

Chinese Bulletin of Botany ›› 2016, Vol. 51 ›› Issue (4): 504-514.DOI: 10.11983/CBB16028
• EXPERIMENTAL COMMUNICATIONS • Previous Articles Next Articles
Tao Liu, Haiying Chen, Haiying Yu, Tingxuan Li, Shangqing Gao, Guangdeng Chen*
Received:2016-02-18
Accepted:2016-05-03
Online:2016-07-01
Published:2016-08-05
Contact:
Chen Guangdeng
About author:# Co-first authors
Tao Liu, Haiying Chen, Haiying Yu, Tingxuan Li, Shangqing Gao, Guangdeng Chen. Characterization of Phosphorus Utilization in Barley Leaf under Low Phosphorus Stress[J]. Chinese Bulletin of Botany, 2016, 51(4): 504-514.
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.165ca | 0.042ba | 0.105ca | 0.030ba | |
| DH147 | 0.162ca | 0.041aba | 0.111ca | 0.029ba | ||
| DH49 | 0.122cb | 0.043ca | 0.104ca | 0.031ba | ||
| P50 | DH110 | 0.176ba | 0.043bb | 0.170ba | 0.029bb | |
| DH147 | 0.177ba | 0.041bb | 0.162bab | 0.031bab | ||
| DH49 | 0.144bb | 0.048ba | 0.151bb | 0.034ba | ||
| P75 | DH110 | 0.230aa | 0.047ab | 0.225aa | 0.039ab | |
| DH147 | 0.227aa | 0.046ab | 0.226aa | 0.036ab | ||
| DH49 | 0.199ab | 0.053aa | 0.220aa | 0.044aa | ||
Table 1 Changes of Pi content in leaves of barley under different P conditions (mg·g-1 FW)
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.165ca | 0.042ba | 0.105ca | 0.030ba | |
| DH147 | 0.162ca | 0.041aba | 0.111ca | 0.029ba | ||
| DH49 | 0.122cb | 0.043ca | 0.104ca | 0.031ba | ||
| P50 | DH110 | 0.176ba | 0.043bb | 0.170ba | 0.029bb | |
| DH147 | 0.177ba | 0.041bb | 0.162bab | 0.031bab | ||
| DH49 | 0.144bb | 0.048ba | 0.151bb | 0.034ba | ||
| P75 | DH110 | 0.230aa | 0.047ab | 0.225aa | 0.039ab | |
| DH147 | 0.227aa | 0.046ab | 0.226aa | 0.036ab | ||
| DH49 | 0.199ab | 0.053aa | 0.220aa | 0.044aa | ||
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.105aa | 0.028bb | 0.109aa | 0.019cb | |
| DH147 | 0.102aa | 0.030cb | 0.110aa | 0.021cb | ||
| DH49 | 0.085bb | 0.044ba | 0.094ab | 0.035ca | ||
| P50 | DH110 | 0.102aa | 0.035bb | 0.105aa | 0.028bb | |
| DH147 | 0.106aa | 0.041bb | 0.109aa | 0.026bb | ||
| DH49 | 0.093abb | 0.058aa | 0.097aa | 0.043ba | ||
| P75 | DH110 | 0.104aa | 0.049ab | 0.108aab | 0.041ab | |
| DH147 | 0.103aa | 0.045ab | 0.113aa | 0.041ab | ||
| DH49 | 0.101aa | 0.063aa | 0.101ab | 0.052aa | ||
Table 2 Changes of ester P content in leaves of barley under different P conditions (mg·g-1 FW)
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.105aa | 0.028bb | 0.109aa | 0.019cb | |
| DH147 | 0.102aa | 0.030cb | 0.110aa | 0.021cb | ||
| DH49 | 0.085bb | 0.044ba | 0.094ab | 0.035ca | ||
| P50 | DH110 | 0.102aa | 0.035bb | 0.105aa | 0.028bb | |
| DH147 | 0.106aa | 0.041bb | 0.109aa | 0.026bb | ||
| DH49 | 0.093abb | 0.058aa | 0.097aa | 0.043ba | ||
| P75 | DH110 | 0.104aa | 0.049ab | 0.108aab | 0.041ab | |
| DH147 | 0.103aa | 0.045ab | 0.113aa | 0.041ab | ||
| DH49 | 0.101aa | 0.063aa | 0.101ab | 0.052aa | ||
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.236ba | 0.074cb | 0.252cb | 0.062cb | |
| DH147 | 0.234ba | 0.067bc | 0.262ba | 0.058cc | ||
| DH49 | 0.185bb | 0.081ba | 0.220cc | 0.074ba | ||
| P50 | DH110 | 0.258aa | 0.077bc | 0.273aba | 0.082bb | |
| DH147 | 0.254aa | 0.085bb | 0.264ba | 0.075bc | ||
| DH49 | 0.243ab | 0.095aa | 0.246bb | 0.094aa | ||
| P75 | DH110 | 0.253aa | 0.096aa | 0.277aab | 0.088ab | |
| DH147 | 0.252aa | 0.094aa | 0.283aa | 0.096aab | ||
| DH49 | 0.246aa | 0.096aa | 0.268ab | 0.093aa | ||
Table 3 Changes of nucleic P concentration in leaves of barley under different P conditions (mg·g-1 FW)
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.236ba | 0.074cb | 0.252cb | 0.062cb | |
| DH147 | 0.234ba | 0.067bc | 0.262ba | 0.058cc | ||
| DH49 | 0.185bb | 0.081ba | 0.220cc | 0.074ba | ||
| P50 | DH110 | 0.258aa | 0.077bc | 0.273aba | 0.082bb | |
| DH147 | 0.254aa | 0.085bb | 0.264ba | 0.075bc | ||
| DH49 | 0.243ab | 0.095aa | 0.246bb | 0.094aa | ||
| P75 | DH110 | 0.253aa | 0.096aa | 0.277aab | 0.088ab | |
| DH147 | 0.252aa | 0.094aa | 0.283aa | 0.096aab | ||
| DH49 | 0.246aa | 0.096aa | 0.268ab | 0.093aa | ||
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.044aa | 0.031aa | 0.043aa | 0.032aa | |
| DH147 | 0.046aa | 0.030aa | 0.042aa | 0.029aa | ||
| DH49 | 0.037bb | 0.031aa | 0.035bb | 0.030aa | ||
| P50 | DH110 | 0.046aa | 0.030aa | 0.043aa | 0.030aa | |
| DH147 | 0.045aa | 0.030aa | 0.043aa | 0.028aa | ||
| DH49 | 0.042aa | 0.031aa | 0.041aa | 0.030aa | ||
| P75 | DH110 | 0.047aa | 0.029aa | 0.043aa | 0.027ab | |
| DH147 | 0.046aa | 0.031aa | 0.042aa | 0.028aab | ||
| DH49 | 0.044aa | 0.029aa | 0.042aa | 0.030aa | ||
Table 4 Changes of insoluble P concentration in leaves of barley under different P conditions (mg·g-1 FW)
| Treatments | Genotypes | Booting stage | Heading stage | |||
|---|---|---|---|---|---|---|
| Upper leaves | Bottom leaves | Upper leaves | Bottom leaves | |||
| P25 | DH110 | 0.044aa | 0.031aa | 0.043aa | 0.032aa | |
| DH147 | 0.046aa | 0.030aa | 0.042aa | 0.029aa | ||
| DH49 | 0.037bb | 0.031aa | 0.035bb | 0.030aa | ||
| P50 | DH110 | 0.046aa | 0.030aa | 0.043aa | 0.030aa | |
| DH147 | 0.045aa | 0.030aa | 0.043aa | 0.028aa | ||
| DH49 | 0.042aa | 0.031aa | 0.041aa | 0.030aa | ||
| P75 | DH110 | 0.047aa | 0.029aa | 0.043aa | 0.027ab | |
| DH147 | 0.046aa | 0.031aa | 0.042aa | 0.028aab | ||
| DH49 | 0.044aa | 0.029aa | 0.042aa | 0.030aa | ||
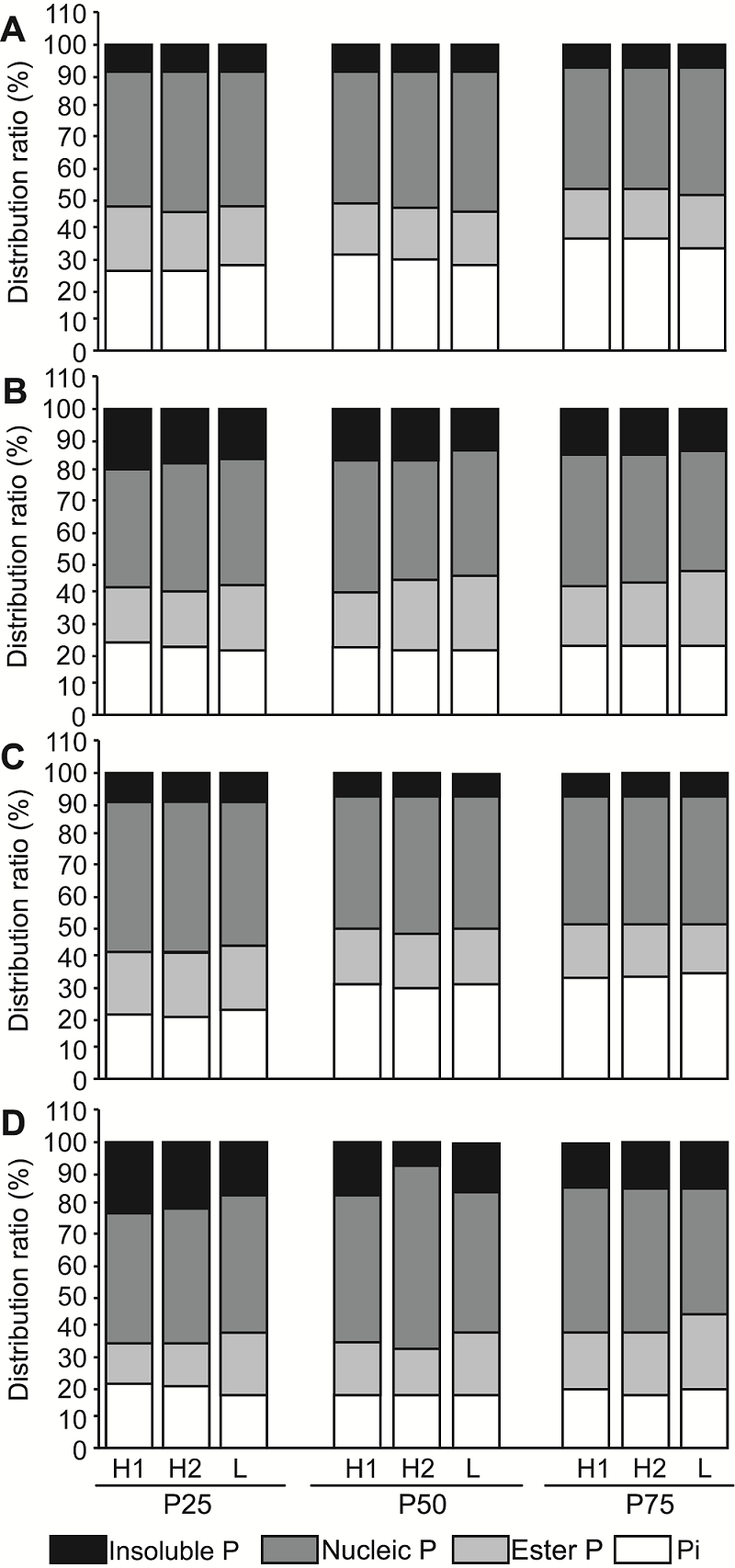
Figure 2 The differences in distribution of P fractions in upper (A, C) and bottom (B, D) leaves of barley under different P conditions at booting (A, B) and heading (C, D) stages H1, H2 and L represent P efficient barley genotypes DH110, DH147 and P inefficient barley genotype DH49, respectively.
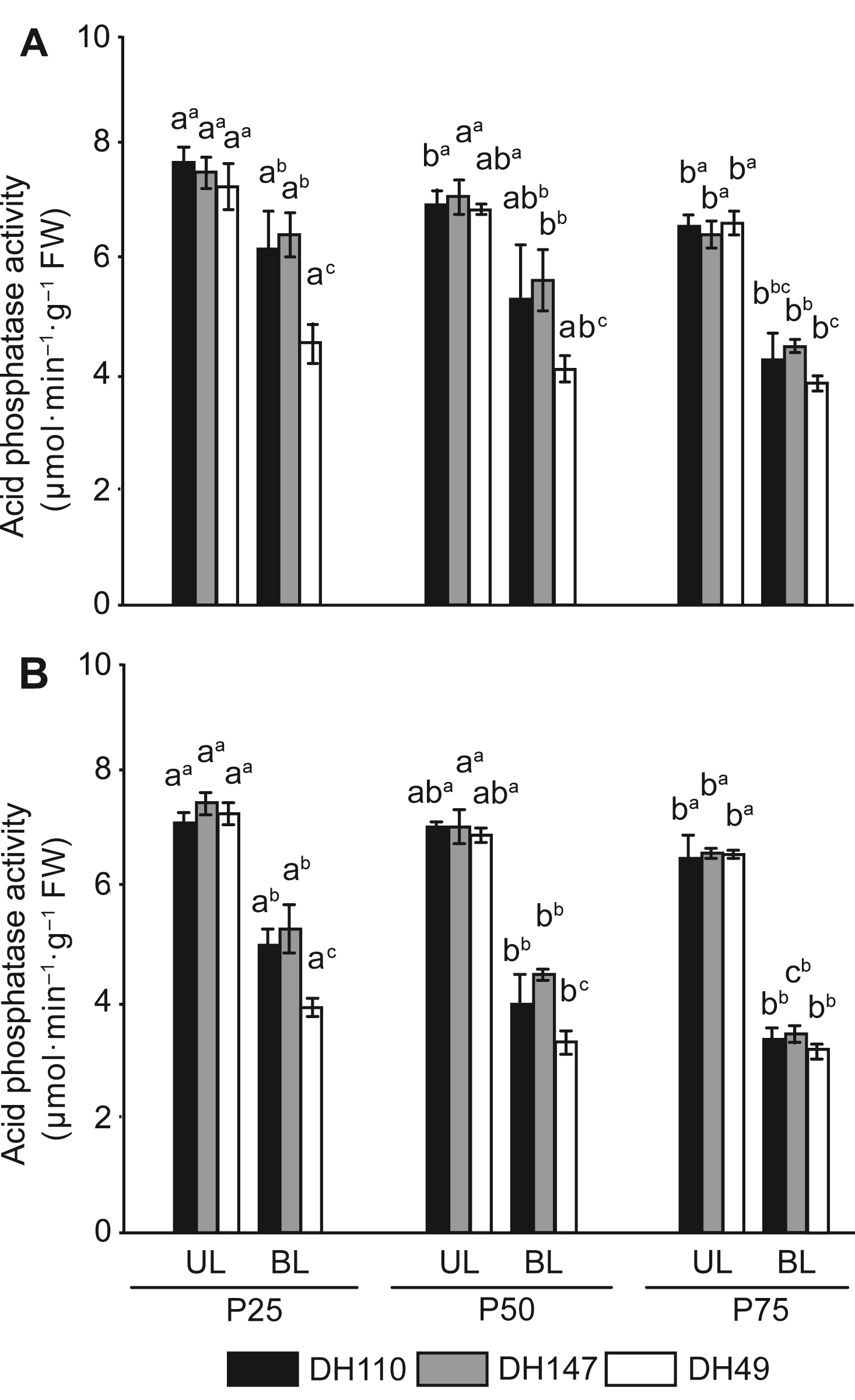
Figure 3 Changes of nucleic acid phosphatase activity in leaves of barley under different P conditions at booting (A) and heading (B) stages UL: Upper leaves; BL: Bottom leaves. Different letters indicate significant difference at P<0.05 among different P conditions; while different superscript letters indicate significant difference at P<0.05 between the two genotypes.
| Organs | Indexes | Acid phosphatase activity (μmol·min-1·g-1 FW) | PUE for dry matter (g·g-1 P) |
|---|---|---|---|
| Upper leaves | Pi | -0.433 | -0.599** |
| Ester P | 0.122 | 0.286 | |
| Nucleic P | -0.332 | -0.166 | |
| Insoluble P | 0.391 | 0.058 | |
| Bottom leaves | Pi | 0.005 | -0.508* |
| Ester P | -0.742** | -0.906** | |
| Nucleic P | -0.657** | -0.922** | |
| Insoluble P | 0.412 | 0.410 |
Table 5 Relationship between P fractions and acid phosphatase activity, P utilization efficiency of barley in different parts of leaves
| Organs | Indexes | Acid phosphatase activity (μmol·min-1·g-1 FW) | PUE for dry matter (g·g-1 P) |
|---|---|---|---|
| Upper leaves | Pi | -0.433 | -0.599** |
| Ester P | 0.122 | 0.286 | |
| Nucleic P | -0.332 | -0.166 | |
| Insoluble P | 0.391 | 0.058 | |
| Bottom leaves | Pi | 0.005 | -0.508* |
| Ester P | -0.742** | -0.906** | |
| Nucleic P | -0.657** | -0.922** | |
| Insoluble P | 0.412 | 0.410 |
| 1 | 陈海英, 余海英, 陈光登, 李廷轩 (2015). 低磷胁迫下磷高效基因型大麦的根系形态特征. 应用生态学报 26, 3020-3026. |
| 2 | 段海燕, 徐芳森, 王运华 (2002). 甘蓝型油菜不同磷效率品种苗期根系生长及磷营养的差异. 植物营养与肥料学报 8, 65-69. |
| 3 | 郭程瑾, 李宾兴, 王斌, 李雁鸣, 肖凯 (2006). 小麦高效吸收和利用磷素的生理机制. 作物学报 32, 827-832. |
| 4 | 黄彩霞, 柴守玺, 赵德明, 常磊, 程宏波, 杨长刚 (2015). 氮磷肥配施对冬小麦灌浆期光合参数及产量的影响. 植物学报 50, 47-54. |
| 5 | 刘渊, 李喜焕, 孙星, 张彩英 (2012). 磷胁迫下大豆酸性磷酸酶活性变化及磷效率基因型差异分析. 植物遗传资源学报13, 521-528. |
| 6 | 鲁如坤 (2000). 土壤农业化学分析方法. 北京: 中国农业科技出版社. pp. 56-57. |
| 7 | 石磊, 梁宏玲, 徐芳森, 王运华 (2008). 甘蓝型油菜幼苗体内磷组分差异与磷高效关系的研究. 植物营养与肥料学报 14, 351-356. |
| 8 | 王丽, 王芹芹, 王幼群 (2014). 蚕豆叶片小叶脉不同发育时期ATP酶和酸性磷酸酶的细胞化学超微结构定位. 植物学报 49, 78-86. |
| 9 | 吴沂珀, 张锡洲, 李廷轩, 阳显斌, 吴德勇 (2013). 小麦不同磷效率品种对不同磷源的利用差异及酸性磷酸酶的作用. 核农学报 27, 351-357. |
| 10 | 易雪梅 (2012). 大麦磷高效基因型筛选及其生理生化特征. 硕士论文. 成都: 四川农业大学. pp. 8-28. |
| 11 | 袁硕, 李春俭, 彭正萍, 彭云峰, 王红, 张丽娟 (2011). 磷对不同玉米品种生长, 体内磷循环和分配的影响. 植物营养与肥料学报 17, 310-316. |
| 12 | 张海伟, 黄宇, 叶祥盛, 徐芳森 (2010). 低磷胁迫下甘蓝型油菜酸性磷酸酶对磷效率的贡献分析. 中国科学: 生命科学 40, 418-427. |
| 13 | 赵华, 徐芳森, 石磊, 王运华 (2006). 植物根系形态对低磷胁迫应答的研究进展. 植物学通报 23, 409-417. |
| 14 | Bargaz A, Faghire M, Abdi N, Farissi M, Sifi B, Drevon JJ, Ikbal MC, Ghoulam C (2012). Low soil phosphorus availability increases acid phosphatases activities and affects P partitioning in nodules, seeds and rhizosphere of Phaseolus vulgaris.Agriculture 2, 139-153. |
| 15 | Cai J, Chen L, Qu H, Lian J, Liu W, Hu YB, Xu GH (2012). Alteration of nutrient allocation and transporter genes expression in rice under N, P, K, and Mg deficiencies.Acta Physiol Plant 34, 939-946. |
| 16 | Chapin III FS, Kedrowski RA (1983). Seasonal changes in nitrogen and phosphorus fractions and autumn retranslocation in evergreen and deciduous taiga trees.Ecology 64, 376-391. |
| 17 | Close DC, Beadle CL (2004). Total, and chemical fractions, of nitrogen and phosphorus in Eucalyptus seedling leaves: effects of species, nursery fertiliser management and transplanting.Plant Soil 259, 85-95. |
| 18 | Dention MD, Veneklaas EJ, Freimoser FM, Lambers H (2007). Banksia species (Proteaceae) from severely pho- sphorus-impoverished soils exhibit extreme efficiency in the use and remobilization of phosphorus.Plant Cell Environ 30, 1557-1565. |
| 19 | Feuillet C, Muehlbauer GJ (2009). Genetics and Genomics of the Triticeae, Plant Genetics and Genomics: Crops and Models. Berlin: Springer. Vol. 7. pp. 685-711. |
| 20 | George TS, Gregory PJ, Hocking P, Richardson AE (2008). Variation in root-associated phosphatase activities in wheat contributes to the utilization of organic P substrates in vitro, but does not explain differences in the P-nutrition of plants when grown in soils. Environ Exp Bot 64, 239-249. |
| 21 | Hammond JP, Broadley MR, White PJ, King GJ, Bowen HC, Hayden RM, Meacham MC, Mead A, Overs TS, William P, Greenwood DJ (2009). Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits.J Exp Bot 60, 1953-1968. |
| 22 | Hart AL, Jessop D (1983). Phosphorus fractions in trifoliate leaves of white clover and lotus at various levels of phosphorus supply.New Zeal J Agr Res 26, 357-361. |
| 23 | Hidaka A, Kitayama K (2011). Allocation of foliar phosphorus fractions and leaf traits of tropical tree species in response to decreased soil phosphorus availability on Mount Kinabalu, Borneo.J Eco 99, 849-857. |
| 24 | Huang CY, Shirley N, Genc Y, Shi BJ, Langridge P (2011). Phosphate utilization efficiency correlates with expression of low-affinity phosphate transporters and noncoding RNA, IPS1, in barley. Plant Physiol 156, 1217-1229. |
| 25 | Karthikeyan AS, Varadarajan DK, Jain A, Held MA, Carpita NC, Raghothama KG (2007). Phosphate starvation responses are mediated by sugar signaling in Arabidopsis. Planta 225, 907-918. |
| 26 | Kedrowski RA (2013). Extraction and analysis of nitrogen, phosphorus and carbon fractions in plant material.J Plant Nutr 6, 989-1011. |
| 27 | Lambers H, Finnegan PM, Laliberté E, Pearse SJ, Ryan MH, Shane MW, Veneklaas EJ (2011). Phosphorus nutrition of Proteaceae in severely phosphorus-impoveri- shed soils: are there lessons to be learned for future crops.Plant Physiol 156, 1058-1066. |
| 28 | Parentoni SN, Souza Júnior CL (2008). Phosphorus acquisition and internal utilization efficiency in tropical maize genotypes.Pesqui Agropecu Bras 43, 893-901. |
| 29 | Peng Z, Li C (2005). Transport and partitioning of phosphorus in wheat as affected by P withdrawal during flag-leaf expansion.Plant Soil 268, 1-11. |
| 30 | Pratt J, Boisson AM, Gout E, Bligny R, Douce R, Aubert S (2009). Phosphate (Pi) starvation effect on the cytosolic Pi concentration and Pi exchanges across the tonoplast in plant cells: an in vivo 31P-nuclear magnetic resonance study using methylphosphonate as a Pi analog.Plant Physio 151, 1646-1657. |
| 31 | Rose TJ, Hardiputra B, Rengel Z (2010). Wheat, canola and grain legume access to soil phosphorus fractions differs in soils with contrasting phosphorus dynamics.Plant Soil 326, 159-170. |
| 32 | Rose TJ, Rengel Z, Ma Q, Bowden JW (2008). Post- flowering supply of P, but not K, is required for maximum canola seed yields.Eur J Agron 28, 371-379. |
| 33 | Rose TJ, Rose MT, Pariasca-Tanaka J, Heuer S, Wissuwa M (2011). The frustration with utilization: why have improvements in internal phosphorus utilization efficiency in crops remained so elusive.Front Plant Sci 10, 371-379. |
| 34 | Rouached H, Stefanovic A, Secco D, Bulak A, Gout E, Bligny R, Poirier Y (2011). Uncoupling phosphate deficiency from its major effects on growth and transcriptome via PHO1 expression in Arabidopsis.Plant J 65, 557-570. |
| 35 | Su J, Xiao Y, Li M, Liu Q, Li B, Tong Y, Jia J, Li Z (2006). Mapping QTLs for phosphorus-deficiency tolerance at wheat seedling stage.Plant Soil 281, 25-36. |
| 36 | Tabaldi LA, Ruppenthal R, Cargnelutti D, Morsch VM, Pereira LB, Schetinger MRC (2007). Effects of metal elements on acid phosphatase activity in cucumber (Cucumis sativus L.) seedlings.Environ Exp Bot 59, 43-48. |
| 37 | Tian J, Wang C, Zhang Q, He XW, Whelan J, Shou HX (2012a). Overexpression of OsPAP10a, a root-associated acid phosphatase, increased extracellular organic phosphorus utilization in rice.J Integr Plant Biol 54, 631-639. |
| 38 | Tian J, Wang X, Tong Y, Chen X, Liao H (2012b). Bioengineering and management for efficient phosphorus utilization in crops and pastures.Curr Opin Biotech 23, 866-871. |
| 39 | Veneklaas EJ, Lambers H, Bragg J, Finnegan PM, Lovelock CE, Plaxton WC, Price CA, Scheible WR, Shane MW, White PJ (2012). Opportunities for improving phosphorus-use efficiency in crop plants.New Phytol 195, 306-320. |
| 40 | Wieneke J (1990). Phosphorus efficiency and phosphorus remobilization in two sorghum (Sorghum bicolor (L.) Moench) cultivars.Plant Soil 123, 139-145. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||