

Chinese Bulletin of Botany ›› 2016, Vol. 51 ›› Issue (3): 322-334.DOI: 10.11983/CBB15024 cstr: 32102.14.CBB15024
• EXPERIMENTAL COMMUNICATIONS • Previous Articles Next Articles
Heyu Yang1, 3, Haiyan Wei2*, Manjie Sang1, 2, Zhonghui Shang1, 2, Yajuan Mao1, 2, Xiaorui Wang1, 3, Fang Liu1, 3, Wei Gu1,
Received:2015-02-12
Accepted:2015-09-07
Online:2016-05-01
Published:2016-05-24
Contact:
Wei Haiyan,Gu Wei
About author:? These authors contributed equally to this paper
Heyu Yang, Haiyan Wei, Manjie Sang, Zhonghui Shang, Yajuan Mao, Xiaorui Wang, Fang Liu, Wei Gu. Phenotypic Plasticity of Schisandra sphenanthera Leaf and the Effect of Environmental Factors on Leaf Phenotype[J]. Chinese Bulletin of Botany, 2016, 51(3): 322-334.
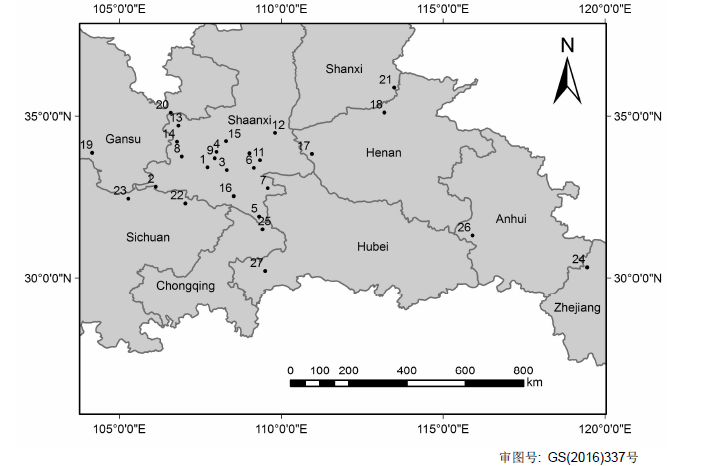
Fig. 1 Figure 1 Map of sampling sites of Schisandra sphenanthera(1: Maoping in Shaanxi (SN-MP); 2: Ningqiang in Shaanxi (SN-NQ); 3: Ningshan in Shaanxi (SN-NS); 4: Taibai in Shaanxi (SN-TB); 5: Zhenping in Shaanxi (SN-ZP); 6: Zhen’an in Shaanxi (SN-ZA); 7: Xunyang in Shaanxi (SN-XY); 8: Liuba in Shaanxi (SN-LB); 9: Foping in Shaanxi (SN-FP); 10: Yingpan in Shaanxi (SN-YP); 11: Fengzhen in Shaanxi (SN-FZ); 12: Huaxian in Shaanxi (SN-HX); 13: Longxian in Shaanxi (SN-LX); 14: Fengxian in Shaanxi (SN-FX); 15: Meixian in Shaanxi (SN-MX); 16: Ziyang in Shaanxi (SN-ZY); 17: Lushi in Henan (HN-LS); 18: Xiuwu in Henan (HN-XW); 19: Zhouqu in Gansu (GS-ZQ); 20: Huating in Gansu (GS-HT); 21: Lingchuan in Shanxi (SX-LC); 22: Nanjiang in Sichuan (SC-NJ); 23: Qingchuan in Sichuan (SC-QC); 24: Lin’an in Zhejiang (ZJ-LA); 25: Wuxi in Chongqing (CQ-WX); 26: Jinzhai in Anhui (AH-JZ); 27: Enshi in Hubei (HB-ES))
| Populationsa | df | LL | LW | PL | LA | LSI | NLT | NSV | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MS | F value | MS | F value | MS | F value | MS | F value | MS | F value | MS | F value | MS | F value | |||||||||||
| SN-MP | 14 | 5.30 | 2.81** | 2.49 | 4.36** | 1.24 | 2.33** | 234.71 | 4.30** | 0.16 | 1.906* | 22.42 | 5.15** | 21.33 | 14.36** | |||||||||
| SN-NQ | 9 | 8.73 | 9.80** | 2.60 | 8.56** | 1.27 | 4.22** | 241.30 | 10.67** | 0.27 | 3.89** | 13.65 | 5.30** | 12.63 | 9.93** | |||||||||
| SN-NS | 19 | 12.37 | 7.28** | 4.27 | 9.61** | 2.98 | 10.82** | 511.60 | 9.63** | 0.22 | 3.09** | 16.36 | 5.01** | 34.95 | 24.81** | |||||||||
| SN-TB | 16 | 13.98 | 5.98** | 3.61 | 6.27** | 2.42 | 5.18** | 600.34 | 7.06** | 0.29 | 3.06** | 31.66 | 3.80** | 4.90 | 4.75** | |||||||||
| SN-ZP | 24 | 8.76 | 4.27** | 3.75 | 7.36** | 1.13 | 3.21** | 398.01 | 7.46** | 0.28 | 2.085* | 10.84 | 3.19** | 7.29 | 3.89** | |||||||||
| HN-LS | 19 | 4.25 | 2.62** | 2.63 | 4.56** | 3.25 | 7.60** | 191.05 | 3.19** | 0.08 | 4.56** | 3.33 | 1.42 | 1.99 | 3.73** | |||||||||
| HN-XW | 19 | 10.90 | 6.23** | 4.46 | 7.35** | 4.86 | 6.92** | 448.17 | 6.89** | 0.25 | 6.34** | 8.02 | 10.71** | 8.81 | 21.20** | |||||||||
| GS-ZQ | 19 | 4.93 | 5.45** | 3.29 | 6.82** | 2.09 | 4.93** | 235.73 | 4.94** | 0.24 | 10.32** | 3.47 | 3.15** | 5.43 | 8.25** | |||||||||
| SX-LC | 16 | 6.92 | 5.20** | 5.01 | 11.28** | 3.20 | 5.87** | 347.08 | 8.71** | 0.40 | 6.87** | 1.32 | 3.645* | 2.91 | 3.76** | |||||||||
| SC-NJ | 19 | 8.49 | 6.96** | 3.12 | 6.07** | 4.48 | 1.47 | 197.54 | 6.46** | 0.53 | 8.17** | 7.04 | 5.24** | 1.56 | 3.54** | |||||||||
| SC-QC | 19 | 8.66 | 9.59** | 1.79 | 4.29** | 1.45 | 4.07** | 155.13 | 5.87** | 0.34 | 8.08** | 1.51 | 1.37 | 0.65 | 0.93 | |||||||||
| SN-ZA | 19 | 5.57 | 5.42** | 3.02 | 6.83** | 1.65 | 5.92** | 237.55 | 5.45** | 0.13 | 9.22** | 2.14 | 3.89** | 2.02 | 3.42** | |||||||||
| SN-XY | 19 | 5.34 | 5.36** | 3.59 | 11.05** | 1.59 | 3.18** | 311.40 | 10.16** | 0.15 | 3.88** | 3.23 | 2.039* | 4.04 | 4.51** | |||||||||
| SN-LB | 18 | 7.59 | 8.04** | 5.58 | 13.70** | 2.20 | 5.42** | 349.77 | 10.91** | 0.18 | 6.65** | 8.53 | 14.65** | 3.19 | 7.96** | |||||||||
| SN-FP | 19 | 5.06 | 9.48** | 1.90 | 14.08** | 2.67 | 2.44** | 156.47 | 11.57** | 0.28 | 10.41** | 2.16 | 1.78 | 29.72 | 1.00** | |||||||||
| SN-YP | 9 | 6.20 | 5.48** | 3.59 | 6.69** | 1.98 | 3.37** | 164.29 | 4.16** | 0.70 | 10.22** | 3.03 | 1.80** | 3.39 | 7.86** | |||||||||
| SN-FZ | 13 | 7.28 | 7.55** | 5.00 | 10.18** | 3.21 | 10.27** | 353.67 | 10.35** | 0.27 | 6.31** | 3.04 | 1.87 | 3.00 | 5.903** | |||||||||
| SN-HX | 9 | 8.72 | 5.37** | 3.39 | 6.58** | 4.29 | 6.19** | 367.31 | 5.71** | 0.16 | 6.66** | 10.11 | 20.85** | 1.65 | 3.10** | |||||||||
| SN-LX | 10 | 12.20 | 9.12** | 2.82 | 3.91** | 3.27 | 4.32** | 378.26 | 6.18** | 0.27 | 3.98** | 9.06 | 16.55 | 3.43 | 7.253** | |||||||||
| SN-FX | 9 | 10.70 | 15.19** | 9.47 | 26.50** | 1.63 | 3.42** | 943.12 | 31.33** | 0.15 | 4.04** | 0.69 | 1.01 | 3.75 | 11.16** | |||||||||
| ZJ-LA | 10 | 21.23 | 6.55** | 5.90 | 5.67** | 1.09 | 3.12** | 1168.80 | 6.00** | 0.13 | 5.30** | 3.60 | 4.32** | 2.27 | 2.72** | |||||||||
| CQ-WX | 14 | 15.17 | 6.84** | 6.64 | 10.91** | 4.90 | 16.15** | 592.90 | 8.20** | 0.20 | 5.56** | 6.47 | 4.77** | 2.44 | 4.90** | |||||||||
| AH-JZ | 10 | 19.67 | 8.93** | 9.29 | 11.94** | 2.82 | 11.10** | 1101.73 | 11.89** | 0.35 | 5.77** | 11.65 | 19.38** | 6.71 | 18.51** | |||||||||
| GS-HT | 9 | 21.70 | 8.12** | 8.99 | 11.72** | 2.62 | 5.13** | 818.39 | 9.44** | 0.86 | 8.75** | 2.82 | 3.83* | 2.89 | 4.56** | |||||||||
| HB-ES | 15 | 4.09 | 3.97** | 6.19 | 10.36** | 3.66 | 8.80** | 397.61 | 8.93** | 0.34 | 5.39** | 6.77 | 4.47 | 2.21 | 3.16** | |||||||||
| SN-MX | 14 | 14.56 | 15.60** | 2.16 | 6.25** | 2.64 | 6.21** | 400.21 | 12.39** | 0.44 | 11.28** | 3.23 | 1.71 | 8.51 | 17.20** | |||||||||
| SN-ZY | 14 | 4.99 | 2.33** | 3.26 | 2.78** | 1.24 | 1.82** | 264.18 | 3.45** | 0.07 | 1.02 | 0.30 | 0.19 | 3.58 | 5.19** | |||||||||
Table 1 Results of ANOVA analyses of leave phenotypes within populations in Schisandra sphenanthera
| Populationsa | df | LL | LW | PL | LA | LSI | NLT | NSV | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MS | F value | MS | F value | MS | F value | MS | F value | MS | F value | MS | F value | MS | F value | |||||||||||
| SN-MP | 14 | 5.30 | 2.81** | 2.49 | 4.36** | 1.24 | 2.33** | 234.71 | 4.30** | 0.16 | 1.906* | 22.42 | 5.15** | 21.33 | 14.36** | |||||||||
| SN-NQ | 9 | 8.73 | 9.80** | 2.60 | 8.56** | 1.27 | 4.22** | 241.30 | 10.67** | 0.27 | 3.89** | 13.65 | 5.30** | 12.63 | 9.93** | |||||||||
| SN-NS | 19 | 12.37 | 7.28** | 4.27 | 9.61** | 2.98 | 10.82** | 511.60 | 9.63** | 0.22 | 3.09** | 16.36 | 5.01** | 34.95 | 24.81** | |||||||||
| SN-TB | 16 | 13.98 | 5.98** | 3.61 | 6.27** | 2.42 | 5.18** | 600.34 | 7.06** | 0.29 | 3.06** | 31.66 | 3.80** | 4.90 | 4.75** | |||||||||
| SN-ZP | 24 | 8.76 | 4.27** | 3.75 | 7.36** | 1.13 | 3.21** | 398.01 | 7.46** | 0.28 | 2.085* | 10.84 | 3.19** | 7.29 | 3.89** | |||||||||
| HN-LS | 19 | 4.25 | 2.62** | 2.63 | 4.56** | 3.25 | 7.60** | 191.05 | 3.19** | 0.08 | 4.56** | 3.33 | 1.42 | 1.99 | 3.73** | |||||||||
| HN-XW | 19 | 10.90 | 6.23** | 4.46 | 7.35** | 4.86 | 6.92** | 448.17 | 6.89** | 0.25 | 6.34** | 8.02 | 10.71** | 8.81 | 21.20** | |||||||||
| GS-ZQ | 19 | 4.93 | 5.45** | 3.29 | 6.82** | 2.09 | 4.93** | 235.73 | 4.94** | 0.24 | 10.32** | 3.47 | 3.15** | 5.43 | 8.25** | |||||||||
| SX-LC | 16 | 6.92 | 5.20** | 5.01 | 11.28** | 3.20 | 5.87** | 347.08 | 8.71** | 0.40 | 6.87** | 1.32 | 3.645* | 2.91 | 3.76** | |||||||||
| SC-NJ | 19 | 8.49 | 6.96** | 3.12 | 6.07** | 4.48 | 1.47 | 197.54 | 6.46** | 0.53 | 8.17** | 7.04 | 5.24** | 1.56 | 3.54** | |||||||||
| SC-QC | 19 | 8.66 | 9.59** | 1.79 | 4.29** | 1.45 | 4.07** | 155.13 | 5.87** | 0.34 | 8.08** | 1.51 | 1.37 | 0.65 | 0.93 | |||||||||
| SN-ZA | 19 | 5.57 | 5.42** | 3.02 | 6.83** | 1.65 | 5.92** | 237.55 | 5.45** | 0.13 | 9.22** | 2.14 | 3.89** | 2.02 | 3.42** | |||||||||
| SN-XY | 19 | 5.34 | 5.36** | 3.59 | 11.05** | 1.59 | 3.18** | 311.40 | 10.16** | 0.15 | 3.88** | 3.23 | 2.039* | 4.04 | 4.51** | |||||||||
| SN-LB | 18 | 7.59 | 8.04** | 5.58 | 13.70** | 2.20 | 5.42** | 349.77 | 10.91** | 0.18 | 6.65** | 8.53 | 14.65** | 3.19 | 7.96** | |||||||||
| SN-FP | 19 | 5.06 | 9.48** | 1.90 | 14.08** | 2.67 | 2.44** | 156.47 | 11.57** | 0.28 | 10.41** | 2.16 | 1.78 | 29.72 | 1.00** | |||||||||
| SN-YP | 9 | 6.20 | 5.48** | 3.59 | 6.69** | 1.98 | 3.37** | 164.29 | 4.16** | 0.70 | 10.22** | 3.03 | 1.80** | 3.39 | 7.86** | |||||||||
| SN-FZ | 13 | 7.28 | 7.55** | 5.00 | 10.18** | 3.21 | 10.27** | 353.67 | 10.35** | 0.27 | 6.31** | 3.04 | 1.87 | 3.00 | 5.903** | |||||||||
| SN-HX | 9 | 8.72 | 5.37** | 3.39 | 6.58** | 4.29 | 6.19** | 367.31 | 5.71** | 0.16 | 6.66** | 10.11 | 20.85** | 1.65 | 3.10** | |||||||||
| SN-LX | 10 | 12.20 | 9.12** | 2.82 | 3.91** | 3.27 | 4.32** | 378.26 | 6.18** | 0.27 | 3.98** | 9.06 | 16.55 | 3.43 | 7.253** | |||||||||
| SN-FX | 9 | 10.70 | 15.19** | 9.47 | 26.50** | 1.63 | 3.42** | 943.12 | 31.33** | 0.15 | 4.04** | 0.69 | 1.01 | 3.75 | 11.16** | |||||||||
| ZJ-LA | 10 | 21.23 | 6.55** | 5.90 | 5.67** | 1.09 | 3.12** | 1168.80 | 6.00** | 0.13 | 5.30** | 3.60 | 4.32** | 2.27 | 2.72** | |||||||||
| CQ-WX | 14 | 15.17 | 6.84** | 6.64 | 10.91** | 4.90 | 16.15** | 592.90 | 8.20** | 0.20 | 5.56** | 6.47 | 4.77** | 2.44 | 4.90** | |||||||||
| AH-JZ | 10 | 19.67 | 8.93** | 9.29 | 11.94** | 2.82 | 11.10** | 1101.73 | 11.89** | 0.35 | 5.77** | 11.65 | 19.38** | 6.71 | 18.51** | |||||||||
| GS-HT | 9 | 21.70 | 8.12** | 8.99 | 11.72** | 2.62 | 5.13** | 818.39 | 9.44** | 0.86 | 8.75** | 2.82 | 3.83* | 2.89 | 4.56** | |||||||||
| HB-ES | 15 | 4.09 | 3.97** | 6.19 | 10.36** | 3.66 | 8.80** | 397.61 | 8.93** | 0.34 | 5.39** | 6.77 | 4.47 | 2.21 | 3.16** | |||||||||
| SN-MX | 14 | 14.56 | 15.60** | 2.16 | 6.25** | 2.64 | 6.21** | 400.21 | 12.39** | 0.44 | 11.28** | 3.23 | 1.71 | 8.51 | 17.20** | |||||||||
| SN-ZY | 14 | 4.99 | 2.33** | 3.26 | 2.78** | 1.24 | 1.82** | 264.18 | 3.45** | 0.07 | 1.02 | 0.30 | 0.19 | 3.58 | 5.19** | |||||||||
| Phenotype | Range | Mean value | Mean square | df | F |
|---|---|---|---|---|---|
| LL (cm) | 7.03 | 8.21 | 8.01 | 26 | 8.66** |
| LW (cm) | 8.39 | 5.19 | 3.36 | 26 | 6.86** |
| PL (cm) | 7.55 | 2.71 | 2.77 | 26 | 8.00** |
| LA (cm2) | 53.92 | 28.55 | 309.22 | 26 | 7.31** |
| LSI | 1.26 | 1.60 | 0.29 | 26 | 10.29** |
| NLT | 9.30 | 9.02 | 9.56 | 26 | 7.31** |
| NSV | 8.40 | 7.80 | 17.70 | 26 | 18.46** |
Table 2 Results of ANOVA analyses of leave phenotypes among populations in Schisandra sphenanthera
| Phenotype | Range | Mean value | Mean square | df | F |
|---|---|---|---|---|---|
| LL (cm) | 7.03 | 8.21 | 8.01 | 26 | 8.66** |
| LW (cm) | 8.39 | 5.19 | 3.36 | 26 | 6.86** |
| PL (cm) | 7.55 | 2.71 | 2.77 | 26 | 8.00** |
| LA (cm2) | 53.92 | 28.55 | 309.22 | 26 | 7.31** |
| LSI | 1.26 | 1.60 | 0.29 | 26 | 10.29** |
| NLT | 9.30 | 9.02 | 9.56 | 26 | 7.31** |
| NSV | 8.40 | 7.80 | 17.70 | 26 | 18.46** |
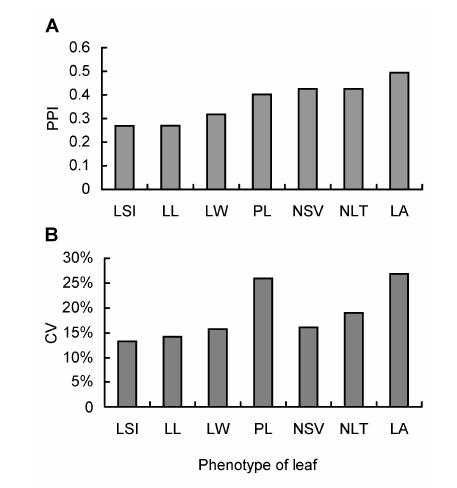
Fig. 2 The phenotypic plasticity index (PPI) and coefficient of variation (CV) of leaves in Schisandra sphenantheraLLLW, PL, LA, LSI, NLT and NSV see Table 1
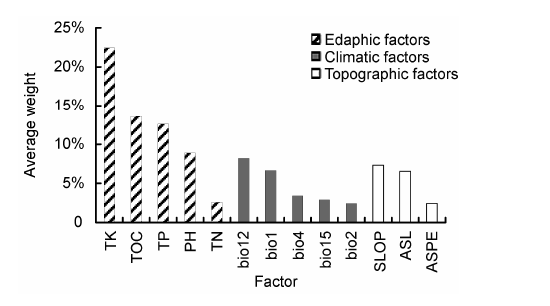
Fig. 3 The average weight of environmental factors on leaf phenotypes in Schisandra sphenanthera(bio1: Annual mean temperature; bio2: Mean diurnal range; bio4: Temperature seasonality; bio12: Annual precipitation; bio15: Precipitation seasonality; ASL: Above sea level; SLOP: Slope; ASPE: Aspect; PH: pH value; TN: Total nitrogen content; TP: Total phosphorus content; TK: Total kalium content; TOC: Total organic carbon content)
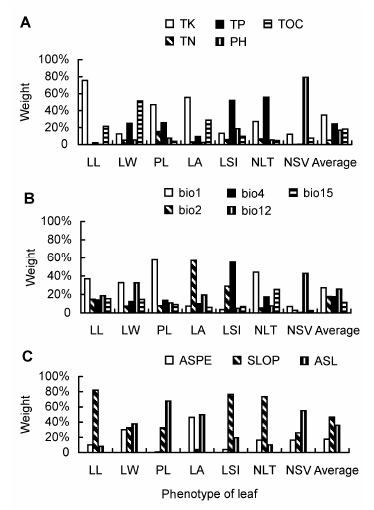
Fig. 4 The weight of edaphic factors (A), climatic factors (B) and topographical factors (C) on leaf phenotypes in Schisandra sphenanthera(LL, LW, PL, LA, LSI, NLT and NSV see Table 1. TK, TN, TP, PH, TOC, bio1, bio2, bio4, bio12, bio15, ASPE, SLOP and ASL see Figure 3.)
| Factors | Phenotypes’ cumulative weight | ||||||
|---|---|---|---|---|---|---|---|
| LL | LW | PL | LA | LSI | NLT | NSV | |
| TK | 68.83% | 70.92% | 43.92% | 41.06% | 64.25% | 87.10% | 86.75% |
| TOC | 88.62% | 40.85% | 99.68% | 62.39% | 80.33% | 100.00% | 90.38% |
| SLOP | 93.71% | 93.22% | 71.28% | 98.82% | 86.54% | 23.48% | 98.88% |
| TP | 95.84% | 60.79% | 80.15% | 86.14% | 39.75% | 68.24% | 99.88% |
| bio1 | 96.79% | 75.49% | 58.36% | 99.47% | 99.68% | 45.37% | 93.88% |
| ASPE | 97.41% | 97.25% | 100.00% | 79.10% | 100.00% | 82.07% | 99.32% |
| ASL | 97.91% | 91.08% | 27.72% | 71.05% | 97.21% | 94.12% | 95.45% |
| bio12 | 98.40% | 80.04% | 91.53% | 97.26% | 99.10% | 90.88% | 43.38% |
| bio15 | 98.80% | 95.27% | 96.40% | 100.00% | 98.30% | 57.94% | 98.18% |
| bio2 | 99.18% | 100.00% | 98.32% | 91.12% | 95.63% | 96.87% | 96.83% |
| bio4 | 99.55% | 98.97% | 88.84% | 98.14% | 72.85% | 76.86% | 99.65% |
| PH | 99.86% | 84.43% | 94.16% | 95.56% | 54.14% | 99.12% | 80.90% |
| TN | 100.00% | 88.58% | 85.46% | 93.61% | 91.10% | 98.12% | 100.00% |
Table 3 The cumulative weight of environmental factors on leaf phenotype in Schisandra sphenanthera
| Factors | Phenotypes’ cumulative weight | ||||||
|---|---|---|---|---|---|---|---|
| LL | LW | PL | LA | LSI | NLT | NSV | |
| TK | 68.83% | 70.92% | 43.92% | 41.06% | 64.25% | 87.10% | 86.75% |
| TOC | 88.62% | 40.85% | 99.68% | 62.39% | 80.33% | 100.00% | 90.38% |
| SLOP | 93.71% | 93.22% | 71.28% | 98.82% | 86.54% | 23.48% | 98.88% |
| TP | 95.84% | 60.79% | 80.15% | 86.14% | 39.75% | 68.24% | 99.88% |
| bio1 | 96.79% | 75.49% | 58.36% | 99.47% | 99.68% | 45.37% | 93.88% |
| ASPE | 97.41% | 97.25% | 100.00% | 79.10% | 100.00% | 82.07% | 99.32% |
| ASL | 97.91% | 91.08% | 27.72% | 71.05% | 97.21% | 94.12% | 95.45% |
| bio12 | 98.40% | 80.04% | 91.53% | 97.26% | 99.10% | 90.88% | 43.38% |
| bio15 | 98.80% | 95.27% | 96.40% | 100.00% | 98.30% | 57.94% | 98.18% |
| bio2 | 99.18% | 100.00% | 98.32% | 91.12% | 95.63% | 96.87% | 96.83% |
| bio4 | 99.55% | 98.97% | 88.84% | 98.14% | 72.85% | 76.86% | 99.65% |
| PH | 99.86% | 84.43% | 94.16% | 95.56% | 54.14% | 99.12% | 80.90% |
| TN | 100.00% | 88.58% | 85.46% | 93.61% | 91.10% | 98.12% | 100.00% |
| [1] | 陈勇, 李芳东, 王海军, 孙冰, 廖绍波, 罗水兴, 蔡刚, 刘东蔚 (2013). 五列木天然群体的表型变异. 中南林业科技大学学报 33(2), 20-23. |
| [2] | 褚延梅, 杨健, 李景吉, 彭培好 (2014). 三种增温情景对入侵植物空心莲子草形态可塑性的影响. 生态学报 34, 1411-1417. |
| [3] | 耿宇鹏, 张文驹, 李博, 陈家宽 (2004). 表型可塑性与外来植物的入侵能力. 生物多样性 12, 447-455. |
| [4] | 顾蔚 (2010). 华中五味子种质资源遗传多样性研究. 博士论文. 西安: 陕西师范大学. |
| [5] | 郭亚奇, 阿里穆斯, 高清竹, 段敏杰, 干珠扎布, 万运帆, 李玉娥, 郭红保 (2011). 灌溉条件下藏北紫花针茅光合特性及其对温度和CO2浓度的短期响应. 植物生态学报 35, 311-321. |
| [6] | 郭彦龙, 顾蔚, 路春燕, 卫海燕 (2013). 基于五味子甲素与乙素的秦岭地区野生华中五味子的潜在空间分布. 植物学报 48, 411-422. |
| [7] | 胡宝清 (2010). 模糊理论基础(第2版). 武汉: 武汉大学出版社. pp. 330-331. |
| [8] | 姜存仓, 郝艳淑, 王晓丽, 夏颖, 王运华 (2011). 钾对不同钾效率棉花基因型叶片解剖结构的影响. 植物营养与肥料学报 17, 1538-1544. |
| [9] | 江建华, 洪德林, 郭媛, 张启武 (2009). 粳稻穗角与谷粒性状的相关性及谷粒性状遗传分析. 植物学报 44, 167-177. |
| [10] | 李乐, 曾辉, 郭大立 (2013). 叶脉网络功能性状及其生态学意义. 植物生态学报 37, 691-698. |
| [11] | 李颖, 姚婧, 杨松, 侯继华 (2014). 东灵山主要树种在不同环境梯度下的叶功能性状研究. 北京林业大学学报 36(1), 72-77. |
| [12] | 刘长柱, 郭强, 池秀莲 (2015). 我国温带山地森林48种常见树种叶片重量-出叶强度的关系. 植物学报 50, 234-240. |
| [13] | 刘柿良, 马明东, 潘远智, 魏刘利, 何成相, 杨开茂 (2013). 不同光环境对桤木幼苗生长和光合特性的影响. 应用生态学报 24, 351-358. |
| [14] | 刘晓, 岳明, 任毅 (2011). 独叶草叶片性状表型多样性研究. 西北植物学报 31, 950-957. |
| [15] | 陆建忠, 裘伟, 陈家宽, 李博 (2005). 入侵种加拿大一枝黄花对土壤特性的影响. 生物多样性 13, 347-356. |
| [16] | 罗成, 顾蔚, 孙茂, 王喆之 (2010). 秦岭地区华中五味子天然居群叶花形态变异研究. 陕西师范大学学报(自然科学版) 38(6), 71-76. |
| [17] | 苗艳明, 吕金枝, 毕润成 (2014). 番薯叶片性状关系模型的建立与验证. 植物学报 49, 190-197. |
| [18] | 潘瑞炽, 陈健源, 温兆清 (1994). 不同钾水平对钾饥饿墨兰生长发育和生理的影响. 热带亚热带植物学报 2(3), 46-53. |
| [19] | 王江民, 陈发棣, 房伟民, 陈素梅, 管志勇, 唐海艳 (2013). 基于叶形特征的切花菊品种鉴别. 植物学报 48, 608-615. |
| [20] | 王坤, 杨继, 陈家宽 (2010). 不同土壤水分和养分条件下喜旱莲子草与同属种生长状况的比较研究. 生物多样性 18, 615-621. |
| [21] | 王蕊, 孙备, 李建东, 王国骄, 孙佳楠, 王欣若, 钟日亭 (2012). 不同光强对入侵种三裂叶豚草表型可塑性的影响. 应用生态学报 23, 1797-1802. |
| [22] | 王志恒, 陈安平, 朴世龙, 方精云 (2004). 高黎贡山种子植物物种丰富度沿海拔梯度的变化. 生物多样性 12, 82-88. |
| [23] | 肖春旺, 张新时, 赵景柱, 吴钢 (2001). 鄂尔多斯高原3种优势灌木幼苗对气候变暖的响应. 植物学报 43, 736-741. |
| [24] | 徐高峰, 申时才, 张付斗, 李天林, 张玉华 (2013). 土壤水分对薇甘菊不同繁殖体单位存活能力和植株表型可塑性影响. 中国农业科学 46, 3134-3141. |
| [25] | 许建平, 张小萍, 曾波, 袁慎鸿, 刘建辉, 刘明智 (2014). 完全水淹环境中光照和溶氧对喜旱莲子草表型可塑性的影响. 生态学报 34, 258-268. |
| [26] | 闫慧, 吴茜, 丁佳, 张守仁 (2013). 不同降水及氮添加对浙江古田山4种树木幼苗光合生理生态特征与生物量的影响. 生态学报 33, 4226-4236. |
| [27] | 杨秉建, 朱敏群, 黄慧青, 陈志锋, 杜小姣 (2014). 不同光照强度对叉花草叶片结构和叶绿素含量的影响. 草原与草坪 34(4), 52-55, 60. |
| [28] | 战伟, 沙伟, 王淼, 师帅, 问青春 (2012). 降水和温度变化对长白山地区水曲柳幼苗生长和光合参数的影响. 应用生态学报 23, 617-624. |
| [29] | 张翠仙, 尼章光, 陈华蕊, 陈于福, 解德宏, 龙亚芹, 张发明 (2014). 怒江干热河谷杧果种质资源的表型和AFLP遗传多样性分析. 植物遗传资源学报 15, 753-758. |
| [30] | 张睿, 国春策, 山红艳, 孔宏智 (2014). 发育重塑与生物多样性. 生物多样性 22, 66-71. |
| [31] | 张亦默, 王卿, 卢蒙, 贾昕, 耿宇鹏, 李博 (2008). 中国东部沿海互花米草种群生活史特征的纬度变异与可塑性. 生物多样性 16, 462-469. |
| [32] | 钟楚, 张明达, 胡雪琼, 朱勇 (2012). 温度变化对烟草光合作用光响应特征的影响. 生态学杂志 31, 337-341. |
| [33] | 中国科学院中国植物志编辑委员会 (1996). 中国植物志(第30卷第1分册). 北京: 科学出版社. pp. 258-260. |
| [34] | 中国科学院西北植物研究所 (1974). 秦岭植物志(第1卷第2分册). 北京: 科学出版社. pp. 341-342. |
| [35] | 中国药典委员会 (2010). 中华人民共和国药典(第1部). 北京: 中国医药科技出版社. pp. 227-228. |
| [36] | 钟梦莹, 樊青丽, 张亚军, 武瑞鑫, 阚雨晨, 王建勋, 邵新庆 (2013). 矮化羊草形态可塑性及生物量分配模式研究. 草地学报 21, 260-264. |
| [37] | 朱建平 (2012). 应用多元统计分析(第2版). 北京: 科学出版社. pp. 93-100. |
| [38] | 朱燕华 (2013). 东亚地区栓皮栎(Quercus variabilis)叶片性状的变异格局及其对环境变化的响应. 博士论文. 上海: 上海交通大学. |
| [39] | 卓露, 管开云, 李文军, 段士民 (2014). 不同生境下细叶鸢尾表型可塑性及生物量分配差异性. 生态学杂志 33, 618-623. |
| [40] | Abraham EM, Kyriazopoulos AP, Parissi ZM, Kosto- poulou P, Karatassiou M, Anjalanidou K, Katsouta C (2014). Growth, dry matter production, phenotypic plasticity, and nutritive value of three natural populations of Dactylis glomerata L. under various shading treatments.Agroforest Syst 88, 287-299. |
| [41] | Böhnke M, Bruelheide H (2013). How do evergreen and deciduous species respond to shade?—Tolerance and plasticity of subtropical tree and shrub species of South- East China.Environ Exp Bot 87, 179-190. |
| [42] | Bradshaw AD (1965). Evolutionary significance of phenol- typic plasticity in plants.Adv Genet 13, 115-155. |
| [43] | Dormann CF, McPherson JM, Araújo MB, Bivand R, Bolliger J, Carl G, Davies RG, Hirzel A, Jetz W, Kissling WD, Kühn I, Ohlemüller R, Peres-Neto PR, Reineking B, Schröder B, Schurr FM, Wilson R (2007). Methods to account for spatial auto correlation in the analysis of species distributional data: a review.Ecography 30, 609-628. |
| [44] | Droste T, Flory SL, Clay K (2010). Variation for phenotypic plasticity among populations of an invasive exotic grass.Plant Ecol 207, 297-306. |
| [45] | Gianoli E, Valladares F (2012). Studying phenotypic plas- ticity: the advantages of a broad approach.Biol J Linn Soc 105, 1-7. |
| [46] | Griffith AB, Andonian K, Weiss CP, Loik ME (2014). Variation in phenotypic plasticity for native and invasive populations of Bromus tectorum.Biol Invasions 16, 2627-2638. |
| [47] | Jaryan V, Datta A, Uniyal SK, Kumar A, Gupta RC, Singh RD (2013). Modelling potential distribution of Sapium sebiferum—an invasive tree species in western Himalaya.Curr Sci India 105, 1282-1288. |
| [48] | Lamarque LJ, Lortie CJ, Porté AJ, Delzon S (2014). Ge- netic differentiation and phenotypic plasticity in life-history traits between native and introduced populations of invasive maple trees.Biol Invasions 17, 1109-1122. |
| [49] | Lu CY, Gu W, Dai AH, Wei HY (2012). Assessing habitat suitability based on geographic information system (GIS) and fuzzy: a case study of Schisandra sphenanthera Rehd. et Wils. in Qinling Mountains, China.Ecol Model 242, 105-115. |
| [50] | Peltonen-Sainio P, Jauhiainen L, Sadras VO (2011). Phenotypic plasticity of yield and agronomic traits in cereals and rapeseed at high latitudes.Field Crop Res 124, 261-269. |
| [51] | Pigliucci M, Tyler GA, Schlichting CD (1998). Mutational effects on constraints on character evolution and phenol- typic plasticity in Arabidopsis thaliana.J Genet 77, 95-103. |
| [52] | Rejmánek M, Richardson DM (1996). What attributes make some plant species more invasive?Ecology 77, 1655-1661. |
| [53] | Royer DL, McElwain JC, Adams JM, Wilf P (2008). Sensitivity of leaf size and shape to climate within Acer rubrum and Quercus kelloggii.New Phytol 179, 808-817. |
| [54] | Royer DL, Wilf P, Janesko DA, Kowalski EA, Dilcher DL (2005). Correlations of climate and plant ecology to leaf size and shape: potential proxies for the fossil record.Am J Bot 92, 1141-1151. |
| [55] | Schlichting CD (1986). The evolution of phenotypic plas- ticity in plants.Annu Rev Ecol Syst 17, 667-693. |
| [56] | Siddiqui F, Herial NA, Ali II (2010). Cumulative effect of vagus nerve stimulators on intractable seizures observed over a period of 3 years.Epilepsy Behav 18, 299-302. |
| [57] | Sultan SE (1995). Phenotypic plasticity and plant adapta- tion.Acta Bot Neerl 44, 363-383. |
| [58] | Sultan SE (2000). Phenotypic plasticity for plant development, function and life history.Trends Plant Sci 5, 537-542. |
| [59] | Sultan SE (2010). Plant developmental responses to the environment eco-devo.Curr Opin Plant Biol 13, 96-101. |
| [60] | Valladares F, Sánchez-Gómez D, Zavala MA (2006). Quantitative estimation of phenotypic plasticity: bridging the gap between the evolutionary concept and its ecological applications.J Ecol 94, 1103-1116. |
| [61] | Weber TC (2011). Maximum entropy modelling of mature hardwood forest distribution in four U.S. states.Forest Ecol Manag 261, 779-788. |
| [62] | Yang XQ, Kushwaha SPS, Saran S, Xu JC, Roy PS (2013). Maxent modelling for predicting the potential distribution of medicinal plant, Justicia adhatoda L. in Lesser Himalayan foothills.Ecol Eng 51, 83-87. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||