

Chinese Bulletin of Botany ›› 2016, Vol. 51 ›› Issue (6): 790-800.DOI: 10.11983/CBB15226
Previous Articles Next Articles
Ruixue Yang†, Haiyang Liu†, Shengli Liu, Tingqiao Yu, Yuzhen Chen*, Cunfu Lu*
Received:2015-12-30
Accepted:2016-03-21
Online:2016-11-01
Published:2016-12-02
Contact:
Chen Yuzhen,Lu Cunfu
About author:# Co-first authors
Ruixue Yang, Haiyang Liu, Shengli Liu, Tingqiao Yu, Yuzhen Chen, Cunfu Lu. Optimization of Detection Methods for Zea mays ABA Receptor ZmPYL1 by Isothermal Titration Calorimetry[J]. Chinese Bulletin of Botany, 2016, 51(6): 790-800.
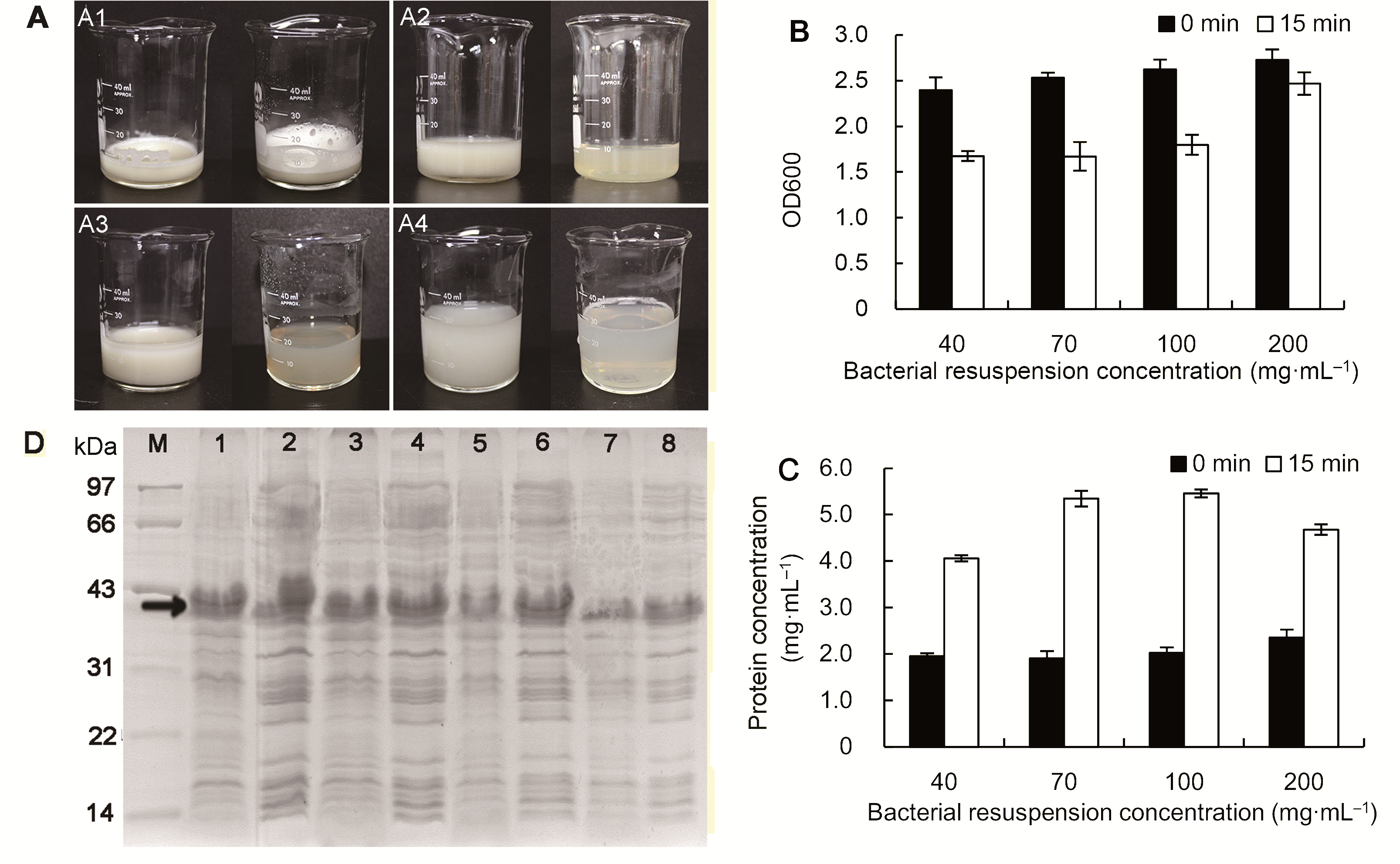
Figure 2 Ultrasonic treatments for bacterial resuspension of different concentrations (A) A1-A4 indicate the comparison of 200, 100, 70 and 40 mg·mL-1 bacterial suspension before (left beaker) and after (right beaker) ultrasonic treatments; (B) Effect of bacterial resuspension of different concentrations on the OD600 before (0 min) and after (15 min) ultrasonic treatments; (C) Effect of bacterial resuspension of different concentrations on the protein content before (0 min) and after (15 min) ultrasonic treatments; (D) SDS-PAGE detection of protein content in sediment and supernatant of different bacterial concentration, lane 1, 3, 5, 7 represent the protein content in sediment of 200, 100, 70 and 40 mg·mL-1 bacterial suspension, respectively; and lane 2, 4, 6, 8 represent the protein content in supernatant of 200, 100, 70 and 40 mg·mL-1 bacterial suspension, respectively. Target protein band is showed by arrow.

Figure 3 Ultrasonic treatment of inclusion body ZmPYL4 (A) Effect of total ultrasonic time on the OD600 of bacterial resuspension and the protein content in supernatant; (B) Bacterial resuspension before (left beaker) and after (right beaker) ultrasonic treatment for 30 min; (C) SDS-PAGE detection of protein level in sediment and supernatant of bacterial suspension. Lane 1: The protein level in sediment before ultrasonic treatment. Lane 2: The protein level in supernatant before ultrasonic treatment. Lane 3: The protein level in sediment after ultrasonic treatment for 30 min. Lane 4: The protein level in supernatant after ultrasonic treatment for 30 min.
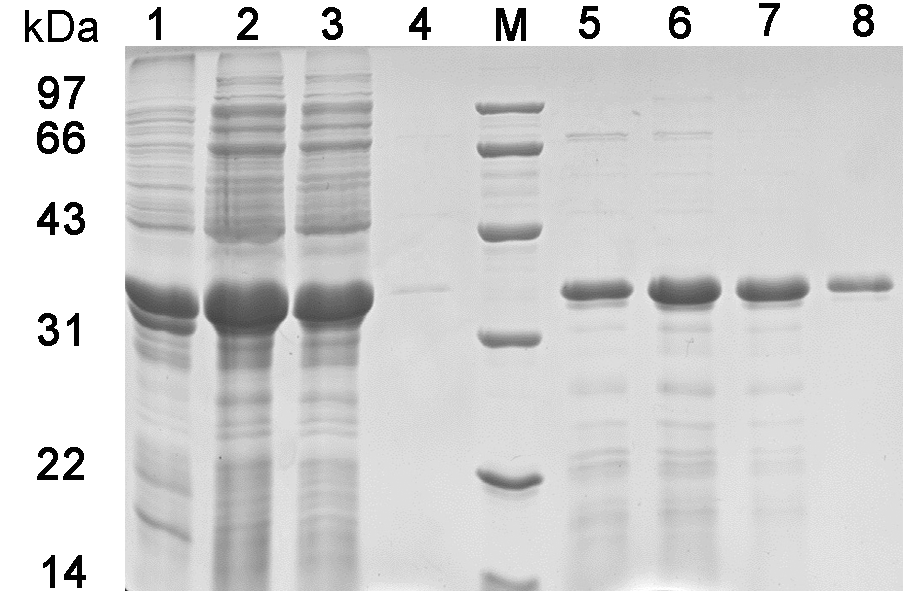
Figure 6 SDS-PAGE detection of purified ZmPYL1 protein Lane 1: The protein level in sediment after ultrasonic treatment; Lane 2: The protein level in supernatant after ultrasonic treatment; Lane 3: The collecting liquid of the supernatant through Ni-NTA; Lane 4: The collecting liquid of wash buffer through Ni-NTA; Lane 5-8: The collecting liquid of elution buffer through Ni-NTA for the first, second, third and fourth time, respectively.
| Thermodynamic parameters | 2 mmol·L-1 (±)-ABA+ 0.005 mmol·L-1 ZmPYL1 | 2 mmol·L-1 (±)-ABA+ 0.1 mmol·L-1 ZmPYL1 | 4 mmol·L-1 (±)-ABA+ 0.1 mmol·L-1 ZmPYL1 |
|---|---|---|---|
| N (sites) | 15.5±5.52 | 0.651±0.190 | 0.821±0.069 |
| Kd (μmol·L-1) | 135.32 | 72.99 | 72.46 |
Table 1 The thermodynamic parameters of (±)-ABA and ZmPYL1 protein in different concentrations
| Thermodynamic parameters | 2 mmol·L-1 (±)-ABA+ 0.005 mmol·L-1 ZmPYL1 | 2 mmol·L-1 (±)-ABA+ 0.1 mmol·L-1 ZmPYL1 | 4 mmol·L-1 (±)-ABA+ 0.1 mmol·L-1 ZmPYL1 |
|---|---|---|---|
| N (sites) | 15.5±5.52 | 0.651±0.190 | 0.821±0.069 |
| Kd (μmol·L-1) | 135.32 | 72.99 | 72.46 |
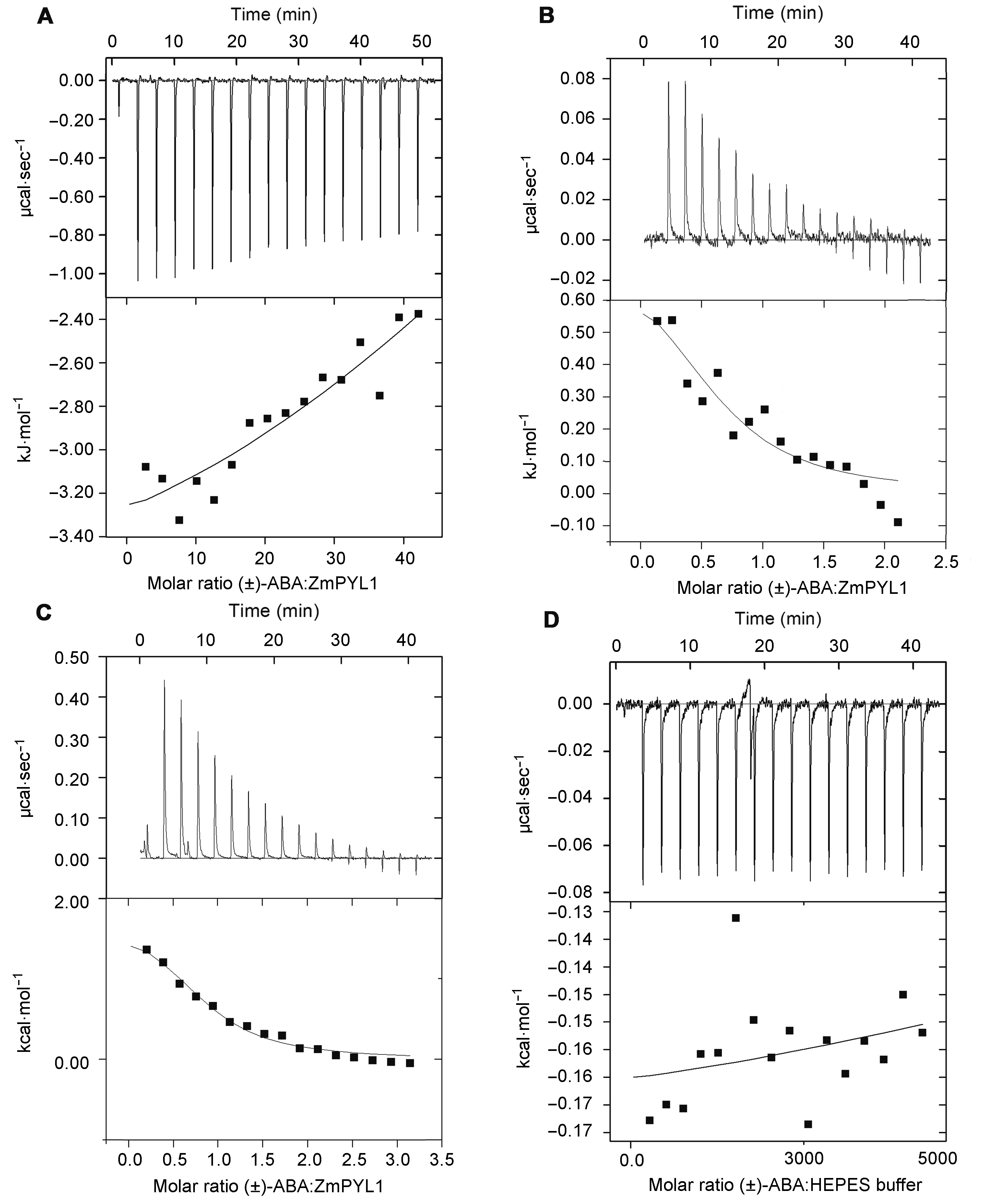
Figure 7 The variation of differential power with time and the heat variation of injection with the molar ratio(A) 2 mmol·L-1 (±)-ABA was titrated into 0.05 mmol·L-1 ZmPYL1; (B) 2 mmol·L-1 (±)-ABA was titrated into 0.1 mmol·L-1 ZmPYL1; (C) 4 mmol·L-1 (±)-ABA was titrated into 0.1 mmol·L-1 ZmPYL1; (D) 4 mmol·L-1 (±)-ABA was titrated into HEPES solution without protein. The solid lines in bottom panels represent the fitting curves, and the closer the fitting curve is to experimental dots, the more credible the fitting result is.
| [1] | 邓洁, 程龙, 刘谊, 白林含, 于昕, 鲍锦库, 曹毅, 乔代蓉 (2006). 根肿(Plasmodiophora brassicae)休眠孢子的纯化和超声波破碎方法研究. 应用与环境生物学报 12, 269-272. |
| [2] | 黄卓楠 (2013). 微量量热技术测定酶催化反应的研究进展. 分析仪器 5, 1-5. |
| [3] | 金德善, 卢存福, 兰小中, 刘雪萍, 乔佩, 李红英, 陈玉珍 (2012). 雪莲愈伤组织蛋白质提取及双向电泳分析. 西北植物学报 32, 1903-1909. |
| [4] | 李永霞, 曾海英, 秦礼康 (2010). 酵母细胞破碎条件优化及高肽酶菌株筛选. 食品科学 13, 302-306. |
| [5] | 石会刚, 陈东戎 (2015). 等温滴定量热法研究核糖开关acc与氨基糖苷类抗生素的相互作用. 生物物理学报 31, 241-250. |
| [6] | 吴蕾, 雷鸣, 洪建辉, 甘一如 (2002). 超声破碎重组大肠杆菌释放包含体的过程研究. 化学工业与工程 19, 422-425. |
| [7] | 易文凯, 王佳, 杨辉, 田云, 卢向阳 (2012). 植物ABA受体及其介导的信号转导通路. 植物学报 47, 515-524. |
| [8] | 周洁, 付强, 玉延华 (2013). 等温滴定微量热检测中样品预处理的优化研究. 化学与生物工程 30(9), 72-74. |
| [9] | 周洁, 玉延华, 朱平川, 付强, 胡炜 (2014). 圆二色和等温滴定微量热技术研究盐酸麻黄碱、盐酸伪麻黄碱与人血清白蛋白的作用机制. 分析试验室 33, 432-435. |
| [10] | Bradford MM (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding.Anal Biochem 72, 248-254. |
| [11] | Canas B, Pineiro C, Calvo E, Lopez-Ferrer D, Gallardo JM (2007). Trends in sample preparation for classical and second generation proteomics.J Chromatogr A 1153, 235-258. |
| [12] | Ding F, Sun Y, Diao JX, Li XN, Yang XL, Sun Y, Zhang L (2012). Features of the complex of food additive hesperidin to hemoglobin.J Photochem Photobiol B 106, 53-60. |
| [13] | Draczkowski P, Matosiuk D, Jozwiak K (2014). Isothermal titration calorimetry in membrane protein research.J Ph- arm Biomed Anal 87, 313-325. |
| [14] | Falconera RJ, Collinsb BM (2011). Survey of the year 2009: applications of isothermal titration calorimetry.J Mol Recognit 24, 1-16. |
| [15] | Florine D, Santiago J, Betz K, Twycross J, Park SY, Rodriguez L, Guzman MG, Jensen MR, Krasnogor N, Blackledge M, Holdsworth M, Cutler SR, Rodriguez PL, Márquez JA (2011). A thermodynamic switch modulates abscisic acid receptor sensitivity.EMBO J 30, 4171-4184. |
| [16] | Hanson WM, Domek GJ, Horvath MP, Goldenberg DP (2007). Rigidification of a flexible potease inhibitor variant upon binding to trypsin.J Mol Biol 366, 230-243. |
| [17] | Hao Q, Yin P, Li WQ, Wang L, Yan CY, Lin ZH, Wu JZ, Wang JW, Yan SF, Yan N (2011). The molecular basis of ABA-independent inhibition of PP2Cs by a subclass of PYL proteins. Mol Cell 42, 662-672. |
| [18] | Hytonen VP, Maatta JAE, Niskanan EA, Huuskonen J, Helttunen KJ, Halling KK, Nordlund HR, Rissanen K, Johnson MS, Salminen TA, Kulomaa MS, Laitinen OH, Airenne TT (2007). Structure and characterization of a novel chicken biotin-binding protein A (BBP-A).BMC Struct Biol 7(8), 1-20. |
| [19] | Li G, Xin H, Zheng XF, Li S, Hu Z (2012). Identification of the abscisic acid receptor VvPYL1 in Vitis vinifera.Plant Biol 14, 244-248. |
| [20] | Ma Y, Szostkiewicz I, Korte A, Moes D, Yang Y, Christmann A, Grill E (2009). Regulators of PP2C phosphatase activity function as abscisic acid sensors.Science 324, 1064-1068. |
| [21] | Madrid KP, Jardim A (2005). Peroxin 5-peroxin 14 association in the protozoan Leishmania donovani involves a novel protein-protein interaction motif.J Biochem 391, 105-114. |
| [22] | Park SY, Fung P, Nishimura N, Jensen DR, Fujii H, Zhao Y, Lumba S, Santiago J, Rodrigues A, Chow TF, Alfred SE, Bonetta D, Finkelstein R, Provart NJ, Desveaux D, Rodriguez PL, McCourt P, Zhu JK, Schroeder JI, Volkman BF, Cutler SR (2009). Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins.Science 324, 1068-1071. |
| [23] | Pierce MM, Raman CS, Nall BT (1999). Isothermal titration calorimetry of protein-protein interactions.Methods 19, 213-221. |
| [24] | Privalov PL, Dragan AI (2007). Microcalorimetry of biological macromolecules.Biophys Chem 126, 16-24. |
| [25] | Santiago J, Rodrigues A, Saez A, Rubio S, Antoni R, Dupeux F, Park SY, Márquez JA, Cutler SR, Rodriguez PL (2009). Modulation of drought resistance by the abscisic acid receptor PYL5 through inhibition of clade A PP2Cs.Plant J 60, 575-588. |
| [26] | Volkova N, Ibrahim V, Hatti-Kaul R (2012). Laccase catalysed oxidation of syringic acid: calorimetric determination of kinetic parameters.Enzyme Microb Technol 50, 233-237. |
| [27] | Wang WS, Ji JJ, Li X, Wang J, Li SS, Pan GH, Fan KQ, Yang KQ (2014). Angucyclines as signals modulate the behaviors of Streptomyces coelicolor.Proc Natl Acad Sci USA 111, 5688-5693. |
| [28] | Wei KF, Wang YM, Zhong XJ, Pan S (2014). Protein kinase structure, expression and regulation in maize drought sig- naling.Mol Breed 34, 583-602. |
| [29] | Zhang XL, Jiang L, Wang GQ, Yu L, Zhang Q, Xin Q, Wu W, Gong ZZ, Chen ZZ (2013). Structural insights into the abscisic acid stereospecificity by the ABA receptors PYR/ PYL/RCAR.PLoS One 8, e67477. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||