

收稿日期: 2024-12-07
录用日期: 2025-02-09
网络出版日期: 2025-02-10
基金资助
大学生创新创业基金资助项目(XDC2023031)
Rapid Propagation Technology of Microsorum punctatum in Vitro
Received date: 2024-12-07
Accepted date: 2025-02-09
Online published: 2025-02-10
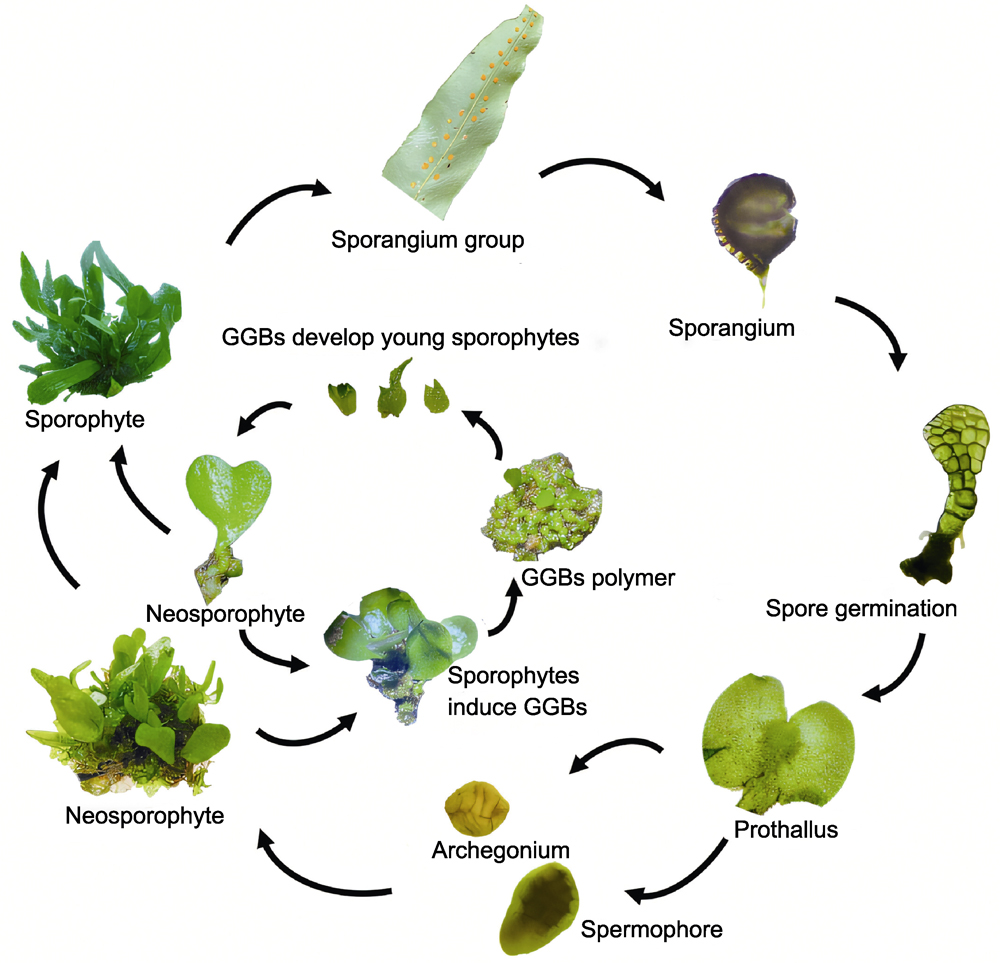
为加强野生蕨类植物的保护与开发, 优化星蕨(Microsorum punctatum)孢子萌发方法和条件, 比较分析了不同因素对原叶体增殖、孢子体诱导、绿色球状小体(GGBs)诱导及其发育为幼孢子体的影响, 建立人工高效快繁技术体系。以成熟孢子为材料, 分别以MS、1/2MS、1/3MS和1/4MS为基本培养基在无菌条件下萌发; 通过L9(34)正交试验, 研究无机盐浓度、植物生长调节剂及其质量浓度对原叶体发生和增殖的影响。当原叶体增殖到一定数量时, 再以MS、1/2MS、1/3MS和1/4MS为基本培养基筛选适宜诱导孢子体的培养基; 随后, 以幼孢子体为材料诱导GGBs发育为新的幼孢子体并炼苗移栽。适宜孢子萌发的培养基为1/2MS, 原叶体在MS+0.3 mg·L-1 6-BA+1.5 mg·L-1 NAA培养基中大量增殖, 60天后增殖系数约为9.6; 将原叶体切割后接入1/4MS培养基, 加无菌水培养90天后, 幼孢子体发生系数约为10.0; 幼孢子体在1/2MS+1.5 mg·L-1 6-BA+0.1 mg·L-1 NAA培养基中可诱导出GGBs, 诱导率达93.3%, GGBs在此培养基中的增殖系数达32.0; 在1/2MS培养基中GGBs的分化成苗率较高, 最高约为92%; 试管苗经炼苗移栽成活率在90%以上。该研究建立了原叶体-受精-孢子体和幼孢子体-GGBs-幼孢子体2个技术体系, 尤其是GGBs的产生, 极大缩短了植株的再生周期。研究结果可为优质种苗及其它蕨类植物的人工繁育提供技术支撑。
葛晓青 , 李梦瑶 , 黄衡宇 , 张爱丽 . 星蕨体外快繁技术[J]. 植物学报, 2025 , 60(6) : 944 -956 . DOI: 10.11983/CBB24190
INTRODUCTION: The wild populations of Microsorum punctatum face endangerment due to habitat degradation and low spore reproductive efficiency. Fern life cycles involve alternating gametophyte and sporophyte generations, where gametophyte development and sporophyte transition represent critical bottlenecks in in vitro propagation, heavily influenced by environmental factors and culture conditions. Although asexual propagation techniques such as green globular bodies (GGBs) have been successfully applied in some fern species, low sporophyte induction efficiency and proliferation challenges persist, hindering large-scale production. This study employed M. punctatum spores to systematically investigate sterile germination mechanisms, gametophyte proliferation, and sporophyte regeneration. A dual-pathway rapid propagation system was established, integrating high-efficiency prothallus proliferation with GGBs induction, aiming to provide both theoretical insights and practical solutions for conserving endangered fern resources and advancing industrial-scale cultivation.
RATIONALE: The unique alternation of generations life cycle in ferns, characterized by independent gametophyte survival, provides a theoretical framework for in vitro propagation. Studies have demonstrated that gametophyte homogenization culture and GGBs induction can overcome sporophyte regeneration barriers, while medium composition and phytohormone ratios critically regulate developmental phase transitions. To address the challenges of low spore propagation efficiency and habitat sensitivity in M. punctatum, this study leverages its gametophyte proliferation potential and rhizome meristematic activity in sporophytes. By optimizing aseptic systems and induction conditions, as well as mimicking the natural fertilization microenvironment, a dual-path regeneration system integrating prothallus proliferation and GGB-based propagation was established, laying a theoretical foundation for efficient conservation of endangered ferns.
RESULTS: Spore germination was optimally achieved in 1/2MS medium. Prothalli exhibited vigorous proliferation in MS medium supplemented with 0.3 mg·L-1 6-BA and 1.5 mg·L-1 NAA, reaching a proliferation coefficient of 9.6 after 60 days of culture. Fragmented prothalli transferred to 1/4MS medium with sterile water supplementation achieved a young sporophyte induction coefficient of 10.0 following 90 day cultivation. GGBs were successfully induced from young sporophytes in 1/2MS medium containing 1.5 mg·L-1 6-BA and 0.1 mg·L-1 NAA, showing 93.3% induction efficiency and a remarkable proliferation coefficient of 32.0. The GGB differentiation into plantlets was most efficient in 1/2MS medium, yielding a conversion rate of 92%. Acclimatized plantlets demonstrated over 90% survival rate post-transplantation.
CONCLUSION: This study successfully established an efficient in vitro rapid propagation system for M. punctatum spores. Optimization of sterilization duration and culture medium types significantly enhanced spore germination rates. A prothallus culture protocol with a high proliferation coefficient was developed, overcoming bottlenecks in gametophyte mass propagation. Liquid immersion-assisted fertilization technology enabled efficient induction of young sporophytes, while the GGBs induction system markedly shortened the regeneration cycle. For the first time, a dual-pathway rapid propagation strategy—“prothallus proliferation-sporophyte induction” combined with “GGBs cyclic regeneration” was proposed. The study demonstrated that the meristematic properties of M. punctatum GGBs are distinct from callus tissue, providing a robust technical framework for the conservation of endangered ferns and industrial-scale seedling production.
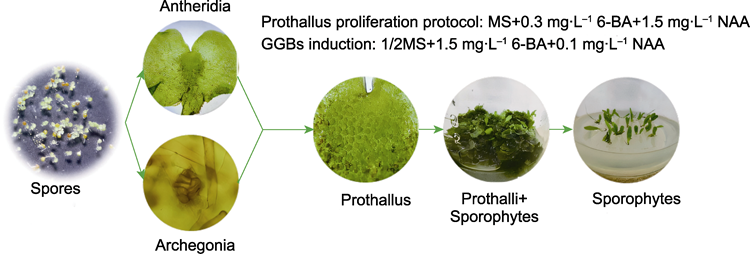
Formation of antheridia, archegonia, and sporophyte production in Microsorum punctatum

| [1] | Aldea F, Banciu C, Brezeanu A, Helepciuc FE, Soare LC (2016). In vitro micropropagation of fern species (Pteridophyta) of biotechnological interest, for ex situ conservation. Oltenia Studii Si Comunic?ri ?tiin?ele Naturii 32, 27-35. |
| [2] | Banks JA (1999). Gametophyte development in ferns. Annu Rev Plant Biol 50, 163-186. |
| [3] | Bertrand AM, Albuerne MA, Fernández H, González A, Sánchez-Tamés R (1999). In vitro organogenesis of Polypodium cambricum. Plant Cell Tissue Organ Cult 57, 65-69. |
| [4] | Chen P, Yang LL, Peng Y, Zhang SZ, Xu GH, Yang JF, Zhang SZ (2021). In vitro regeneration of Osmunda mildei. Guihaia 41, 301-307. (in Chinese) |
| 陈朋, 杨蕾蕾, 彭杨, 张苏州, 徐桂红, 杨建芬, 张寿洲 (2021). 粤紫萁离体再生技术研究. 广西植物 41, 301-307. | |
| [5] | Cho JS, Lee CH (2017). Several factors affecting mass production of Microlepia strigosa (Thunb.) C. Presl sporophytes. Hortic Sci Technol 35, 46-58. |
| [6] | Dela Cruz RY, Ang AMG, Doblas GZ, Librando IL, Porquis HC, Batoctoy BCLS, Cabresos CC, Jacalan DRY, Amoroso VB (2017). Phytochemical screening, antioxidant and anti-inflammatory activities of the three fern (poly- podiaceae) species in Bukidnon, Philippines. Bull Environ Pharmacol Life Sci 6, 28-33. |
| [7] | Duan JQ, Sun DK, Xiang JY, Tang JR (2021). Study on tissue culture and rapid propagation technology of Asplenium nidus. Seed 40(5), 84-90. (in Chinese) |
| 段杰秋, 孙大宽, 向建英, 唐军荣 (2021). 巢蕨组培快繁技术研究. 种子 40(5), 84-90. | |
| [8] | Fernández H, Revilla MA (2003). In vitro culture of ornamental ferns. Plant Cell Tissue Organ Cult 73, 1-13. |
| [9] | Flora of China Editorial Committee (2000). Flora of China (vol. 6). Beijing: Science Press. pp. 226. (in Chinese) |
| 中国科学院中国植物志编辑委员会 (2000). 中国植物志(第6卷, 第2分册). 北京: 科学出版社. pp. 226. | |
| [10] | Gao XW, Zhou SY, Bian ZJ, Wang QH, Dai XL (2018). Effects on morphological development of gametophytes of three fern species under different moisture conditions. Jo urnal of Shanghai Normal University (Nat Sci) 47, 726-733. (in Chinese) |
| 高晓雯, 周施雨, 卞竹箐, 王清华, 戴锡玲 (2018). 不同水分条件对3种蕨类植物配子体形态发育的影响. 上海师范大学学报(自然科学版) 47, 726-733. | |
| [11] | Greer GK, McCarthy BC (2000). Patterns of growth and reproduction in a natural population of the fern Polystichum acrostichoides. Am Fern J 90, 60-76. |
| [12] | Hegde S, D’Souza L (2000). Recent advances in biotechnology of ferns. In: Trivedi PC, ed. Plant Biotechnology—Recent Advances. New Delhi: Panima Publishing Corporation. pp. 213-237. |
| [13] | IUCN (2024). The IUCN Red List of Threatened Species. https://www.iucnredlist.org. 2024-03-09. |
| [14] | Jang BK, Cho JS, Lee KC, Lee CH (2017). Culture conditions affecting spore germination, prothallus propagation and sporophyte formation of Dryopteris nipponensis Koidz. Hortic Sci Technol 35, 480-489. |
| [15] | Jang BK, Cho JS, Park K, Lee CH (2019). A methodology for large-scale Athyrium sheareri gametophyte proliferation and sporophyte production using tissue culture. In Vitro Cell Dev Biol Plant 55, 519-526. |
| [16] | Korpelainen H (1994). Growth, sex determination and reproduction of Dryopteris filix-mas (L.) Schott gametophytes under varying nutritional conditions. Bot J Linn Soc 114, 357-366. |
| [17] | Korpelainen H (1995). Growth and reproductive characteristics in artificially formed clonal gametophytes of Dryopteris filix-mas (Dryopteridaceae). Plant Syst Evol 196, 195-206. |
| [18] | Krieg CP, Chambers SM (2022). The ecology and physiology of fern gametophytes: a methodological synthesis. Appl Plant Sci 10, e11464. |
| [19] | Lin CW, Shih CH, Chang HC, Kao CY (2024). Regeneration of endangered fern Adiantum reniforme var. sinense. Indian J Agric Res 58, 56-62. |
| [20] | Murashige T, Skoog F (1962). A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15, 473-497. |
| [21] | Park K, Jang BK, Lee CH (2019). Culture condition for gametophyte and sporophyte masspropagation of bamboo fern (Coniogramme japonica) using tissue culture. J Plant Biotechnol 46, 119-126. |
| [22] | Park K, Jang BK, Lee HM, Cho JS, Lee CH (2020). An efficient method for in vitro shoot-tip culture and sporophyte production using Selaginella martensii Spring sporophyte. Plants 9, 235. |
| [23] | Petchsri S, Boonkerd T, Baum BR, Karladee D, Suriyong S, Lungkaphin A (2012). Phenetic study of the Microsorum punctatum complex (Polypodiaceae). Sci Asia 38, 1-12. |
| [24] | Ravi BX, Robert J, Gabriel M (2014). In vitro spore germination and gametophytic growth development of a critically endangered fern Pteris tripartita Sw. Afr J Biotechnol 13, 2350-2358. |
| [25] | Ravi BX, Varuvel GVA, Kilimas R, Robert J (2015). Apogamous sporophyte development through spore reproduction of a South Asia’s critically endangered fern: Pteris tripartita Sw. Asian Pac J Reprod 4, 135-139. |
| [26] | Rybczyński JJ, Tomiczak K, Grzyb M, Miku?a A (2018). Morphogenic events in ferns:single and multicellular explants in vitro. In: In: Fernández H, ed. Current Advances in Fern Research. Cham: Springer. pp. 99-120. |
| [27] | Sanders HL, Darrah PR, Langdale JA (2011). Sector analysis and predictive modelling reveal iterative shoot-like development in fern fronds. Development 138, 2925-2934. |
| [28] | Somer M, Arbesú R, Menéndez V, Revilla MA, Fernández H (2010). Sporophyte induction studies in ferns in vitro. Euphytica 171, 203-210. |
| [29] | Vasco A, Moran RC, Ambrose BA (2013). The evolution, morphology, and development of fern leaves. Front Plant Sci 4, 345. |
| [30] | Yang FC (2009). Special topics on tropical pteridophyte (XIII) cultivation and management of Microsorum punctatum. China Flowers and Horticulture (24), 20-21. (in Chinese) |
| 杨逢春 (2009). 热带蕨类植物专题(十三) 星蕨的栽培管理. 中国花卉园艺 (24), 20-21. | |
| [31] | Yu RP, Li F, Wang GX, Ruan JW, Wu LF, Wu M, Yang CM, Shan QL (2021). In vitro regeneration of the colorful fern Pteris aspericaulis var. tricolor via green globular bodies system. In Vitro Cell Dev Biol Plant 57, 225-234. |
| [32] | Yu RP, Zhang GF, Li H, Cao H, Mo XJ, Gui M, Zhou XH, Jiang YL, Li SC, Wang JH (2017). In vitro propagation of the endangered tree fern Cibotium barometz through formation of green globular bodies. Plant Cell Tissue Organ Cult 128, 369-379. |
| [33] | Zhang DR, Bu ZG, Chen LL, Chang Y (2020). Establishment of a tissue culture and rapid propagation system of Dryopteris fragrans. Chin Bull Bot 55, 760-767. (in Chinese) |
| 张冬瑞, 卜志刚, 陈玲玲, 常缨 (2020). 香鳞毛蕨的组织培养和快速繁殖体系构建. 植物学报 55, 760-767. | |
| [34] | Zhang W, Wang Q, Chen GH, Chen HF (2020). Partial tissue culture and rapid propagation of Phymatosorus scolopendria spores. Plant Physiology Journal 56, 856-862. (in Chinese) |
| 张薇, 王强, 陈国华, 陈红锋 (2020). 瘤蕨孢子的不完全组织培养与快速繁殖. 植物生理学报 56, 856-862. |
/
| 〈 |
|
〉 |