-663. (in Chinese) |
| 张长生, 魏滔, 周玉萍, 范甜, 吕天晓, 田长恩 (2021). FLC调控植物成花的分子机制研究新进展. 植物学报 56, 651-663. | |
| [37] | Zhang XJ, Nomoto M, Garcia-León M, Takahashi N, Kato M, Yura K, Umeda M, Rubio V, Tada Y, Furumoto T, Aoyama T, Tsuge T (2022). CFI 25 subunit of cleavage factor I is important for maintaining the diversity of 3′ UTR lengths in Arabidopsis thaliana (L.) heynh. Plant Cell Physiol 63, 369-383. |
/
| 〈 |
|
〉 |